Home > Specialist ctrs & depts. > Clinical Laboratories
Clinical Laboratories
Satoshi Nakajima, Shigeyuki Hasuo, Masahiro Uchikawa, Susumu Wakai, Naoshi Sasaki, Motoi Miyakoshi, Koji Ono, Hiroshi Yamakawa, Ryuzaburo Ohtake, Akashi Koseki, Yoji Hashimoto, Yasuo Shibuki, Hiroki Kakishima, Tomohiro Nakatani, Michi Shoji, Tsutomu Watanabe, Rie Matsuo, Yukie Nakajima, Sachiko Kobayashi, Katsuhide Ikeda, Kazuya Tokita, Satoe Miyaki, Kumiko Nagasaki, Noriko Takahashi, Mizuho Fujima, Daisuke Asahina, Tomoe Ito, Midori Hashimoto, Kaori Ueki, Fumie Watanabe, Akino Chiba, Takako Takada, Kyoko Osanai, Yuko Adegawa, Ruriko Miyake, Asuka Matsunaga, Hiroshi Chigira, Go Sato, Sakiko Yoshimura, Yuu Aruga, Saori Kobayashi, Kaori Yamaguchi, Ryoko Uegaki, Kensyo Kashiwaya, Saori Nakabayashi, Shingo Nakajima, Hideya Matsubayashi, Saeko Shirahama, Akiko Takayanagi, Mei Fukuhara, Kumi Ohashi, Momoko Kitou, Moemi Kasane, Kazuki Ito, Haruki Sasaki, Asuka Takaku, Keiko Arai, Yuri Kurosawa, Megumi Masuda, Yoshiko Shibata, Naomi Fujiki, Ritsuko Tohyama, Chieko Nozawa, Kozaburo Endou, Keiko Mizukoshi, Kiyoaki Nomoto, Masahiko Ushigome, Minami Takahashi, Sachiko Katayama, Shigeru Tamura, Megumi Miura
Routine activities / Research activities
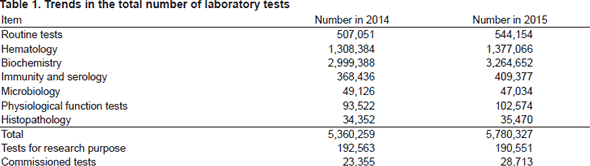
The services of the Clinical Laboratories are organized into five sections: in vitro diagnostics ( routine tests, hematology, biochemistry, immunity and serology, bacteriology, and genetic diagnosis), transfusion and cell therapy testing, blood collection, physiological examination, and histopathology. The Clinical Laboratories have obtained accreditation under ISO 15189, the international standards for quality management and technical competence in medical laboratories, after successful completion of the second surveillance stage. The In Vitro Diagnosis Section has undertaken a complete renewal of hematology and coagulation test systems to improve its ability for timely provision of accurate test results. The Transfusion and Cell Therapy Testing Section is using KM-CART (novel cell-free and concentrated reinfusion therapy) for the management of patients with refractory ascites. It also has renewed the fully automated pre-transfusion testing system, achieving better safety in the management of blood products. The Histopathology Section has been reinforced with the introduction of an automatic thin section machine in response to the increase in the number of specimens, and is preparing it for full-scale operation. The Physiological Examination Section has been experiencing increases in electrocardiography and echocardiography in clinical trials and ultrasonography of lower extremities for the close assessment of deep vein thrombosis. In response, the Section has been refurbished with the renewal of the electrocardiography filing system and respiratory function test equipment, as well as the expansion of circulatory function tests. The Genetic Diagnosis Section has opened the Sysmex Cancer Innovation Laboratory (SCI-Lab), which performs exhaustive gene analysis using the next-generation sequencer (NGS) in collaboration with Sysmex. The total number of laboratory tests in 2015 was 5,780,327, recording a 7% increase from the previous year. In particular, genetic diagnosis tests and physiological examination increased by 9% and 10%, respectively (Tables 1 and 2). The Clinical Laboratories are also cooperating in the promotion of academy-industry-government joint research, clinical trials, and biobank projects.
Research achievement
The Clinical Laboratories are working actively toward the improvement of accuracy and standardization in genetic diagnosis.
Education
The Clinical Laboratories conducted a revision of their original system for accreditation in blood collection, which has been in place since the previous year. The system is being continued with the elongation of the period of training and enrichment of technical training, including education in reception for blood collection, handling of specimens, response to mechanical problems, and dealing with patients. Young technologists are assigned to specialist sections after mastering the basics of laboratory work in the In Vitro Diagnostics Section. A workshop focusing on ISO 15189 was held to ensure the adherence to the quality management system under ISO. Because reinforcement of individual sections is essential to the improvement of the Clinical Laboratories as a whole, monthly study meetings were held and personnel were encouraged to participate in and give presentations at external seminars and academic conferences. As a new initiative, the Clinical Laboratories launched an internship program with universities, aiming to promote the appeal of medical laboratory work.
Future prospects
The Clinical Laboratories intend to construct a system in which medical laboratory technologists play a part in the services of SCI-Lab and participate in the development of advanced medical techniques. In the Microbiology Section, we intend to shorten the time to reporting of test results through the use of a mass spectrometer mainly in blood culture tests, where immediacy is demanded. In ultrasound tests, we intend to establish a system to evaluate the cardiotoxicity of anticancer agents. In response to the increasing number of patients, we strive to shorten waiting time with an emphasis on TAT (turnaround time) from blood collection to analysis and the reporting of results.