HOME > Publication & Reports > Annual Report 2016 > Hospital East
Department of Pathology and Clinical Laboratories / Clinical Laboratories
Takeshi Kuwata, Masato Sugano, Genichiro Ishii, Satoshi Fujii, Motohiro Kojima, Eiichi Yoshikawa, Shigehisa Yoshida, Masahiro Karibe, Seiji Iwasaki, Miki Goto, Masaki Takeda, Hiromi Kimura, Yasuharu Hashimoto, Yukihiro Okano, Mari Hisano, Mitunori Tajima, Mika Sasanuma, Aya Koike, Takuya Yamaguchi, Takuya Aiba, Keiko Nakai, Ayumi Setsuta, Mayumi Kasai, Ayumi Nakanishi, Emi Hasegawa, Masahiro Inoue, Yasuko Yoshihara, Izumi Suzuki, Kazumi Yamaguchi, Sayuri Shibayama, Ikuko Tkahashi, Saki Nakamura, Kazuki Motohashi, Chihiro Kodama, Yukina Fukuda, Eriko Iwamoto, Yasuteru Yamagishi, Nagisa Bouno, Kazumi Tamura, Tomomi Sekine, Masayuki Ito, Rie Kuroiwa, Michiko Iida, Yuki Soeda, Natsuki Nagamine, Takuya Hanazawa, Emiko Yoshikawa, Emi Imamiya, Yoshiko Ohtake, Megumi Yamaguchi, Miwa Yamada
Introduction
The Department of Pathology and Clinical Laboratories (DPCL) has two divisions; the Pathology Division (PD) and the Clinical Laboratory Division (CLD). Both divisions play a fundamental role in routine hospital services and support research activities at the National Cancer Center Hospital East (NCCHE).
The DPCL received ISO15189:2007 accreditation in 2012, and successfully transited to the newest version (ISO15189:2012) in 2014, ensuring quality control and quality assurance of the testing, including the one for clinical trials, performed in the departments. In 2015, two sections, Physiology and Supporting laboratory testing in clinical studies, received ISO15189:2012, ensuring the quality control and quality assurance of the testing, including the one for clinical trials, performed in the departments with global standard.
Routine activities
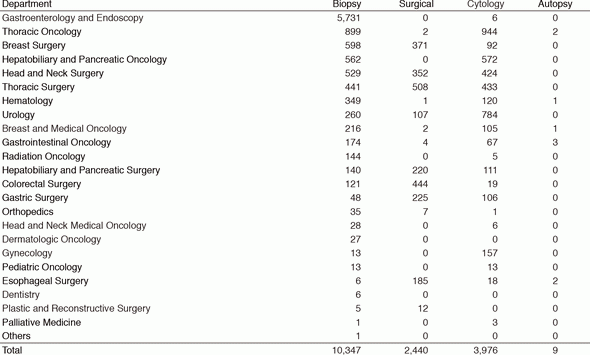
Primarily, the routine activity at the PD is surgical pathology. The Number of samples examined in our department in 2016 is listed in Table 1.
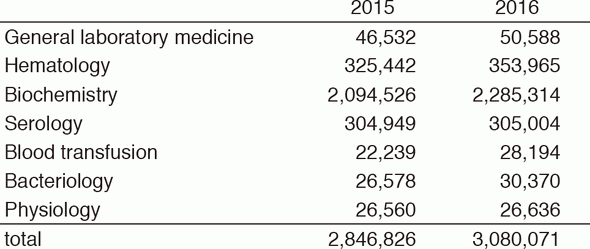
The CLD consists of six sections; i) General Laboratory Medicine, ii) Hematology, iii) Biochemistry/Serology, iv) Physiology, v) Bacteriology, and vi) Blood Transfusion. The numbers of test performed in each section are listed in Tables 2 and 3. The total number of tests performed in the DPCL in 2016 increased by 8.2% compared with the previous year; including 26.8% and 14.3% increase in Blood Transfusion and Bacteriology sections, respectively.
Research activities
All of the pathologists were involved in research activities at the Exploratory Oncology Research & Clinical Trial Center (EPOC). All the technologists working in our department are also highly motivated to develop advanced diagnostic technologies and various results are presented in several meetings.
Clinical trials
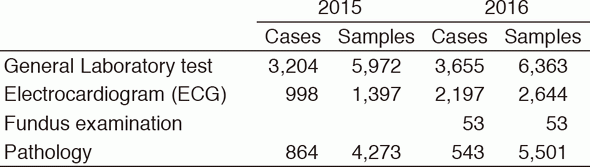
Practically, the CLD participated in all of the clinical trials operated at the NCCHE by providing laboratory data. The section for supporting laboratory testing in clinical studies was transferred to the DPCL in June 2014. This section, coordinating with the pathology and physiology sections, reinforces quality control and quality assurance for clinical tests performed in clinical trials at the NCCHE.
Education
Clinicopathological conferences are held regularly with each clinical department/section. In the PD, conference-style training sessions are open weekly for the residents.
Future prospects
Pathological diagnosis and laboratory tests play a fundamental role not only in routine hospital work but also in medical research. As an ISO15189-certified clinical laboratory, the DPCL will be continuously involved in investigating new diagnostic technologies, developing new drugs, and conducting translational /clinical researches in the NCCHE.
Table 1. Number of pathology and cytology samples examined at Pathology Division in 2016
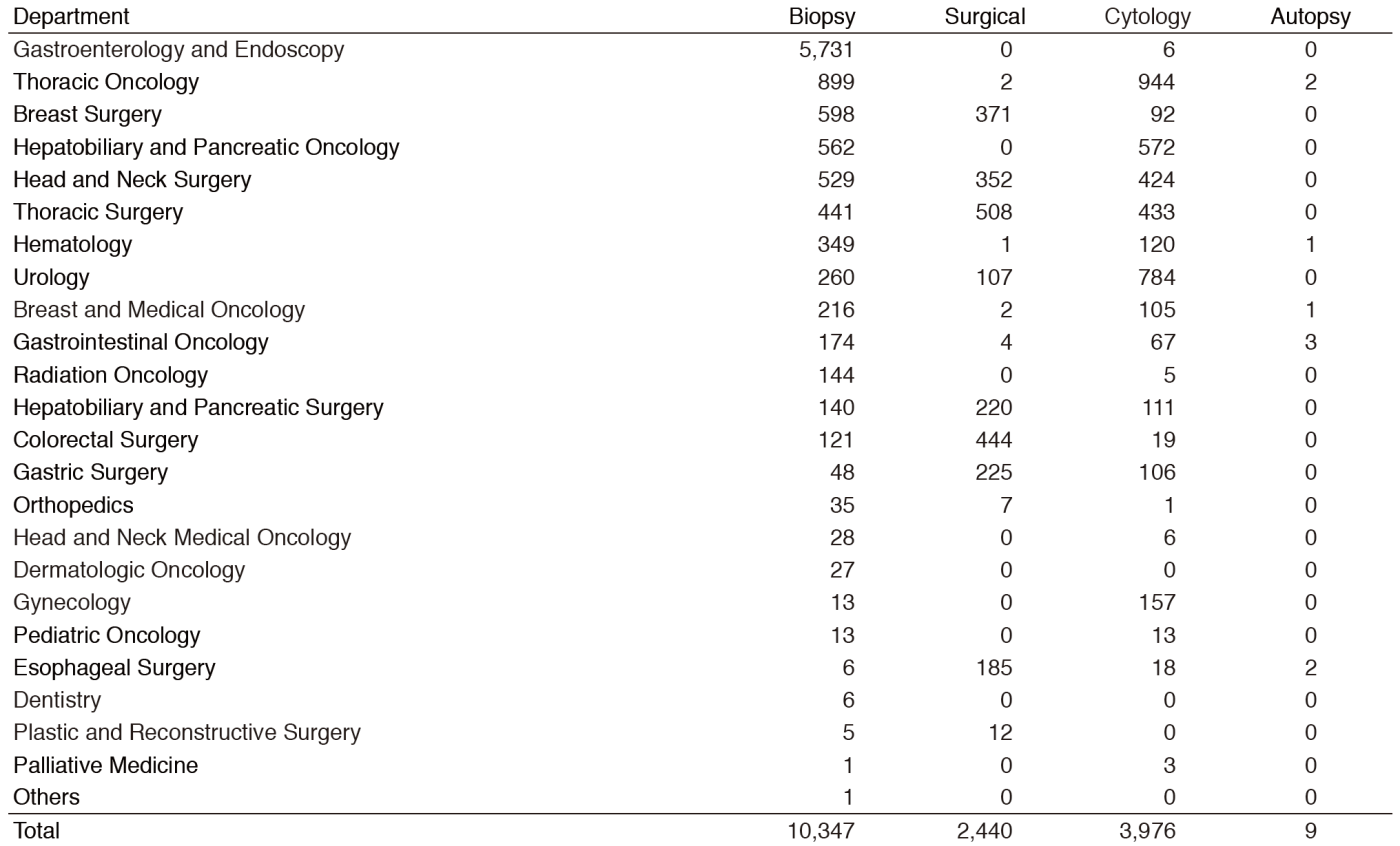
Table 2. Number of laboratory tests exmined in the Clinical Laboratory Division in 2015 & 2016
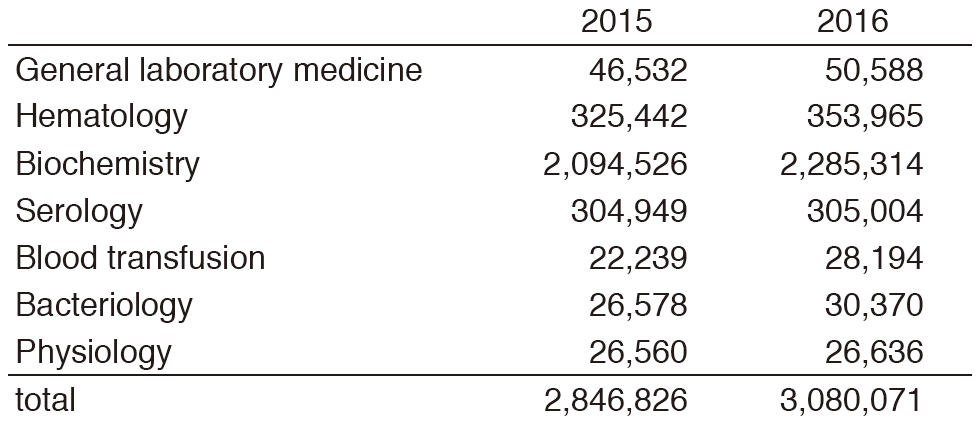
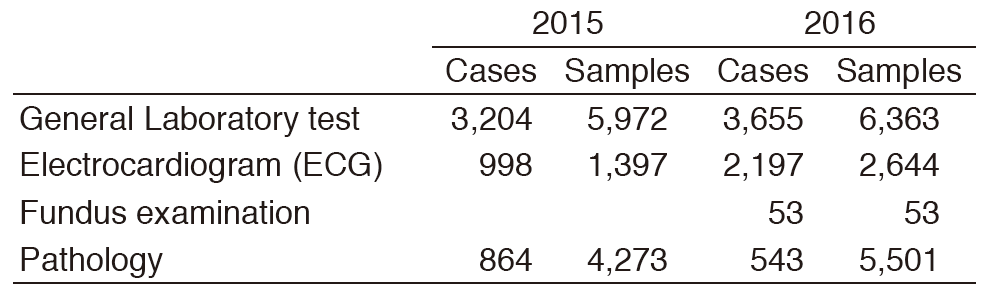
Table 3. Number of cases and samples prepared in the Clinical Laboratory Division for clinical trials in 2015 & 2016
List of papers published in 2016
Journal
1.Kuboki Y, Yamashita S, Niwa T, Ushijima T, Nagatsuma A, Kuwata T, Yoshino T, Doi T, Ochiai A, Ohtsu A. Comprehensive analyses using next-generation sequencing and immunohistochemistry enable precise treatment in advanced gastric cancer. Ann Oncol, 27:127-133, 2016
2.Yokoyama T, Nakatake M, Kuwata T, Couzinet A, Goitsuka R, Tsutsumi S, Aburatani H, Valk PJM, Delwel R, Nakamura T. MEIS1-mediated transactivation of synaptotagmin-like 1 promotes CXCL12/CXCR4 signaling and leukemogenesis. J Clin Invest, 126:1664-1678, 2016
3.Hisakane K, Saruwatari K, Fujii S, Kirita K, Umemura S, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Kuwata T, Ochiai A, Gemma A, Tsuboi M, Goto K, Ishii G. Unique intravascular tumor microenvironment predicting recurrence of lung squamous cell carcinoma. J Cancer Res Clin Oncol, 142:593-600, 2016
4.Sekihara K, Hishida T, Ikemura S, Saruwatari K, Morise M, Kuwata T, Fujii S, Kojima M, Ochiai A, Funai K, Aokage K, Yoshida J, Tsuboi M, Ishii G. The association of intravascular stromal cells with prognosis in high-grade neuroendocrine carcinoma of the lung. J Cancer Res Clin Oncol, 142:905-912, 2016
5.Saruwatari K, Ikemura S, Sekihara K, Kuwata T, Fujii S, Umemura S, Kirita K, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Ochiai A, Kohrogi H, Tsuboi M, Goto K, Ishii G. Aggressive tumor microenvironment of solid predominant lung adenocarcinoma subtype harboring with epidermal growth factor receptor mutations. Lung Cancer, 91:7-14, 2016
6.Matsuzawa R, Kirita K, Kuwata T, Umemura S, Matsumoto S, Fujii S, Yoh K, Kojima M, Niho S, Ohmatsu H, Ochiai A, Tsuboi M, Goto K, Ishii G. Factors influencing the concordance of histological subtype diagnosis from biopsy and resected specimens of lung adenocarcinoma. Lung Cancer, 94:1-6, 2016
7.Suzuki S, Aokage K, Hishida T, Yoshida J, Kuwata T, Yamauchi C, Tsuboi M, Ishii G. Interstitial growth as an aggressive growth pattern in primary lung cancer. J Cancer Res Clin Oncol, 142:1591-1598, 2016
8.Naito M, Aokage K, Saruwatari K, Hisakane K, Miyoshi T, Hishida T, Yoshida J, Masato S, Kojima M, Kuwata T, Fujii S, Ochiai A, Sato Y, Tsuboi M, Ishii G. Microenvironmental changes in the progression from adenocarcinoma in situ to minimally invasive adenocarcinoma and invasive lepidic predominant adenocarcinoma of the lung. Lung Cancer, 100:53-62, 2016