Annual Report 2017
Department of Hepatobiliary and Pancreatic Oncology
Masafumi Ikeda, Shuichi Mitsunaga, Izumi Ohno, Hiroshi Imaoka, Yusuke Hashimoto, Hideaki Takahashi, Mitsuhito Sasaki, Kazuo Watanabe, Kumiko Umemoto, Gen Kimura, Yuko Suzuki, Motoyasu Kan
Introduction
The Department of Hepatobiliary and Pancreatic Oncology is responsible for the diagnosis and treatment of patients with hepatic, biliary, and pancreatic cancers as well as the interventional management by endoscopic or percutaneous procedure (Tables 1, 2). Our goals are to provide high-quality cancer treatment with adequate palliative care, and to develop novel and effective treatments and procedures through well-designed clinical trials and research.
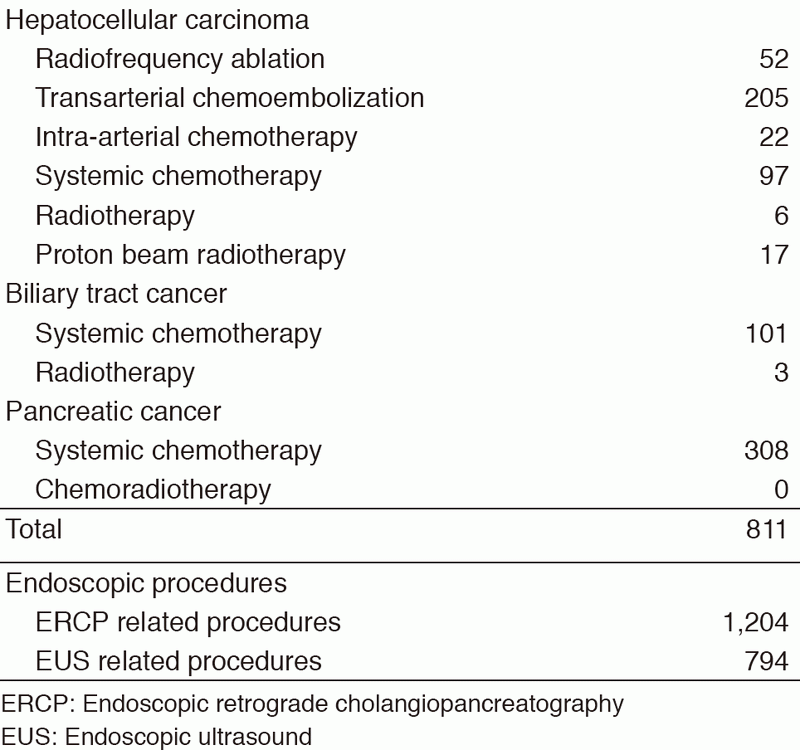
Table 1. Number of new patients

Our team and what we do
Our department is composed of seven staff oncologists and five residents, with the average of 54 beds in the hospital. These doctors are divided into two teams and each team conducts clinical rounds for each admitted patient every morning and evening. The treatment strategies on individual patients are discussed in weekly tumor board conferences attended by medical oncologists, surgeons, radiologists, radiation oncologists, and pharmacists. Furthermore, we are also responsible for endoscopic abdominal ultrasonographic examinations, endoscopic or percutaneous
ultrasound-guided biopsies of abdominal masses,
local ablative therapy for liver tumors, endoscopic or percutaneous biliary drainage, abscess drainage and stenting for obstructive jaundice, etc. (Table 2)
Research activities
1. Hepatocellular carcinoma (HCC)
Sorafenib and lenvatinib for first line and regorafenib for second line are currently acknowledged as standard chemotherapy for patients with advanced HCC. All agents are multi-kinase inhibitors, and have some troublesome toxicities. The management against adverse events are essential to bring out the maximum antitumor effects of these agents. Therefore, we constituted a medical team, such as "Team Nexavar", "Team Regorafenib" and "Team Lenvatinib", for the management and supportive care for toxicities. Also, some immune checkpoint inhibitors, including nivolumab and pembrolizumab, showed the promising efficacy and safety in patients with unresectable HCC. The development of multi-kinase inhibitors and immune checkpoints inhibitors are underway to intensify the efficacy.
2. Biliary tract cancer (BTC)
Both gemcitabine plus cisplatin and gemcitabine plus S-1 are currently available standard of care for advanced BTC as first line treatment. However, no standard regimens have been established as second line treatment. We found that the overexpression of HER3 or HER4 as well as HER2 were frequently observed in BTC patients. At present, the development of Pan HER inhibitor, such as varlitinib, is planning.
3. Pancreatic cancer (PC)
Gemcitabine (Gem) plus nab-paclitaxel and modified FOLFIRINOX has been established as first line treatment of advanced PC, and both regimens are often used for advanced PC on daily practice. We conducted the retrospective comparison study to clarify which regimen was better as first line chemotherapy, Gem plus nab-paclitaxel or modified FOLFIRINOX, and which regimen was better as second line chemotherapy after treatment of Gem plus nab-paclitaxel, modified FOLFIRINOX or S-1 alone.
4. Neuroendocrine neoplasms (NEN)
Since NENs are relatively rare and indolent neoplasms, progression-free survival (PFS) is more frequently used in phase III trials for NENs. But this surrogacy of PFS for overall survival (OS) has never been validated. We performed a literature-based analysis of phase II and III trials for NENs. PFS is an acceptable surrogate for OS in clinical trials for NENs.
5. Other research
We investigated the value on diagnosis and response evaluation of serum microRNAs on highly sensitive microarray in PC and BTC and the usefulness of endoscopic procedures, such as celiac plexus neurolysis, placement of metallic biliary stent or fine needle aspiration.
Clinical trials
Fifty-two clinical trials (sponsored: 30 trials, investigator-initiated: 22 trials) are ongoing, and 12 clinical trials (sponsored: eight trials, investigator-initiated: five trials) are being planned for the upcoming year.
1. HCC
A randomized phase II trial comparing transarterial chemoembolization (TACE) using drug eluting Beads vs. conventional TACE (PRESIDENT) is ongoing. Some sponsored trials of lenvatinib plus pembrolizumab, lenvatinib plus nivolumab, bevacizumab plus atezolizumab, sorafenib plus PDR-001, as first line chemotherapy, and nivolumab, durvalumab plus tremelimumab, PF-4518600, as second line chemotherapy are ongoing.
2. BTC
The enrollment of patients of JCOG1202, which was a randomized phase III trial comparing adjuvant S-1 with observation in patients with resected BTC, was completed. Some sponsored trials of Gem cisplatin plus nivolumab as first line chemotherapy and resminostat plus S-1, nivolumab, capecitabine plus varlitinib, E7090, BAY1436032, etc. for advanced BTCs refractory to Gem are ongoing.
3. PC
Some clinical trials of a phase II/III trial of neoadjuvant S-1 and concurrent radiotherapy vs. Gem plus nab-paclitaxel for borderline resectable PC (GABARNANCE), Gem plus nab-paclitaxel vs. modified FOLFIRINOX for locally advanced PC (JCOG1407), Gem plus oxaliplatin for familial PC (FABRIC) as the first line setting are ongoing. Some sponsored trials of HF10 plus Gem plus nab-paclitaxel, a phase III trial of BBI608 plus Gem plus nab-paclitaxel vs. Gem plus nab-paclitaxel, etc. are ongoing as the first line setting.
Education
For our residents, daily trainings are provided with group discussion on the daily practice of management of inpatients and outpatients, in which they can learn the indication, administration, and management of adverse events from loco-regional treatments to systemic chemotherapy for hepatic, biliary, and pancreatic cancer patients, and the accompanied procedures to make a diagnosis and interventional management, and so on. In addition, they can make presentations of their research domestically, hold worldwide meetings, and make a paper in English under the instruction of staff physicians.
Future prospects
The prognosis of patients with hepatic, biliary, and pancreatic cancers remains dismal, and standard treatments for these cancers is limited. In Japan, the incidences of these types of cancer, especially HCC and BTC, are higher than those in western countries. Therefore, we must conduct a lot of novel and promising clinical trials and research that take the lead worldwide. And it is necessary to develop biomarker research and endoscopic management accompanying cancer treatment.
List of papers published in January 2017 - March 2018
Journal
1. Ueno M, Li CP, Ikeda M, Ishii H, Mizuno N, Yamaguchi T, Ioka T, Oh DY, Ichikawa W, Okusaka T, Matsuyama Y, Arai D, Chen LT, Park YS, Furuse J. A randomized phase II study of gemcitabine plus Z-360, a CCK2 receptor-selective antagonist, in patients with metastatic pancreatic cancer as compared with gemcitabine plus placebo. Cancer Chemother Pharmacol, 80:307-315, 2017
2. Kudo M, Hatano E, Ohkawa S, Fujii H, Masumoto A, Furuse J, Wada Y, Ishii H, Obi S, Kaneko S, Kawazoe S, Yokosuka O, Ikeda M, Ukai K, Morita S, Tsuji A, Kudo T, Shimada M, Osaki Y, Tateishi R, Sugiyama G, Abada PB, Yang L, Okusaka T, Zhu AX. Ramucirumab as second-line treatment in patients with advanced hepatocellular carcinoma: Japanese subgroup analysis of the REACH trial. J Gastroenterol, 52:494-503, 2017
3. Ikeda K, Kudo M, Kawazoe S, Osaki Y, Ikeda M, Okusaka T, Tamai T, Suzuki T, Hisai T, Hayato S, Okita K, Kumada H. Phase 2 study of lenvatinib in patients with advanced hepatocellular carcinoma. J Gastroenterol, 52:512-519, 2017
4. Imaoka H, Sasaki M, Takahashi H, Hashimoto Y, Ohno I, Mitsunaga S, Watanabe K, Umemoto K, Kimura G, Suzuki Y, Ikeda M. Progression-free survival as a surrogate endpoint in advanced neuroendocrine neoplasms. Endocr Relat Cancer, 24:475-483, 2017
5. Okuyama H, Ikeda M, Takahashi H, Ohno I, Hashimoto Y, Mitsunaga S, Sakamoto Y, Kondo S, Morizane C, Ueno H, Kobayashi T, Arai Y, Okusaka T. Transarterial (Chemo)Embolization for Liver Metastases in Patients with Neuroendocrine Tumors. Oncology, 92:353-359, 2017
6. Ikeda M, Okusaka T, Sato Y, Furuse J, Mitsunaga S, Ueno H, Morizane C, Inaba Y, Kobayashi T, Arai Y. A Phase I/II trial of continuous hepatic intra-arterial infusion of 5-fluorouracil, mitoxantrone and cisplatin for advanced hepatocellular carcinoma. Jpn J Clin Oncol, 47:512-519, 2017
7. Kimura G, Hashimoto Y, Ikeda M. Successful obliteration of bleeding duodenal varices by EUS-guided injection of N-butyl-2-cyanoacrylate. VIDEOGIE, 2:317-319, 2017
8. Ikeda M, Takahashi H, Kondo S, Lahn MMF, Ogasawara K, Benhadji KA, Fujii H, Ueno H. Phase 1b study of galunisertib in combination with gemcitabine in Japanese patients with metastatic or locally advanced pancreatic cancer. Cancer Chemother Pharmacol, 79:1169-1177, 2017
9. Hijioka S, Hosoda W, Matsuo K, Ueno M, Furukawa M, Yoshitomi H, Kobayashi N, Ikeda M, Ito T, Nakamori S, Ishii H, Kodama Y, Morizane C, Okusaka T, Yanagimoto H, Notohara K, Taguchi H, Kitano M, Yane K, Maguchi H, Tsuchiya Y, Komoto I, Tanaka H, Tsuji A, Hashigo S, Kawaguchi Y, Mine T, Kanno A, Murohisa G, Miyabe K, Takagi T, Matayoshi N, Yoshida T, Hara K, Imamura M, Furuse J, Yatabe Y, Mizuno N. Rb Loss and KRAS Mutation Are Predictors of the Response to Platinum-Based Chemotherapy in Pancreatic Neuroendocrine Neoplasm with Grade 3: A Japanese Multicenter Pancreatic NEN-G3 Study. Clinical cancer research, 23:4625-4632, 2017
10. Kudo M, Moriguchi M, Numata K, Hidaka H, Tanaka H, Ikeda M, Kawazoe S, Ohkawa S, Sato Y, Kaneko S, Furuse J, Takeuchi M, Fang X, Date Y, Okusaka T. S-1 versus placebo in patients with sorafenib-refractory advanced hepatocellular carcinoma (S-CUBE): a randomised, double-blind, multicentre, phase 3 trial. The lancet. Gastroenterology & hepatology, 2:407-417, 2017
11. Ikeda M, Shimizu S, Sato T, Morimoto M, Kojima Y, Inaba Y, Hagihara A, Kudo M, Nakamori S, Kaneko S, Sugimoto R, Tahara T, Ohmura T, Yasui K, Sato K, Ishii H, Furuse J, Okusaka T. Reply to the Letter to the editor 'Sorafenib plus hepatic arterial infusion chemotherapy with cisplatin versus Sorafenib for advanced hepatocellular carcinoma: randomized phase II trial' by Fornaro et al. Ann Oncol, 28:903-904, 2017
12. Mitsunaga S, Okusaka T, IkedaM, Ozaka M, Ohkawa S, Ioka T, Shimura T, Sato K, Terao K, Ochiai A, Furuse J. Multicenter, Open-Label, Phase I/II Study of Tocilizumab, an Anti-Interleukin-6 Receptor Monoclonal Antibody, Combined with Gemcitabine in Patients with Advanced Pancreatic Cancer. J Med Diagn Meth, 6:234, 2017
13. Ikeda M, Kudo M, Aikata H, Nagamatsu H, Ishii H, Yokosuka O, Torimura T, Morimoto M, Ikeda K, Kumada H, Sato T, Kawai I, Yamashita T, Horio H, Okusaka T. Transarterial chemoembolization with miriplatin vs. epirubicin for unresectable hepatocellular carcinoma: a phase III randomized trial. J Gastroenterol, 53:281-290, 2018
14. Ikeda M, Ioka T, Fukutomi A, Morizane C, Kasuga A, Takahashi H, Todaka A, Okusaka T, Creasy CL, Gorman S, Felitsky DJ, Kobayashi M, Zhang F, Furuse J. Efficacy and safety of trametinib in Japanese patients with advanced biliary tract cancers refractory to gemcitabine. Cancer Sci, 109:215-224, 2018
15. Ikeda M, Morizane C, Ueno M, Okusaka T, Ishii H, Furuse J. Chemotherapy for hepatocellular carcinoma: current status and future perspectives. Jpn J Clin Oncol, 48:103-114, 2018