HOME > Publication & Reports > Annual Report 2016 > Exploratory Oncology Research & Clinical Trial Center
Division of Experimental Therapeutics
[Kashiwa Campus] Toshihiko Doi, Kiyotaka Yoh, Yoichi Naito, Takahiro Kogawa, Kohei Shitara, Hideaki Takahashi, Yasutoshi Kuboki
[Tsukiji Campus]Noboru Yamamoto, Toshio Shimizu, Yutaka Fujiwara, Kan Yonemori, Kiyoshi Yoshimura, Shigehisa Kitano, Shunsuke Kondo, Satoru Iwasa, Akihiko Shimomura, Takafumi Koyama
Introduction
The Division of Experimental Therapeutics supports efforts toward the creation of new drugs and other products from breakthroughs originating from academic institutions by achieving results at the level of basic research. We evaluate and discover excellent research and development proposals, and provide integrated management of the program so that basic research outcomes are linked through to clinical research and clinical trial for approval. We have conducted several IITs (Investigator Initiated Trials) as applications for approval and also have promoted research and development in the field of medicine.
Our team and what we do
This division plays a key role in the new anti-cancer drug development in Japan as well as in Asia. The top priority is to conduct FIH (First-in-Human) trials, and to also perform phase I trials for solid tumors (i.e., all comers). Recently, we have been joining global phase I trials to accelerate new drug development in Japan. Web- and tel.-conferences are held with EU and US sites, and we discuss about the patient enrollment as well as further developmental strategy. Routine web-conferences are also held between the National Cancer Center (NCC)-Hospital (Tokyo) and the NCC-Hospital East (Chiba) every Friday morning, and we are sharing information about adverse events, patient enrollment, and are referring candidates to each other to accelerate enrollment.
Routine activities
Engaging in medical R&D and establishment /maintenance of medical research environment.
Research activities
We promote the following activities for appropriate conducting of research and trials.
* To plan the clinical trial as IIT for achieving POC (proof of concept) of new academic seeds
* Explanation meetings for researchers and office employees on compliance of laws, ordinances and guidelines
* To consider development design to create a new with the goal of creating innovative drugs, medical devices, as well as ensuring that research projects on promising results proceed more rapidly and in greater depth
Clinical trials
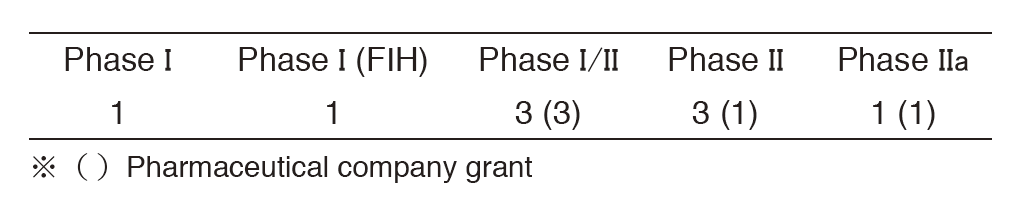
We have conducted several IITs using unapproved drugs from academia and pharmaceutical companies (Table shown below).
List of papers published in 2016
Journal
1.Shitara K, Muro K, Shimada Y, Hironaka S, Sugimoto N, Komatsu Y, Nishina T, Yamaguchi K, Segawa Y, Omuro Y, Tamura T, Doi T, Yukisawa S, Yasui H, Nagashima F, Gotoh M, Esaki T, Emig M, Chandrawansa K, Liepa AM, Wilke H, Ichimiya Y, Ohtsu A. Subgroup analyses of the safety and efficacy of ramucirumab in Japanese and Western patients in RAINBOW: a randomized clinical trial in second-line treatment of gastric cancer. Gastric Cancer, 19:927-938, 2016
2.Mitsunaga S, Ikeda M, Shimizu S, Ohno I, Takahashi H, Okuyama H, Ueno H, Morizane C, Kondo S, Sakamoto Y, Okusaka T, Ochiai A. C-Reactive Protein Level Is an Indicator of the Aggressiveness of Advanced Pancreatic Cancer. Pancreas, 45:110-116, 2016
3.Harano K, Yonemori K, Hirakawa A, Shimizu C, Katsumata N, Gemma A, Fujiwara Y, Tamura K. The influence of familial factors on the choice of the place of death for terminally ill breast cancer patients: a retrospective single-center study. Breast Cancer, 23:797-806, 2016
4.Nokihara H, Yamada Y, Fujiwara Y, Yamamoto N, Wakui H, Nakamichi S, Kitazono S, Inoue K, Harada A, Taube T, Takeuchi Y, Tamura T. Phase I trial of volasertib, a Polo-like kinase inhibitor, in Japanese patients with advanced solid tumors. Invest New Drugs, 34:66-74, 2016
5.Kurihara H, Shimizu C, Miyakita Y, Yoshida M, Hamada A, Kanayama Y, Yonemori K, Hashimoto J, Tani H, Kodaira M, Yunokawa M, Yamamoto H, Watanabe Y, Fujiwara Y, Tamura K. Molecular imaging using PET for breast cancer. Breast Cancer, 23:24-32, 2016
6.Shimizu C, Mogushi K, Morioka MS, Yamamoto H, Tamura K, Fujiwara Y, Tanaka H. Fc-Gamma receptor polymorphism and gene expression of peripheral blood mononuclear cells in patients with HER2-positive metastatic breast cancer receiving single-agent trastuzumab. Breast Cancer, 23:624-632, 2016
7.Sawada T, Watanabe M, Fujimura Y, Yagishita S, Shimoyama T, Maeda Y, Kanda S, Yunokawa M, Tamura K, Tamura T, Minami H, Koh Y, Koizumi F. Sensitive cytometry based system for enumeration, capture and analysis of gene mutations of circulating tumor cells. Cancer Sci, 107:307-314, 2016
8.Shimomura A, Fujiwara Y, Kondo S, Kodaira M, Iwasa S, Kitano S, Tanabe Y, Tamura K, Yamamoto N. Tremelimumab-associated tumor regression following after initial progression: two case reports. Immunotherapy, 8:9-15, 2016
9.Shoji H, Morizane C, Sakamoto Y, Kondo S, Ueno H, Takahashi H, Ohno I, Shimizu S, Mitsunaga S, Ikeda M, Okusaka T. Phase I clinical trial of oral administration of S-1 in combination with intravenous gemcitabine and cisplatin in patients with advanced biliary tract cancer. Jpn J Clin Oncol, 46:132-137, 2016
10.Yamamoto S, Suga K, Maeda K, Maeda N, Yoshimura K, Oka M. Breast sentinel lymph node navigation with three-dimensional computed tomography-lymphography: a 12-year study. Breast Cancer, 23:456-462, 2016
11.Yamashita M, Kitano S, Aikawa H, Kuchiba A, Hayashi M, Yamamoto N, Tamura K, Hamada A. A novel method for evaluating antibody-dependent cell-mediated cytotoxicity by flowcytometry using cryopreserved human peripheral blood mononuclear cells. Sci Rep, 6:19772, 2016
12.Kuboki Y, Yamashita S, Niwa T, Ushijima T, Nagatsuma A, Kuwata T, Yoshino T, Doi T, Ochiai A, Ohtsu A. Comprehensive analyses using next-generation sequencing and immunohistochemistry enable precise treatment in advanced gastric cancer. Ann Oncol, 27:127-133, 2016
13.Seki Y, Fujiwara Y, Kohno T, Takai E, Sunami K, Goto Y, Horinouchi H, Kanda S, Nokihara H, Watanabe S, Ichikawa H, Yamamoto N, Kuwano K, Ohe Y. Picoliter-Droplet Digital Polymerase Chain Reaction-Based Analysis of Cell-Free Plasma DNA to Assess EGFR Mutations in Lung Adenocarcinoma That Confer Resistance to Tyrosine-Kinase Inhibitors. Oncologist, 21:156-164, 2016
14.Takahashi N, Furuta K, Taniguchi H, Sasaki Y, Shoji H, Honma Y, Iwasa S, Okita N, Takashima A, Kato K, Hamaguchi T, Shimada Y, Yamada Y. Serum level of hepatocyte growth factor is a novel marker of predicting the outcome and resistance to the treatment with trastuzumab in HER2-positive patients with metastatic gastric cancer. Oncotarget, 7:4925-4938, 2016
15.Fuse N, Kuboki Y, Kuwata T, Nishina T, Kadowaki S, Shinozaki E, Machida N, Yuki S, Ooki A, Kajiura S, Kimura T, Yamanaka T, Shitara K, Nagatsuma AK, Yoshino T, Ochiai A, Ohtsu A. Prognostic impact of HER2, EGFR, and c-MET status on overall survival of advanced gastric cancer patients. Gastric Cancer, 19:183-191, 2016
16.Ikeda M, Sato A, Mochizuki N, Toyosaki K, Miyoshi C, Fujioka R, Mitsunaga S, Ohno I, Hashimoto Y, Takahashi H, Hasegawa H, Nomura S, Takahashi R, Yomoda S, Tsuchihara K, Kishino S, Esumi H. Phase I trial of GBS-01 for advanced pancreatic cancer refractory to gemcitabine. Cancer Sci, 107:1818-1824, 2016
17.Ikeda M, Yamamoto H, Kaneko M, Oshima H, Takahashi H, Umemoto K, Watanabe K, Hashimoto Y, Ohno I, Mitsunaga S, Okusaka T. Screening rate for hepatitis B virus infection in patients undergoing chemotherapy in Japan. Int J Clin Oncol, 21:1162-1166, 2016
18.Shitara K, Yonesaka K, Denda T, Yamazaki K, Moriwaki T, Tsuda M, Takano T, Okuda H, Nishina T, Sakai K, Nishio K, Tokunaga S, Yamanaka T, Boku N, Hyodo I, Muro K. Randomized study of FOLFIRI plus either panitumumab or bevacizumab for wild-type KRAS colorectal cancer-WJOG 6210G. Cancer Sci, 107:1843-1850, 2016
19.Shitara K, Ohtsu A. Advances in Systemic Therapy for Metastatic or Advanced Gastric Cancer. J Natl Compr Canc Netw, 14:1313-1320, 2016
20.Kasi PM, Kotani D, Cecchini M, Shitara K, Ohtsu A, Ramanathan RK, Hochster HS, Grothey A, Yoshino T. Chemotherapy induced neutropenia at 1-month mark is a predictor of overall survival in patients receiving TAS-102 for refractory metastatic colorectal cancer: a cohort study. BMC Cancer, 16:467, 2016
21.Araki T, Takashima A, Hamaguchi T, Honma Y, Iwasa S, Okita N, Kato K, Yamada Y, Hashimoto H, Taniguchi H, Kushima R, Nakao K, Boku N, Shimada Y. Amrubicin in patients with platinum-refractory metastatic neuroendocrine carcinoma and mixed adenoneuroendocrine carcinoma of the gastrointestinal tract. Anticancer Drugs, 27:794-799, 2016
22.Bun S, Yunokawa M, Ebata T, Shimomura A, Shimoi T, Kodaira M, Yonemori K, Shimizu C, Fujiwara Y, Kato T, Makino Y, Hayashi Y, Tamura K. Feasibility of dose-dense paclitaxel/carboplatin therapy in elderly patients with ovarian, fallopian tube, or peritoneal cancer. Cancer Chemother Pharmacol, 78:745-752, 2016
23.Ebata T, Yunokawa M, Bun S, Shimomura A, Shimoi T, Kodaira M, Yonemori K, Shimizu C, Fujiwara Y, Kato T, Tamura K. Dose-dense paclitaxel plus carboplatin as neoadjuvant chemotherapy for advanced ovarian, fallopian tube, or primary peritoneal carcinomas. Cancer Chemother Pharmacol, 78:1283-1288, 2016
24.Fujiwara K, Shintani D, Nishikawa T. Clear-cell carcinoma of the ovary. Ann Oncol, 27 Suppl 1:i50-i52, 2016
25.Fujiwara Y, Hamada A, Mizugaki H, Aikawa H, Hata T, Horinouchi H, Kanda S, Goto Y, Itahashi K, Nokihara H, Yamamoto N, Ohe Y. Pharmacokinetic profiles of significant adverse events with crizotinib in Japanese patients with ABCB1 polymorphism. Cancer Sci, 107:1117-1123, 2016
26.Fujiwara Y, Tamura K, Kondo S, Tanabe Y, Iwasa S, Shimomura A, Kitano S, Ogasawara K, Turner PK, Mori J, Asou H, Chan EM, Yamamoto N. Phase 1 study of abemaciclib, an inhibitor of CDK 4 and 6, as a single agent for Japanese patients with advanced cancer. Cancer Chemother Pharmacol, 78:281-288, 2016
27.Funakoshi Y, Fujiwara Y, Kiyota N, Mukohara T, Shimada T, Toyoda M, Imamura Y, Chayahara N, Tomioka H, Umezu M, Otsuki N, Nibu K, Minami H. Validity of new methods to evaluate renal function in cancer patients treated with cisplatin. Cancer Chemother Pharmacol, 77:281-288, 2016
28.Harano K, Hirakawa A, Yunokawa M, Nakamura T, Satoh T, Nishikawa T, Aoki D, Ito K, Ito K, Nakanishi T, Susumu N, Takehara K, Watanabe Y, Watari H, Saito T. Optimal cytoreductive surgery in patients with advanced uterine carcinosarcoma: A multi-institutional retrospective study from the Japanese gynecologic oncology group. Gynecol Oncol, 141:447-453, 2016
29.Hatogai K, Kitano S, Fujii S, Kojima T, Daiko H, Nomura S, Yoshino T, Ohtsu A, Takiguchi Y, Doi T, Ochiai A. Comprehensive immunohistochemical analysis of tumor microenvironment immune status in esophageal squamous cell carcinoma. Oncotarget, 7:47252-47264, 2016
30.Horinouchi H, Goto Y, Kanda S, Fujiwara Y, Nokihara H, Yamamoto N, Sumi M, Tamura T, Ohe Y. Candidates for Intensive Local Treatment in cIIIA-N2 Non-Small Cell Lung Cancer: Deciphering the Heterogeneity. Int J Radiat Oncol Biol Phys, 94:155-162, 2016
31.Iwasa S, Yamada Y, Heike Y, Shoji H, Honma Y, Komatsu N, Matsueda S, Yamada A, Morita M, Yamaguchi R, Tanaka N, Kawahara A, Kage M, Shichijo S, Sasada T, Itoh K. Phase I study of a new cancer vaccine of ten mixed peptides for advanced cancer patients. Cancer Sci, 107:590-600, 2016
32.Kanda S, Goto K, Shiraishi H, Kubo E, Tanaka A, Utsumi H, Sunami K, Kitazono S, Mizugaki H, Horinouchi H, Fujiwara Y, Nokihara H, Yamamoto N, Hozumi H, Tamura T. Safety and efficacy of nivolumab and standard chemotherapy drug combination in patients with advanced non-small-cell lung cancer: a four arms phase Ib study. Ann Oncol, 27:2242-2250, 2016
33.Kataoka T, Kiyota N, Shimada T, Funakoshi Y, Chayahara N, Toyoda M, Fujiwara Y, Nibu K, Komori T, Sasaki R, Mukohara T, Minami H. Randomized trial of standard pain control with or without gabapentin for pain related to radiation-induced mucositis in head and neck cancer. Auris Nasus Larynx, 43:677-684, 2016
34.Katsuya Y, Horinouchi H, Asao T, Kitahara S, Goto Y, Kanda S, Fujiwara Y, Nokihara H, Yamamoto N, Watanabe S, Tsuta K, Ohe Y. Expression of programmed death 1 (PD-1) and its ligand (PD-L1) in thymic epithelial tumors: Impact on treatment efficacy and alteration in expression after chemotherapy. Lung Cancer, 99:4-10, 2016
35.Kim Y, Kobayashi E, Kubota D, Suehara Y, Mukaihara K, Akaike K, Ito A, Kaneko K, Chuman H, Kawai A, Kitano S. Reduced argininosuccinate synthetase expression in refractory sarcomas: Impacts on therapeutic potential and drug resistance. Oncotarget, 7:70832-70844, 2016
36.Kimbara S, Kondo S. Immune checkpoint and inflammation as therapeutic targets in pancreatic carcinoma. World J Gastroenterol, 22:7440-7452, 2016
37.Makino Y, Watanabe M, Makihara RA, Nokihara H, Yamamoto N, Ohe Y, Sugiyama E, Sato H, Hayashi Y. Simultaneous optimization of limited sampling points for pharmacokinetic analysis of amrubicin and amrubicinol in cancer patients. Asia Pac J Clin Oncol, 12:259-264, 2016
38.Matsukuma S, Yoshimura K, Ueno T, Oga A, Inoue M, Watanabe Y, Kuramasu A, Fuse M, Tsunedomi R, Nagaoka S, Eguchi H, Matsui H, Shindo Y, Maeda N, Tokuhisa Y, Kawano R, Furuya-Kondo T, Itoh H, Yoshino S, Hazama S, Oka M, Nagano H. Calreticulin is highly expressed in pancreatic cancer stem-like cells. Cancer Sci, 107:1599-1609, 2016
39.Matsuo K, Takazawa Y, Ross MS, Elishaev E, Podzielinski I, Yunokawa M, Sheridan TB, Bush SH, Klobocista MM, Blake EA, Takano T, Matsuzaki S, Baba T, Satoh S, Shida M, Nishikawa T, Ikeda Y, Adachi S, Yokoyama T, Takekuma M, Fujiwara K, Hazama Y, Kadogami D, Moffitt MN, Takeuchi S, Nishimura M, Iwasaki K, Ushioda N, Johnson MS, Yoshida M, Hakam A, Li SW, Richmond AM, Machida H, Mhawech-Fauceglia P, Ueda Y, Yoshino K, Yamaguchi K, Oishi T, Kajiwara H, Hasegawa K, Yasuda M, Kawana K, Suda K, Miyake TM, Moriya T, Yuba Y, Morgan T, Fukagawa T, Wakatsuki A, Sugiyama T, Pejovic T, Nagano T, Shimoya K, Andoh M, Shiki Y, Enomoto T, Sasaki T, Mikami M, Shimada M, Konishi I, Kimura T, Post MD, Shahzad MM, Im DD, Yoshida H, Omatsu K, Ueland FR, Kelley JL, Karabakhtsian RG, Roman LD. Significance of histologic pattern of carcinoma and sarcoma components on survival outcomes of uterine carcinosarcoma. Ann Oncol, 27:1257-1266, 2016
40.Minami H, Ando Y, Ma BBY, Hsiang Lee J, Momota H, Fujiwara Y, Li L, Fukino K, Ito K, Tajima T, Mori A, Lin C-C. Phase I, multicenter, open-label, dose-escalation study of sonidegib in Asian patients with advanced solid tumors. Cancer Sci, 107:1477-1483, 2016
41.Miyasaka A, Nishikawa T, Kozawa E, Yasuda M, Fujiwara K, Hasegawa K. Advanced Mucinous Adenocarcinoma Arising from a Mature Cystic Teratoma: A Case Report and Literature Review. Case Rep Oncol, 9:331-337, 2016
42.Asao T, Fujiwara Y, Sunami K, Kitahara S, Goto Y, Kanda S, Horinouchi H, Nokihara H, Yamamoto N, Ichikawa H, Kohno T, Tsuta K, Watanabe S, Takahashi K, Ohe Y. Medical treatment involving investigational drugs and genetic profile of thymic carcinoma. Lung Cancer, 93:77-81, 2016
43.Mizugaki H, Yamamoto N, Murakami H, Kenmotsu H, Fujiwara Y, Ishida Y, Kawakami T, Takahashi T. Phase I dose-finding study of monotherapy with atezolizumab, an engineered immunoglobulin monoclonal antibody targeting PD-L1, in Japanese patients with advanced solid tumors. Invest New Drugs, 34:596-603, 2016
44.Nagao S, Iwasa N, Kurosaki A, Nishikawa T, Hanaoka T, Hasegawa K, Fujiwara K. The Efficacy of Low-Dose Paclitaxel Added to Combination Chemotherapy of Carboplatin and Gemcitabine or Pegylated Liposomal Doxorubicin. Int J Gynecol Cancer, 26:443-448, 2016
45.Nakamura Y, Kitano S, Takahashi A, Tsutsumida A, Namikawa K, Tanese K, Abe T, Funakoshi T, Yamamoto N, Amagai M, Yamazaki N. Nivolumab for advanced melanoma: pretreatment prognostic factors and early outcome markers during therapy. Oncotarget, 7:77404-77415, 2016
46.Nokihara H, Yamamoto N, Ohe Y, Hiraoka M, Tamura T. Pharmacokinetics of Weekly Paclitaxel and Feasibility of Dexamethasone Taper in Japanese Patients with Advanced Non-small Cell Lung Cancer. Clin Ther, 38:338-347, 2016
47.Sasaki Y, Hamaguchi T, Yamada Y, Takahashi N, Shoji H, Honma Y, Iwasa S, Okita N, Takashima A, Kato K, Nagai Y, Taniguchi H, Boku N, Ushijima T, Shimada Y. Value of KRAS, BRAF, and PIK3CA Mutations and Survival Benefit from Systemic Chemotherapy in Colorectal Peritoneal Carcinomatosis. Asian Pac J Cancer Prev, 17:539-543, 2016
48.Nomura M, Iwasa S, Tsushima T, Kato K, Yasui H, Boku N, Muto M, Muro K. Active salvage chemotherapy versus best supportive care for patients with recurrent or metastatic squamous cell carcinoma of the esophagus refractory or intolerable to fluorouracil, platinum, and taxane. Cancer Chemother Pharmacol, 78:1209-1216, 2016
49.Nonagase Y, Okamoto K, Iwasa T, Yoshida T, Tanaka K, Takeda M, Kaneda H, Shimizu T, Tsurutani J, Nakagawa K. Afatinib-refractory brain metastases from EGFR-mutant non-small-cell lung cancer successfully controlled with erlotinib: a case report. Anticancer Drugs, 27:251-253, 2016
50.Ogiya R, Niikura N, Kumaki N, Bianchini G, Kitano S, Iwamoto T, Hayashi N, Yokoyama K, Oshitanai R, Terao M, Morioka T, Tsuda B, Okamura T, Saito Y, Suzuki Y, Tokuda Y. Comparison of tumor-infiltrating lymphocytes between primary and metastatic tumors in breast cancer patients. Cancer Sci, 107:1730-1735, 2016
51.Okuma HS, Koizumi F, Hirakawa A, Nakatochi M, Komori O, Hashimoto J, Kodaira M, Yunokawa M, Yamamoto H, Yonemori K, Shimizu C, Fujiwara Y, Tamura K. Clinical and microarray analysis of breast cancers of all subtypes from two prospective preoperative chemotherapy studies. Br J Cancer, 115:411-419, 2016
52.Okuma HS, Kondo S. Trends in the development of MET inhibitors for hepatocellular carcinoma. Future Oncol, 12:1275-1286, 2016
53.Sasada S, Kodaira M, Shimoi T, Shimomura A, Yunokawa M, Yonemori K, Shimizu C, Fujiwara Y, Tamura K. Ifosfamide and Etoposide Chemotherapy in the Treatment of Recurrent/Refractory Rhabdomyosarcoma in Adults. Anticancer Res, 36:2429-2432, 2016
54.Satake H, Tahara M, Mochizuki S, Kato K, Hara H, Yokota T, Kiyota N, Kii T, Chin K, Zenda S, Kojima T, Bando H, Yamazaki T, Iwasa S, Honma Y, Hamauchi S, Tsushima T, Ohtsu A. A prospective, multicenter phase I/II study of induction chemotherapy with docetaxel, cisplatin and fluorouracil (DCF) followed by chemoradiotherapy in patients with unresectable locally advanced esophageal carcinoma. Cancer Chemother Pharmacol, 78:91-99, 2016
55.Shiba S, Morizane C, Hiraoka N, Sasaki M, Koga F, Sakamoto Y, Kondo S, Ueno H, Ikeda M, Yamada T, Shimada K, Kosuge T, Okusaka T. Pancreatic neuroendocrine tumors: A single-center 20-year experience with 100 patients. Pancreatology, 16:99-105, 2016
56.Shigeta K, Kosaka T, Kitano S, Yasumizu Y, Miyazaki Y, Mizuno R, Shinojima T, Kikuchi E, Miyajima A, Tanoguchi H, Hasegawa S, Oya M. High Absolute Monocyte Count Predicts Poor Clinical Outcome in Patients with Castration-Resistant Prostate Cancer Treated with Docetaxel Chemotherapy. Ann Surg Oncol, 23:4115-4122, 2016
57.Shimizu T, Fukuoka K, Takeda M, Iwasa T, Yoshida T, Horobin J, Keegan M, Vaickus L, Chavan A, Padval M, Nakagawa K. A first-in-Asian phase 1 study to evaluate safety, pharmacokinetics and clinical activity of VS-6063, a focal adhesion kinase (FAK) inhibitor in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol, 77:997-1003, 2016
58.Shimizu T, Seto T, Hirai F, Takenoyama M, Nosaki K, Tsurutani J, Kaneda H, Iwasa T, Kawakami H, Noguchi K, Shimamoto T, Nakagawa K. Phase 1 study of pembrolizumab (MK-3475; anti-PD-1 monoclonal antibody) in Japanese patients with advanced solid tumors. Invest New Drugs, 34:347-354, 2016
59.Tada K, Kitano S, Shoji H, Nishimura T, Shimada Y, Nagashima K, Aoki K, Hiraoka N, Honma Y, Iwasa S, Okita N, Takashima A, Kato K, Yamada Y, Katayama N, Boku N, Heike Y, Hamaguchi T. Pretreatment Immune Status Correlates with Progression-Free Survival in Chemotherapy-Treated Metastatic Colorectal Cancer Patients. Cancer Immunol Res, 4:592-599, 2016
60.Takahashi N, Iwasa S, Fukahori M, Sudo K, Sasaki Y, Shoji H, Honma Y, Okita NT, Takashima A, Hamaguchi T, Boku N, Shimada Y, Honda K, Yamada T, Yamada Y. A phase I study of the combination of panitumumab and bevacizumab in KRAS wild-type colorectal cancer patients previously treated with fluoropyrimidine, oxaliplatin, irinotecan and bevacizumab. Cancer Chemother Pharmacol, 78:567-575, 2016
61.Takahashi N, Iwasa S, Sasaki Y, Shoji H, Honma Y, Takashima A, Okita NT, Kato K, Hamaguchi T, Yamada Y. Serum levels of soluble programmed cell death ligand 1 as a prognostic factor on the first-line treatment of metastatic or recurrent gastric cancer. J Cancer Res Clin Oncol, 142:1727-1738, 2016
62.Takahashi N, Iwasa S, Taniguchi H, Sasaki Y, Shoji H, Honma Y, Takashima A, Okita N, Kato K, Hamaguchi T, Shimada Y, Yamada Y. Prognostic role of ERBB2, MET and VEGFA expression in metastatic colorectal cancer patients treated with anti-EGFR antibodies. Br J Cancer, 114:1003-1011, 2016
63.Tamura K, Hashimoto J, Tanabe Y, Kodaira M, Yonemori K, Seto T, Hirai F, Arita S, Toyokawa G, Chen L, Yamamoto H, Kawata T, Lindemann J, Esaki T. Safety and tolerability of AZD5363 in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol, 77:787-795, 2016
64.Tamura K, Mukai H, Naito Y, Yonemori K, Kodaira M, Tanabe Y, Yamamoto N, Osera S, Sasaki M, Mori Y, Hashigaki S, Nagasawa T, Umeyama Y, Yoshino T. Phase I study of palbociclib, a cyclin-dependent kinase 4/6 inhibitor, in Japanese patients. Cancer Sci, 107:755-763, 2016
65.Tamura Y, Nokihara H, Honda K, Tanabe Y, Asahina H, Yamada Y, Enatsu S, Kurek R, Yamamoto N, Tamura T. Phase I study of the second-generation, recombinant, human EGFR antibody necitumumab in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol, 78:995-1002, 2016
66.Doi T, Lee K-H, Kim T-M, Ohtsu A, Kim TY, Ikeda M, Yoh K, Gallo Stampino C, Hirohashi T, Suzuki A, Fujii Y, Andrew Williams J, Bang Y-J. A phase I study of the human anti-activin receptor-like kinase 1 antibody PF-03446962 in Asian patients with advanced solid tumors. Cancer Med, 5:1454-1463, 2016
67.Tanabe Y, Ichikawa H, Kohno T, Yoshida H, Kubo T, Kato M, Iwasa S, Ochiai A, Yamamoto N, Fujiwara Y, Tamura K. Comprehensive screening of target molecules by next-generation sequencing in patients with malignant solid tumors: guiding entry into phase I clinical trials. Mol Cancer, 15:73, 2016
68.Doi T, Shitara K, Kojima T, Yoshino T, Dontabhaktuni A, Rebscher H, Tang S, Cosaert J, Ohtsu A. A phase I study evaluating cixutumumab, a type 1 insulin-like growth factor receptor inhibitor, given every 2 or 3 weeks in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol, 77:1253-1262, 2016
69.Tanaka R, Yonemori K, Hirakawa A, Kinoshita F, Takahashi N, Hashimoto J, Kodaira M, Yamamoto H, Yunokawa M, Shimizu C, Fujimoto M, Fujiwara Y, Tamura K. Risk Factors for Developing Skeletal-Related Events in Breast Cancer Patients With Bone Metastases Undergoing Treatment With Bone-Modifying Agents. Oncologist, 21:508-513, 2016
70.Yoh K, Doi T, Ohmatsu H, Kojima T, Takahashi H, Zenke Y, Wacheck V, Enatsu S, Nakamura T, Turner K, Uenaka K. A phase I dose-escalation study of LY2875358, a bivalent MET antibody, given as monotherapy or in combination with erlotinib or gefitinib in Japanese patients with advanced malignancies. Invest New Drugs, 34:584-595, 2016
71.Watanabe S, Takeda M, Takahama T, Iwasa T, Tsurutani J, Tanizaki J, Shimizu T, Sakai K, Wada Y, Isogai N, Nishio K, Nakagawa K. Successful human epidermal growth receptor 2-targeted therapy beyond disease progression for extramammary Paget's disease. Invest New Drugs, 34:394-396, 2016
72.Yoh K, Hosomi Y, Kasahara K, Yamada K, Takahashi T, Yamamoto N, Nishio M, Ohe Y, Koue T, Nakamura T, Enatsu S, Lee P, Ferry D, Tamura T, Nakagawa K. A randomized, double-blind, phase II study of ramucirumab plus docetaxel vs placebo plus docetaxel in Japanese patients with stage IV non-small cell lung cancer after disease progression on platinum-based therapy. Lung Cancer, 99:186-193, 2016
73.Yamaguchi T, Iwasa S, Nagashima K, Ikezawa N, Hamaguchi T, Shoji H, Honma Y, Takashima A, Okita N, Kato K, Yamada Y, Shimada Y. Comparison of Panitumumab Plus Irinotecan and Cetuximab Plus Irinotecan for KRAS Wild-type Metastatic Colorectal Cancer. Anticancer Res, 36:3531-3536, 2016
74.Kogawa T, Fouad TM, Liu DD, Wu J, Shen Y, Masuda H, Fujii T, Chavez-MacGregor M, Alvarez RH, Hortobagyi GN, Valero V, Ueno NT. High HER2/Centromeric Probe for Chromosome 17 Fluorescence In Situ Hybridization Ratio Predicts Pathologic Complete Response and Survival Outcome in Patients Receiving Neoadjuvant Systemic Therapy With Trastuzumab for HER2-Overexpressing Locally Advanced Breast Cancer. Oncologist, 21:21-27, 2016
75.Nagatani Y, Shitara K, Bando H, Kuboki Y, Okamoto W, Kojima T, Yoshino T, Nishida T, Ohtsu A, Doi T. Clinical outcomes of patients with gastrointestinal stromal tumor in phase I clinical trials. BMC Cancer, 16:889, 2016
76.Bando H, Doi T, Muro K, Yasui H, Nishina T, Yamaguchi K, Takahashi S, Nomura S, Kuno H, Shitara K, Sato A, Ohtsu A. A multicenter phase II study of TAS-102 monotherapy in patients with pre-treated advanced gastric cancer (EPOC1201). Eur J Cancer, 62:46-53, 2016
77.Doi A, Sumiyoshi T, Omori Y, Oyamada Y, Kumano K, Yoshizaki N, Hirayama M, Suzuki Y, Okushiba S, Kogawa T, Doi T, Kondo H. Double Extramedullary Plasmacytoma of the Stomach with a Long-term Endoscopic Follow-up. Intern Med, 55:3585-3590, 2016
78.Hatake K, Doi T, Uetake H, Takahashi Y, Ishihara Y, Shirao K. Bevacizumab safety in Japanese patients with colorectal cancer. Jpn J Clin Oncol, 46:234-240, 2016
79.Yonemori K, Hirakawa A, Kawachi A, Kinoshita F, Okuma H, Nishikawa T, Tamura K, Fujiwara Y, Takebe N. Drug induced interstitial lung disease in oncology phase I trials. Cancer Sci, 107:1830-1836, 2016
80.Nishida T, Doi T. Pazopanib for both GIST and soft-tissue sarcoma. Lancet Oncol, 17:549-550, 2016
81.Yonemori K, Tamura K, Kodaira M, Fujikawa K, Sagawa T, Esaki T, Shirakawa T, Hirai F, Yokoi Y, Kawata T, Hatano B, Takahashi Y. Safety and tolerability of the olaparib tablet formulation in Japanese patients with advanced solid tumours. Cancer Chemother Pharmacol, 78:525-531, 2016
82.Yoshino T, Kojima T, Bando H, Yamazaki T, Naito Y, Mukai H, Fuse N, Goto K, Ito Y, Doi T, Ohtsu A. Effect of food on the pharmacokinetics of TAS-102 and its efficacy and safety in patients with advanced solid tumors. Cancer Sci, 107:659-665, 2016
83.Saruwatari K, Umemura S, Nomura S, Kirita K, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Ohe Y, Goto K. Prognostic Factor Analysis in Patients With Small-Cell Lung Cancer Treated With Third-Line Chemotherapy. Clin Lung Cancer, 17:581-587, 2016
84.Zenke Y, Umemura S, Sugiyama E, Kirita K, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Goto K. Successful treatment with afatinib after grade 3 hepatotoxicity induced by both gefitinib and erlotinib in EGFR mutation-positive non-small cell lung cancer. Lung Cancer, 99:1-3, 2016
85.Zenke Y, Umemura S, Motegi A, Furukawa K, Kirita K, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Tsuboi M, Akimoto T, Goto K. Acute and Progressive Tracheal Stenosis after Proton Beam Therapy with Concurrent Chemotherapy for Non-Small Cell Lung Cancer. J Thorac Oncol, 11:1181-1183, 2016
86.Zenke Y, Yoh K, Matsumoto S, Umemura S, Niho S, Ohmatsu H, Goto K, Ohe Y. Clinical Impact of Gastric Acid-Suppressing Medication Use on the Efficacy of Erlotinib and Gefitinib in Patients With Advanced Non-Small-Cell Lung Cancer Harboring EGFR Mutations. Clin Lung Cancer, 17:412-418, 2016
87.Tamiya M, Tamiya A, Kaneda H, Nakagawa K, Yoh K, Goto K, Okamoto H, Shimokawa T, Abe T, Tanaka H, Daga H, Takeda K, Hirashima T, Atagi S. A phase II study of pemetrexed plus carboplatin followed by maintenance pemetrexed as first-line chemotherapy for elderly patients with advanced non-squamous non-small cell lung cancer. Med Oncol, 33:2, 2016
88.Niho S, Nokihara H, Nihei K, Akimoto T, Sumi M, Ito Y, Yoh K, Goto K, Ohmatsu H, Horinouchi H, Yamamoto N, Sekine I, Kubota K, Ohe Y, Tamura T. Dose-Escalation Study of Thoracic Radiotherapy in Combination With Pemetrexed Plus Cisplatin in Japanese Patients With Locally Advanced Nonsquamous Non-Small Cell Lung Cancer: A Post Hoc Analysis of Survival and Recurrent Sites. Am J Clin Oncol, 39:132-135, 2016
89.Mukai H, Saeki T, Aogi K, Naito Y, Matsubara N, Shigekawa T, Ueda S, Takashima S, Hara F, Yamashita T, Ohwada S, Sasaki Y. Patritumab plus trastuzumab and paclitaxel in human epidermal growth factor receptor 2-overexpressing metastatic breast cancer. Cancer Sci, 107:1465-1470, 2016
90.Hisakane K, Saruwatari K, Fujii S, Kirita K, Umemura S, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Kuwata T, Ochiai A, Gemma A, Tsuboi M, Goto K, Ishii G. Unique intravascular tumor microenvironment predicting recurrence of lung squamous cell carcinoma. J Cancer Res Clin Oncol, 142:593-600, 2016
91.Shimomura A, Shiino S, Kawauchi J, Takizawa S, Sakamoto H, Matsuzaki J, Ono M, Takeshita F, Niida S, Shimizu C, Fujiwara Y, Kinoshita T, Tamura K, Ochiya T. Novel combination of serum microRNA for detecting breast cancer in the early stage. Cancer Sci, 107:326-334, 2016
92.Saruwatari K, Ikemura S, Sekihara K, Kuwata T, Fujii S, Umemura S, Kirita K, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Ochiai A, Kohrogi H, Tsuboi M, Goto K, Ishii G. Aggressive tumor microenvironment of solid predominant lung adenocarcinoma subtype harboring with epidermal growth factor receptor mutations. Lung Cancer, 91:7-14, 2016
93.Matsuzawa R, Kirita K, Kuwata T, Umemura S, Matsumoto S, Fujii S, Yoh K, Kojima M, Niho S, Ohmatsu H, Ochiai A, Tsuboi M, Goto K, Ishii G. Factors influencing the concordance of histological subtype diagnosis from biopsy and resected specimens of lung adenocarcinoma. Lung Cancer, 94:1-6, 2016
94.Miura N, Kamita M, Kakuya T, Fujiwara Y, Tsuta K, Shiraishi H, Takeshita F, Ochiya T, Shoji H, Huang W, Ohe Y, Yamada T, Honda K. Efficacy of adjuvant chemotherapy for non-small cell lung cancer assessed by metastatic potential associated with ACTN4. Oncotarget, 7:33165-33178, 2016
95.Honma Y, Yamada Y, Terazawa T, Takashima A, Iwasa S, Kato K, Hamaguchi T, Shimada Y, Ohashi M, Morita S, Fukagawa T, Machida N, Katai H. Feasibility of neoadjuvant S-1 and oxaliplatin followed by surgery for resectable advanced gastric adenocarcinoma. Surg Today, 46:1076-1082, 2016
96.Hatogai K, Yano T, Kojima T, Onozawa M, Fujii S, Daiko H, Yoda Y, Hombu T, Doi T, Kaneko K, Ohtsu A. Local efficacy and survival outcome of salvage endoscopic therapy for local recurrent lesions after definitive chemoradiotherapy for esophageal cancer. Radiat Oncol, 11:31, 2016
97.Hatogai K, Fujii S, Kojima T, Daiko H, Kadota T, Fujita T, Yoshino T, Doi T, Takiguchi Y, Ohtsu A. Prognostic significance of tumor regression grade for patients with esophageal squamous cell carcinoma after neoadjuvant chemotherapy followed by surgery. J Surg Oncol, 113:390-396, 2016
98.Hatogai K, Yano T, Kojima T, Onozawa M, Daiko H, Nomura S, Yoda Y, Doi T, Kaneko K, Ohtsu A. Salvage photodynamic therapy for local failure after chemoradiotherapy for esophageal squamous cell carcinoma. Gastrointest Endosc, 83:1130-1139 e1133, 2016
99.Kotani D, Shitara K, Kawazoe A, Fukuoka S, Kuboki Y, Bando H, Okamoto W, Kojima T, Doi T, Ohtsu A, Yoshino T. Safety and Efficacy of Trifluridine/Tipiracil Monotherapy in Clinical Practice for Patients With Metastatic Colorectal Cancer: Experience at a Single Institution. Clin Colorectal Cancer, 15:e109-115, 2016