Home > Clincal depts. > Department of Urology
Department of Urology
Yoshiyuki Matsui, Motokiyo Komiyama, Eijiro Nakamura, Yasuo Shinoda, Aiko Maejima, Ayumu Matsuda, Hiroyuki Fujimoto
Introduction
In the Department of Urology, all urogenital malignant diseases, including adrenal cancer, kidney cancer, urothelial cancer, prostate cancer, testicular germ cell tumors, and retroperitoneal sarcoma, are the subject of diagnosis and treatment through comprehensive approaches, including radical surgery, irradiation, and chemotherapy
The Team and What We Do
The urology team consists of six staff physicians and three residents. In addition, with the participation of a radiation oncologist, multi-disciplinary treatments for advanced disease, including renal cancer, urothelial cancer, hormone-refractory prostate cancer and metastatic germ cell tumors, are performed. Every morning clinical rounds start at 8: 00 a.m., and a weekly conference (with a medical oncologist, radiologist and pharmacist) to discuss inpatient management is held on Monday evenings. Patient statistics by major treatment are summarized in Table 1.
Major urological malignant diseases are treated according to the following strategies:
1) Renal cell carcinoma - M0: partial (robotic-assisted) or radical nephrectomy. M1: chemotherapy with target drugs with TKI or IO with or without palliative nephrectomy. Selected small (less than 3 cm) tumors: cryotherapy.
2) Bladder cancer - Carcinoma in situ: BCG instillation therapy. Ta, T1: transurethral resection of bladder cancer (TURBT), often combined with preoperative or postoperative BCG instillation. T2-T4: radical cystectomy (robotic-assisted) with neoadjuvant chemotherapy with an M-VAC/GC regimen. N+: systemic chemotherapy, radiation; sometimes urinary diversion alone. M+: chemotherapy with an M-VAC /GC or IO regimen.
3) Prostate cancer - Organ-confined disease: active surveillance, robotic-assisted radical prostatectomy, irradiation, or endocrine therapy. Specimen-confined disease: extended radical prostatectomy without neoadjuvant endocrine therapy, radiation therapy with endocrine therapy, or endocrine therapy alone. For high risk prostate cancer, extended pelvic lymph node dissection using robotics is performed. M1: endocrine therapy and palliative radiation if necessary. For castration refractory disease: docetaxel or cabazitaxel chemotherapy is indicated.
4) Testicular germ cell tumor (GCT) - Stage I: careful observation regardless of a pathological element. Stage II or higher: EP or BEP chemotherapy as the first line. In nonseminomatous cases, a salvage operation is performed after induction chemotherapy. In seminoma cases, careful observation rather than surgery is selected.
Table 1. Patients statistics: Major treatment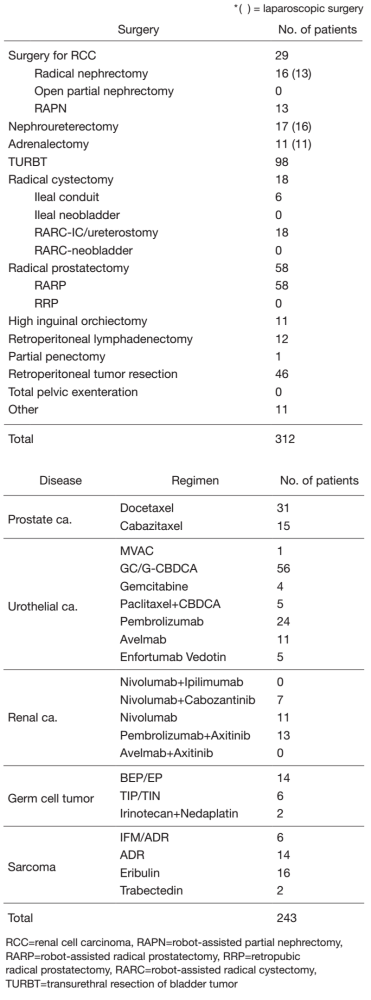
Research activities
We are constantly seeking ways to improve the treatment of malignant urological tumors.
1) Urothelial cancer - A phase III study to confirm the efficacy of pirarubicin in the prevention of bladder recurrence after radical nephroureterectomy for upper tract urothelial carcinoma (JCOG1403) is ongoing. For metastatic disease, IO drugs or enfortumab vedotin has been indicated after standard 1st line chemotherapy.
2) Prostate cancer - A phase II study to evaluate the efficacy of robotic-assisted laparoscopic radical prostatectomy for T1cT3a prostate cancer is no longer recruiting. A new robotic operative method to achieve a complete surgical margin (extended radical prostatectomy) has been developed, and its efficacy in patients with specimen-confined disease has been evaluated without neoadjuvant endocrine therapy. This method was introduced in robotic-assisted laparoscopic radical prostatectomy with extended lymph node dissection.
3) Testicular germ cell tumors (GCTs) - Advanced and/or refractory cases: A so-called "desperate operation", which was designed for patients whose tumor markers do not normalize after induction chemotherapy, has been shown to be both efficacious and clinically significant. For 1st line CDDP-refractory germ cell tumors, a second line TIP/TIN regimen has completed enrollment.
Clinical trials
We are actively involved in the following mainly ongoing protocol studies:
1) A phase III study: A single early intravesical instillation of pirarubicin in the prevention of bladder recurrence after radical nephroureterectomy for upper tract urothelial carcinoma (JCOG1403)
2) A phase III study: Anti PD-L1 antibody (ATEZOLIZUMAB/ MPDL3280A) for muscle invasive bladder cancer
3) An open-label, randomized phase III study: MK-6482 vs Everolimus in advanced renal cell carcinoma progressing after PD-1/L1 and VEGF-targeted therapies.
4) A randomized controlled phase III trial on continued or paused PD-1 pathway blockade for patients with advanced renal cell carcinoma (JCOG1905)
Education
1) Urological residents learn standard laparoscopic surgery to get certified under the urinary laparoscopic technology certification system.
2) Gynecological or colorectal residents are rotating to our department, with the aim of nurturing pelvic surgeons.
Future Prospects
1) To establish a standard protocol for multimodal treatment for complicated urological malignancies or retroperitoneal sarcomas
2) To minimize the invasiveness of treatment through the introduction of robotic or laparoscopic surgery
List of papers published
Journal
1. Kirisawa T, Fukunaga A, Takamori H, Maejima A, Shinoda Y, Komiyama M, Fujimoto H, Yonemori K, Yoshida A, Matsui Y. Cytoreductive robot-assisted prostatectomy for systemic prostate rhabdomyosarcoma presenting as urinary retention. IJU case reports, 5:122-125, 2022
2. Nakajima N, Miyajima A, Shinohara N, Obara W, Kondo T, Kimura G, Kume H, Fujimoto H, Sugiyama T, Nonomura N, Hongo F, Fukumori T, Takahashi M, Kanayama HO, Eto M. Risk factors for recurrence after operation in patients with pT1a renal cell carcinoma: sub-analysis of the multi-institutional national database of the Japanese Urological Association. Japanese journal of clinical oncology, 52:274-280, 2022
3. Inaba K, Tsuchida K, Kashihara T, Umezawa R, Takahashi K, Okuma K, Murakami N, Ito Y, Igaki H, Sumi M, Nakayama Y, Shinoda Y, Hara T, Matsui Y, Komiyama M, Fujimoto H, Itami J. Treatment results of radiotherapy to both the prostate and metastatic sites in patients with bone metastatic prostate cancer. Journal of radiation research, 62:511-516, 2021
4. Fukunaga A, Maejima A, Shinoda Y, Matsui Y, Komiyama M, Fujimoto H, Miyagi Maeshima A. Prognostic implication of staging of seminal vesicle invasion in patients with prostatic adenocarcinoma after prostatectomy. International journal of urology: official journal of the Japanese Urological Association, 28:1039-1045, 2021
5. Hatakeyama S, Narita S, Okita K, Narita T, Iwamura H, Fujita N, Inokuchi J, Matsui Y, Kitamura H, Ohyama C. Management of bladder cancer in older patients. Jpn J Clin Oncol. 2022 Mar;29(3):242-249
6. Nishimura N, Miyake M, Iida K, Miyamoto T, Tomida R, Numakura K, Inokuchi J, Yoneyama T, Matsumura Y, Yajima S, Masuda H, Terada N, Taoka R, Kobayashi T, Kojima T, Matsui Y, Nishiyama N, Kitamura H, Nishiyama H, Fujimoto K. Prognostication in Japanese patients with Bacillus Calmette-Guérin-unresponsive non-muscle-invasive bladder cancer undergoing early radical cystectomy. Int J Urol. 2022 Mar;29(3):242-249.
7. Kato M, Kobayashi T, Matsui Y, Ito K, Hikami K, Yamada T, Ogawa K, Nakamura K, Sassa N, Yokomizo A, Abe T, Tsuchihashi K, Tatarano S, Inokuchi J, Tomida R, Fujiwara M, Takahashi A, Matsumoto K, Shimizu K, Araki H, Kurahashi R, Ozaki Y, Tashiro Y, Uegaki M, Kojima T, Uchida J, Ogawa O, Nishiyama H, Kitamura H; Japan Urological Oncology Group. Impact of the objective response to and number of cycles of platinum-based first-line chemotherapy for metastatic urothelial carcinoma on overall survival of patients treated with pembrolizumab. Int J Urol. 2021;28(12):1261-1267.
8. Mizuno K, Sumiyoshi T, Okegawa T, Terada N, Ishitoya S, Miyazaki Y, Kojima T, Katayama H, Fujimoto N, Hatakeyama S, Shiota M, Yoshimura K, Matsui Y, Narita S, Matsumoto H, Kurahashi R, Kanno H, Ito K, Kimura H, Kamiyama Y, Sunada T, Goto T, Kobayashi T, Yamada H, Tsuchiya N, Kamba T, Matsuyama H, Habuchi T, Eto M, Ohyama C, Ito A, Nishiyama H, Okuno H, Kamoto T, Fujimoto A, Ogawa O, Akamatsu S. Clinical Impact of Detecting Low-Frequency Variants in Cell-Free DNA on Treatment of Castration-Resistant Prostate Cancer. Clin Cancer Res. 2021; 27(22):6164-6173.
