HOME > Publication & Reports > Annual Report 2016 > Sections Directed by President
Center For Research Administration And Support(CRAS)
Kiyohito Nakai, Masafumi Shimokawa, Nobuko Ushirozawa, Ayako Yamashita, Tomoki Yamada, Namiko Aoshima, Fumie Suzuki, Yuichi Amanuma, Shou Kobayashi, Jinko Sato, Kiyoka Nagayama, Shunichi Magara, Yuko Yamada, Atsushi Okazaki, Masahiro Kusuda, Takako Kuwaki, Shizuo Ao, Kazunori Aoki, Masaki Yamamoto, Genta Oono, Yuji Shinoda, Yuko Kineri, Hiromi Nakagawa, Yuichi Nakamura, Ryo Yaguchi, Toshihiro Tanaka, Chieko Yano, Ryoko Noda, Haruka Nakada, Taro Shibata, Aya Kuchiba, Shogo Nomura, Junki Mizusawa, Masashi Wakabayashi, Megumi Kubo, Yasue Kojima, Noriko Yamashita, Rika Ohmi, Yayoi Oofuji, Masahiko Ozaki, Motonori Kakei, Yumiko Shirai, Kuniko Takahashi, Yuki Harada, Mie Yamada, Harumi Maruno, Tomoko Ishida, Junko Okuaki, Natsuko Takagi, Nanako Takatera, Sayaka Tada, Satoko Tabata, Harumi Nakajima, Mai Hashimoto, Junko Horie, Kaori Matsushita, Shimon Tashiro, Akiko Nakada, Mao Shikano, Mari Hirata, Teruhiko Yoshida
Introduction
The Center for Research Administration and Support (CRAS) was established on July 16, 2014. The starting members of the CRAS were approximately 160 staff, who together offer diverse functions and specialties, ranging from research fund administrations, alliance with private sectors, intellectual properties, clinical research coordinators and data managers, monitoring and audit, biostatistics support, offices for research ethics (IRB), and COI committees.
The background and the purpose of the creation of the CRAS was explained by Dr. Tomomitsu Hotta, the President of the National Cancer Center (NCC), in the NCC News 2014 Vol.5/No.3 (in Japanese). Briefly, since its foundation in 1962, the NCC has added several new segments and organizations to evolve as a comprehensive cancer center. Because each segment needed its own research infrastructure, support activities in the NCC had become fragmented and scattered with the possibility of gaps and redundancies. Dr. Hotta asked the Strategic Planning Bureau and assembled the "NCC New Vision" in 2014, in which he proposed integration and communications of various research support functions in the NCC. The CRAS was created in response to the 2014 Vision.
In 2015, the NCC Hospital (NCCH) and the NCC Hospital East (NCCHE) were certified as the Core Clinical Research Hospital under the Medical Care Act in August and September, respectively. It is then required that the support functions for the clinical research, especially those concerning clinical trials, need to be operated under the explicit responsibilities of a hospital director. As a result, the governance of the Research Coordination Division, Research Promotion Division and Regulatory Science Section of the CRAS have now moved to the Clinical Research Support Offices, which belong to the common departments of each hospital. In January 2017, these divisions and section will be officially separated from the CRAS in the NCC organization, and their 2016 annual reports are found in the respective hospital chapters.
(Future prospects)
The NCC has embarked for the new era under the leadership of newly appointed President and Directors of the NCC Research Institute (NCCRI) and both hospitals (NCCH, NCCHE) on April 1, 2016. The Vision and high-priority research targets have been redefined, and re-organization is in progress in various parts of the NCC. However, the core concept of the CRAS stays unchanged to contribute to the NCC mission by promoting organic unity of the NCC as a whole. It is particularly important to respect differences in roles and characteristics of each NCC campus, Tsukiji and Kashiwa, to make the most of their strong points to maximize the research output of the NCC. Collaborations with the Clinical Research Support Offices of both hospitals remain crucial, and the CRAS wishes to play a significant role in bridging the various sections together in the NCC.
Activities and Future prospects of each Division/Section
1.Research Administration Division
1)Research Administration Section
The Research Administration Section is a central office in charge of various administrative work related to research funding including application and reporting. The major external funding sources of the NCC are competitive grants from the government and government-supported agencies, such as the Ministry of Health, Labour and Welfare (MHLW), the Japan Science and Technology Agency (JST), and the Japan Agency for Medical Research and Development (AMED). This section also serves as an administrative office for the NCC Research and Development Fund, which is provided directly from the government to the NCC for fulfillment of its mission as the national core institute of the cancer control. This section organized seminars regarding research funding and its rules to prevent financial misconducts.
(Future prospects)
The Guidelines for Managing and Auditing Public Research Funds at Research Institutes was updated by the Ministry of Education, Culture, Sports, Science and Technology (MEXT) in February 2014 and adopted by the MHLW in March 2014. This section serves as a compliance promotion office of the Guidelines and has established a new system for research fund administration, which is fully compatible with the new Guidelines.
2)Research Administrators
Research administrators (RA) helped the Director of the NCCRI to review the research achievements in 2016 and to develop research plans for 2017. RA also offered an administrative office work for the NCC Seeds Selection Committee, which was established in 2015 to identify promising research and development seeds of cancer diagnostics and therapeutics in the NCC; in 2015, 11 seeds were selected to be funded for a year by the NCC in-house grant.
RA has supported the promotion of the commercial viability of research outcomes based upon a comprehensive alliance with companies. Candidate compounds for clinical development have been provided by collaboration with business enterprises.
(Future prospects)
RA promotes translation of innovative research in the NCC to clinical diagnostic and therapeutic development and to patient care through four major mechanisms: the NCC Seeds Selection Committee, comprehensive alliances with leading companies, participation in the academic Drug Discovery Network, and establishment of NCC spin-off venture companies.
3)Research Auditors
For clinical studies led by the NCC's investigators, seven audits were conducted on GCP (Good Clinical Practice) trials, and two on B type advanced medical service trials. In addition, eight internal audits were conducted on departments, which have been engaged in clinical research, as part of self-inspection required by the ethics guideline. Four audits/investigations were also conducted on clinical research upon request by researchers. Other activities included revision of audit procedure, GCP-related training and consultation.
(Future prospects)
The audit procedure was revised for clinical interventional studies involving invasive procedure for the research subjects to adopt "Ethical Guidelines for Medical and Health Research Involving Human Subjects"; the audit will be conducted by request of researchers, and some requests have already been received. Considering the expected increase of the requests, introduction of the risk-based audit plan will be effective for improvement in our research process. The development and improvement in quality management system will be continued to support clinical research.
4)Research Alliance Section and Intellectual Property Section
To make the NCC research outcomes clinically useful products available to cancer patients, the Research Alliance Section promotes collaborative research arrangements with the private sector. Number of collaborative researches and their funds has been increasing each year (Figure 1).
Figure 1. Collaborative Researches (FY 2008-2015)
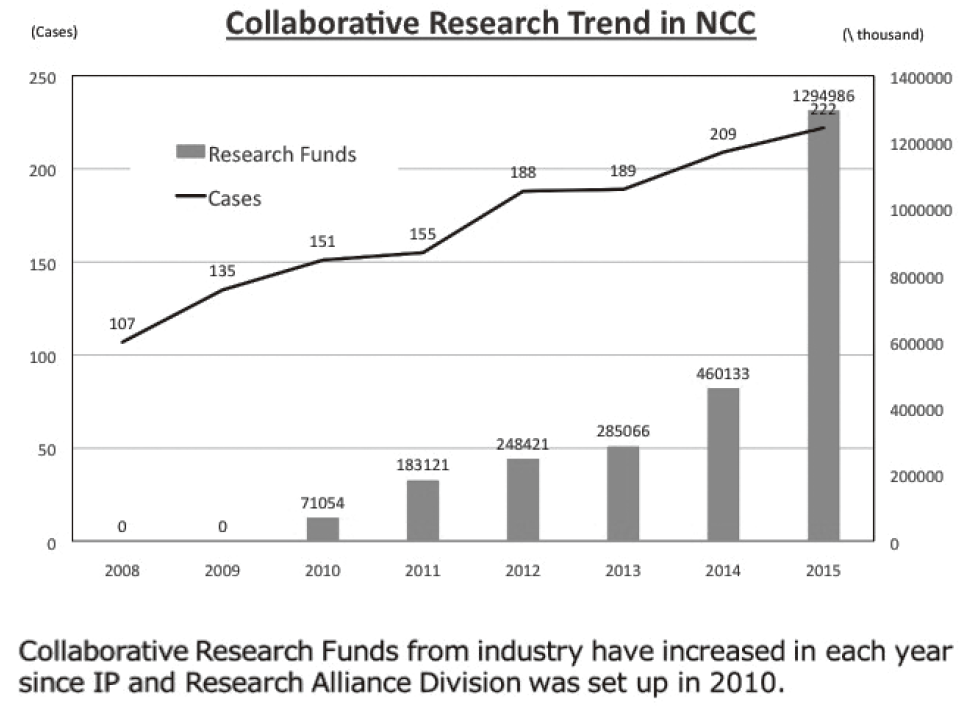
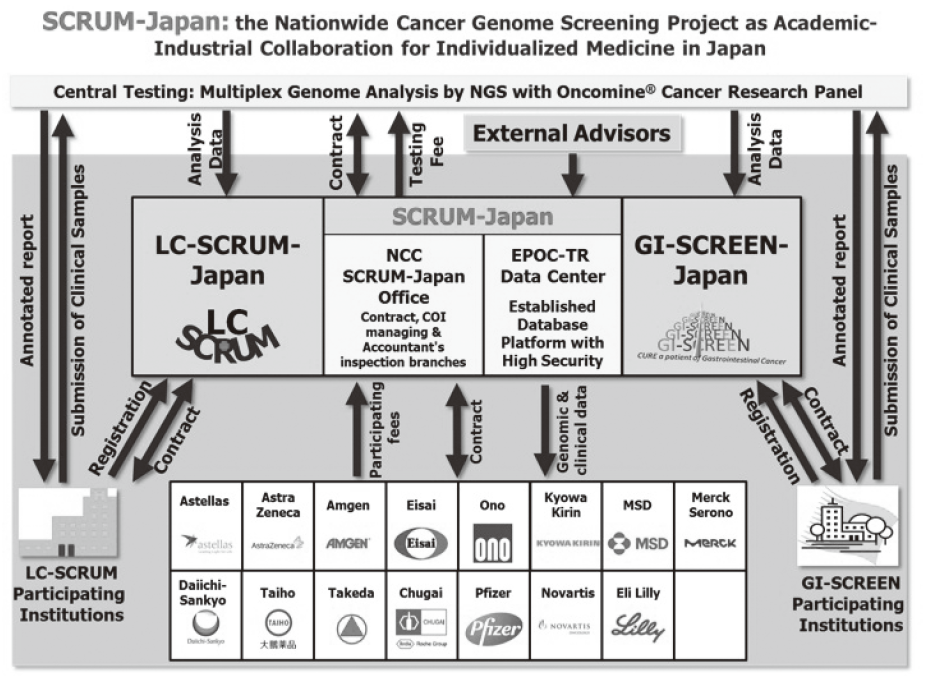
As of December 31, 2016, the number of collaboration was 237, and their research funds amount to ca. 1.5 billion yen, which exceeded those of 2015. The NCC has developed a comprehensive collaboration research system with companies and academic institutions. With the addition of two new collaborative frameworks this year, the NCC has 10 comprehensive collaborative alliances (Figure 2). This section supported a nationwide genomic screening project with the participation of major pharmaceutical companies and institutions across Japan (SCRUM-Japan "Cancer Genome Screening Project for Individualized Medicine in Japan"), which was launched in March 2015. Currently 15 pharmaceutical companies have registered (Figure 3). This section has also assisted regional alliances with medium-sized medical device companies.
Figure 2. The NCC-Industry Partnership
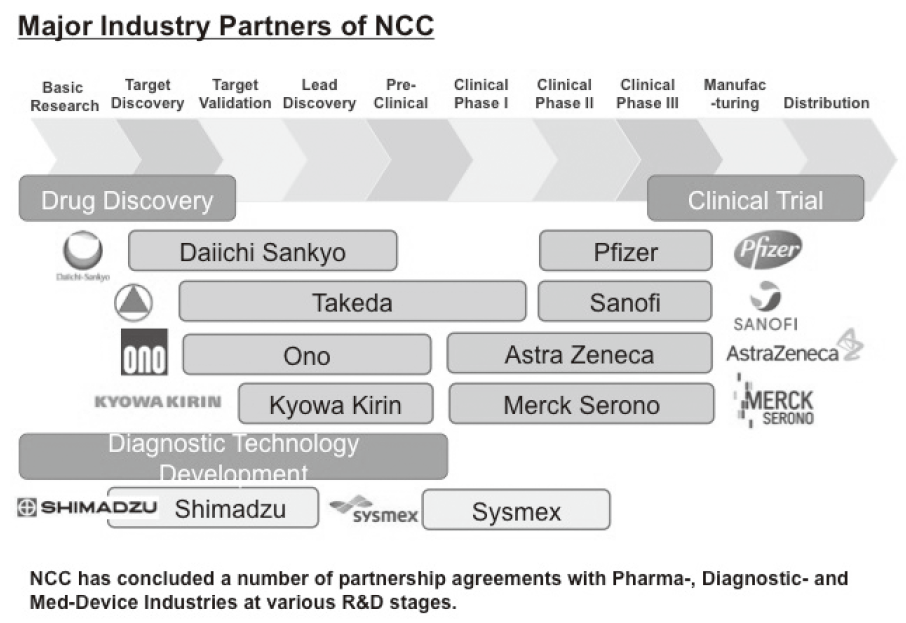
Figure 3. SCRUM-Japan (established in February 2015)
The Intellectual Property (IP) Section constantly reviews IPs and dismisses those that could not find a business sponsor within a certain period; the limited budget would be focused on IPs that are commercially viable. The number of patent licensing agreements in recent years is shown in Figure 4.
Figure 4. Licensing Deals (FY2008-2015)
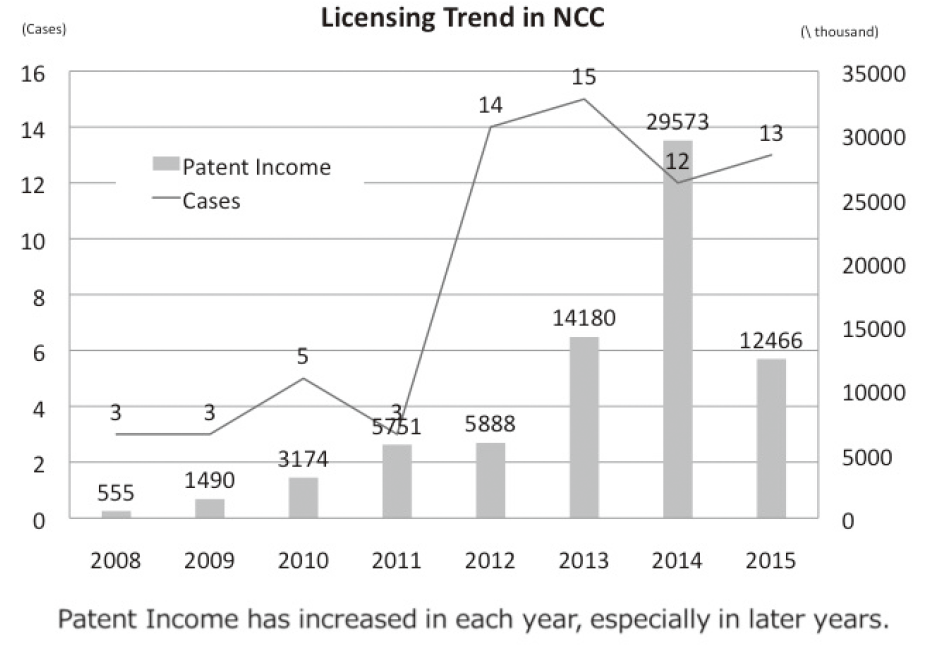
Staff members actively participate in seminars with regard to IP laws and regulations to update their knowledge and elevate their skills to promote academic-industrial alliances. Their competencies in problem solution gained through on-the-job training (OJT) and effective consultation with experts will enable them to face new challenges in innovative fields.
(Future prospects)
In the trend of Open Innovation, this section will keep supporting the creation of systemic and effective collaborative research frameworks. It also foresees the possibility of a new laboratory setup where research is being performed by researchers from both industry and the NCC, and should lead to more functional collaboration.
As to IP management, the NCC employs patent strategies to protect sthe potential value of the invention for industry, through which the translation of academic science and technology is made to the patient's bedside. The IP section plays an important role in assisting NCC's comprehensive decision making, taking various aspects into consideration such as incubation of innovative technology, cost and effect balance, and risk management.
2.Biostatistics Division
The Biostatistics Division has a responsible role in study design, analysis, interpretation, and publication, especially in the Japan Clinical Oncology Group (JCOG) and the Exploratory Oncology Research & Clinical Trial Center (EPOC) clinical trials and the investigator-initiated clinical trials which are led by investigators in the NCC Hospitals. We have also committed to establish an infrastructure to support clinical trials in the NCC Hospitals.
We provided 15 introductory Biostatistics lectures, which contain three newly prepared lectures, for investigators in the NCC to learn and review the elementary aspect of biostatistics. We had a cumulative total of 599 participants. In addition, we hosted two biostatistics lectures to cover the important biostatistical side of various application fields and a cumulative total of 81 investigators participated.
Furthermore, we have provided biostatistical consultation and expertise, which supports NCC investigators working on basic, translational, clinical, and epidemiological researches. We offered advice regarding 120 problems (77 in the Tsukiji campus and 43 in the Kashiwa campus) for which biostatistical consultation was requested.
(Future prospects)
The NCC has a critical role for providing clinical service, education, conducting researches, and making policy recommendation/proposal, which all need decisions on the basis of solid and scientific evidence from reliable data and information. The mission of the Biostatistics Division is to contribute to providing the best evidence and the improvement of clinical practice and public health through the development and application of statistical methods. The Biostatistics Division is expanding its independent and collaborative research within a range of areas, including prevention and policy recommendations/proposals, as well as treatment development. We are also opening up a new methodological research area in which a mathematical approach will serve as a solid basis.
3.Human Research Protection Section
The major role of the Human Research Protection Section has been its function as a secretariat for various research ethics committees (IRBs) in the NCC for human subject research.
Aside from routine responsibilities, the major achievements of this section in 2016 included the establishment of the Kashiwa office for the IRB. In the last two years of the preparation, it is increasingly apparent that the two-office IRB system may actually need a substantial investment, but it would be ready to start in 2017.
The NCC is expected to act in the scope of the nation-wide cancer research and care, in addition to the operation of its own institute. This section has continued its effort to develop the central IRB system, which is being promoted internationally for clinical trials and other medical research. It obtained two AMED grants related to the central IRB projects in 2016 and cooperated with JCOG and SCRUM-Japan, who provided this section with the feedback and opinions on the review.
(Future prospects)
The Human Research Protection Section has still been struggling with the long review-waiting list. The major reason is the relative lack of the adequate personnel in response to the increasing volume and subjects of the work expected for this section. In addition to the request for further resource allocation, this section would keep reviewing its work process to make it more efficient by, for instance, an introduction of paper-less conference.
The CRAS and the Clinical Research Support Offices in both hospitals keep working to further update the research support system in the NCC, which will enable the IRB to concentrate more on its core responsibility of ethics reviews.
In 2016, the Human Research Protection Section applied for the Certified IRBl for the first time and is waiting for the review results from the MHLW.
4.Bioethics Section
The Bioethics Section provides research ethics consultation service to researchers / research support staff, IRB members/staff, and other stakeholders in research enterprises throughout the lifecycle of a study. The research ethics consultation service plays strictly an advisory role and is independent from IRB decisions. The Bioethics Section started providing an ethics consultation service from 2015 and received about 100 calls a year. Also, this section provides ethics education to researchers and IRB members/staff.
(Future prospects)
The Bioethics Section will provide a consultation service and improve quality of the consultation service. This section will also provide advanced education on research ethics as well as basic education.
List of papers published in 2016
Journal
1.Nishina T, Boku N, Gotoh M, Shimada Y, Hamamoto Y, Yasui H, Yamaguchi K, Kawai H, Nakayama N, Amagai K, Mizusawa J, Nakamura K, Shirao K, Ohtsu A. Randomized phase II study of second-line chemotherapy with the best available 5-fluorouracil regimen versus weekly administration of paclitaxel in far advanced gastric cancer with severe peritoneal metastases refractory to 5-fluorouracil-containing regimens (JCOG0407). Gastric Cancer, 19:902-910, 2016
2.Yokota T, Igaki H, Kato K, Tsubosa Y, Mizusawa J, Katayama H, Nakamura K, Fukuda H, Kitagawa Y. Accuracy of preoperative diagnosis of lymph node metastasis for thoracic esophageal cancer patients from JCOG9907 trial. Int J Clin Oncol, 21:283-288, 2016
3.Nishi Y, Fukushima T, Nomura S, Tomoyose T, Nakachi S, Morichika K, Tedokon I, Tamaki K, Shimabukuro N, Taira N, Miyagi T, Karimata K, Ohama M, Yamanoha A, Tamaki K, Hayashi M, Arakaki H, Uchihara J-N, Ohshiro K, Asakura Y, Kuba-Miyara M, Karube K, Masuzaki H. Characterization of patients with aggressive adult T-cell leukemia-lymphoma in Okinawa, Japan: a retrospective analysis of a large cohort. Int J Hematol, 104:468-475, 2016
4.Tanaka K, Hasegawa T, Nojima T, Oda Y, Mizusawa J, Fukuda H, Iwamoto Y. Prospective evaluation of Ki-67 system in histological grading of soft tissue sarcomas in the Japan Clinical Oncology Group Study JCOG0304. World J Surg Oncol, 14:110, 2016
5.Saito S, Fujita S, Mizusawa J, Kanemitsu Y, Saito N, Kinugasa Y, Akazai Y, Ota M, Ohue M, Komori K, Shiozawa M, Yamaguchi T, Akasu T, Moriya Y. Male sexual dysfunction after rectal cancer surgery: Results of a randomized trial comparing mesorectal excision with and without lateral lymph node dissection for patients with lower rectal cancer: Japan Clinical Oncology Group Study JCOG0212. Eur J Surg Oncol, 42:1851-1858, 2016
6.Onda T, Satoh T, Saito T, Kasamatsu T, Nakanishi T, Nakamura K, Wakabayashi M, Takehara K, Saito M, Ushijima K, Kobayashi H, Kawana K, Yokota H, Takano M, Takeshima N, Watanabe Y, Yaegashi N, Konishi I, Kamura T, Yoshikawa H. Comparison of treatment invasiveness between upfront debulking surgery versus interval debulking surgery following neoadjuvant chemotherapy for stage III/IV ovarian, tubal, and peritoneal cancers in a phase III randomised trial: Japan Clinical Oncology Group Study JCOG0602. Eur J Cancer, 64:22-31, 2016
7.Nishio S, Kitagawa R, Shibata T, Yoshikawa H, Konishi I, Ushijima K, Kamura T. Prognostic factors from a randomized phase III trial of paclitaxel and carboplatin versus paclitaxel and cisplatin in metastatic or recurrent cervical cancer: Japan Clinical Oncology Group (JCOG) trial: JCOG0505-S1. Cancer Chemother Pharmacol, 78:785-790, 2016
8.Mizusawa J, Morizane C, Okusaka T, Katayama H, Ishii H, Fukuda H, Furuse J. Randomized Phase III study of gemcitabine plus S-1 versus gemcitabine plus cisplatin in advanced biliary tract cancer: Japan Clinical Oncology Group Study (JCOG1113, FUGA-BT). Jpn J Clin Oncol, 46:385-388, 2016
9.Goto K, Ohe Y, Shibata T, Seto T, Takahashi T, Nakagawa K, Tanaka H, Takeda K, Nishio M, Mori K, Satouchi M, Hida T, Yoshimura N, Kozuki T, Imamura F, Kiura K, Okamoto H, Sawa T, Tamura T. Combined chemotherapy with cisplatin, etoposide, and irinotecan versus topotecan alone as second-line treatment for patients with sensitive relapsed small-cell lung cancer (JCOG0605): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol, 17:1147-1157, 2016
10.Fujitani K, Yang H-K, Mizusawa J, Kim Y-W, Terashima M, Han S-U, Iwasaki Y, Hyung WJ, Takagane A, Park DJ, Yoshikawa T, Hahn S, Nakamura K, Park CH, Kurokawa Y, Bang Y-J, Park BJ, Sasako M, Tsujinaka T. Gastrectomy plus chemotherapy versus chemotherapy alone for advanced gastric cancer with a single non-curable factor (REGATTA): a phase 3, randomised controlled trial. Lancet Oncol, 17:309-318, 2016
11.Eba J, Nakamura K, Mizusawa J, Suzuki K, Nagata Y, Koike T, Hiraoka M, Watanabe S, Ishikura S, Asamura H, Fukuda H. Stereotactic body radiotherapy versus lobectomy for operable clinical stage IA lung adenocarcinoma: comparison of survival outcomes in two clinical trials with propensity score analysis (JCOG1313-A). Jpn J Clin Oncol, 46:748-753, 2016
12.Ikeda M, Sato A, Mochizuki N, Toyosaki K, Miyoshi C, Fujioka R, Mitsunaga S, Ohno I, Hashimoto Y, Takahashi H, Hasegawa H, Nomura S, Takahashi R, Yomoda S, Tsuchihara K, Kishino S, Esumi H. Phase I trial of GBS-01 for advanced pancreatic cancer refractory to gemcitabine. Cancer Sci, 107:1818-1824, 2016
13.Kataoka K, Takeuchi H, Mizusawa J, Ando M, Tsubosa Y, Koyanagi K, Daiko H, Matsuda S, Nakamura K, Kato K, Kitagawa Y. A randomized Phase III trial of thoracoscopic versus open esophagectomy for thoracic esophageal cancer: Japan Clinical Oncology Group Study JCOG1409. Jpn J Clin Oncol, 46:174-177, 2016
14.Akutsu Y, Kato K, Igaki H, Ito Y, Nozaki I, Daiko H, Yano M, Udagawa H, Nakagawa S, Takagi M, Mizusawa J, Kitagawa Y. The Prevalence of Overall and Initial Lymph Node Metastases in Clinical T1N0 Thoracic Esophageal Cancer: From the Results of JCOG0502, a Prospective Multicenter Study. Ann Surg, 264:1009-1015, 2016
15.Hamamoto Y, Mizusawa J, Katayama H, Nakamura K, Kato K, Tsubosa Y, Ishikura S, Igaki H, Shinoda M, Fukuda H, Kitagawa Y, Ando N. Inter-institutional survival heterogeneity in chemoradiation therapy for esophageal cancer: exploratory analysis of the JCOG0303 study. Jpn J Clin Oncol, 46:389-392, 2016
16.Tsushima T, Mizusawa J, Sudo K, Honma Y, Kato K, Igaki H, Tsubosa Y, Shinoda M, Nakamura K, Fukuda H, Kitagawa Y. Risk Factors for Esophageal Fistula Associated With Chemoradiotherapy for Locally Advanced Unresectable Esophageal Cancer: A Supplementary Analysis of JCOG0303. Medicine (Baltimore), 95:e3699, 2016
17.Kurokawa Y, Boku N, Yamaguchi T, Ohtsu A, Mizusawa J, Nakamura K, Fukuda H. Inter-institutional heterogeneity in outcomes of chemotherapy for metastatic gastric cancer: correlative study in the JCOG9912 phase III trial. ESMO Open, 1:e000031, 2016
18.Kataoka K, Katai H, Mizusawa J, Katayama H, Nakamura K, Morita S, Yoshikawa T, Ito S, Kinoshita T, Fukagawa T, Sasako M. Non-Randomized Confirmatory Trial of Laparoscopy-Assisted Total Gastrectomy and Proximal Gastrectomy with Nodal Dissection for Clinical Stage I Gastric Cancer: Japan Clinical Oncology Group Study JCOG1401. J Gastric Cancer, 16:93-97, 2016
19.Hatogai K, Kitano S, Fujii S, Kojima T, Daiko H, Nomura S, Yoshino T, Ohtsu A, Takiguchi Y, Doi T, Ochiai A. Comprehensive immunohistochemical analysis of tumor microenvironment immune status in esophageal squamous cell carcinoma. Oncotarget, 7:47252-47264, 2016
20.Bando H, Doi T, Muro K, Yasui H, Nishina T, Yamaguchi K, Takahashi S, Nomura S, Kuno H, Shitara K, Sato A, Ohtsu A. A multicenter phase II study of TAS-102 monotherapy in patients with pre-treated advanced gastric cancer (EPOC1201). Eur J Cancer, 62:46-53, 2016
21.Saruwatari K, Umemura S, Nomura S, Kirita K, Matsumoto S, Yoh K, Niho S, Ohmatsu H, Ohe Y, Goto K. Prognostic Factor Analysis in Patients With Small-Cell Lung Cancer Treated With Third-Line Chemotherapy. Clin Lung Cancer, 17:581-587, 2016
22.Hatogai K, Yano T, Kojima T, Onozawa M, Daiko H, Nomura S, Yoda Y, Doi T, Kaneko K, Ohtsu A. Salvage photodynamic therapy for local failure after chemoradiotherapy for esophageal squamous cell carcinoma. Gastrointest Endosc, 83:1130-1139.e3, 2016
