Center for Research Administration and Support (CRAS)
Teruhiko Yoshida, Takeyuki Sato, Masayoshi Shibatuji, Nobuko Ushirozawa, Yutaka Inazu, Daiki Suzuki, Fumie Suzuki, Kiyoka Nagayama, Yuko Yamada, Jinko Sato, Yuichi Amanuma, Miki Ito, Mayumi Yoshida, Masaki Yamamoto, Genta Ohno, Yukari Nakayama, Kyoko Chiba, Yuichi Nakamura, Ryu Aoyama, Maho Muraoka, Hanae Takahashi, Kazunori Aoki, Kazuhiko Aoyagi, Taro Shibata, Gakuto Ogawa, Yasue Kojima, Aya Kuchiba, Junki Mizusawa, Masashi Wakabayashi, Ryunosuke Machida, Shogo Nomura, Chihiro Yaita, Yuki Konda, Shimon Tashiro, Tsunakuni Ikka, Akiko Nakada, Mao Shikano, Mari Hirata, Noriko Yamashita, Yayoi Ofuji, Kuniko Takahashi, Harumi Maruno, Shizuka Yamamoto, Norie Enomoto, Satoko Arai, Tomoko Ishida, Shinae Kamiya, Nanako Ninomiya, Tomoko Seki, Haruka Nakada
Introduction
The Center for Research Administration and Support (CRAS) was established on July 16, 2014. The starting members of the CRAS were approximately 160 staff, who together offered diverse functions and specialties, ranging from research fund administration, alliances with private sectors, intellectual properties, clinical research coordinators and data managers, monitoring and audit, biostatistics support, offices for research ethics (IRB), and COI committees.
The background and the purpose of the creation of the CRAS was explained by Dr. Tomomitsu Hotta, the President of the National Cancer Center (NCC), in the NCC News 2014 Vol.5 No.3 (in Japanese). Briefly, since its foundation in 1962, the NCC has added several new segments and organizations to evolve as a comprehensive cancer center. Because each segment needed its own research infrastructure, support activities in the NCC had become fragmented and scattered with the possibility of gaps and redundancies. Dr. Hotta approached the Strategic Planning Bureau and assembled the "NCC New Vision" in 2014, in which he proposed integration and communication for various research support functions in the NCC. The CRAS was created in response to the 2014 Vision.
In 2015, the NCC Hospital (NCCH) and the NCC Hospital East (NCCHE) were certified as the Core Clinical Research Hospital under the Medical Care Act in August and September, respectively.
It was then required that the support functions for clinical research, especially those concerning clinical trials, needed to be operated under the explicit responsibility of a hospital director. As a result, the governance of the Research Coordination Division, Research Promotion Division and Regulatory Science Section of the CRAS has now moved to the Clinical Research Support Offices, which belong to the common departments of each hospital. In January 2017, these divisions and section were officially separated from the CRAS in the NCC organization, and their 2016 annual reports are found in the respective hospital chapters.
Another major reformation of the CRAS in 2017 was the establishment of the Bioethics Division by expanding the former Bioethics Section. The Clinical Trials Act was promulgated on April 14, 2017 and came into effect on April 1, 2018. The Division worked very hard and efficiently in the period of the transitional measures of the law. Despite the serious scarcity of human resources in the specialty field, the NCC has been endowed with strong staff, who have been contributing to work not just inside the NCC but also all over Japan with regard to research ethics-related capabilities. However, it was with great regret that the first Chief of the Bioethics Division, Dr. Shimon Tashiro, moved to a university at the end of fiscal 2018. Dr. Tashiro is highly respected for his distinguished skills, decision-making, knowledge, passion and personality both inside and outside the CRAS.
In 2018, the concept of the RA (research administrator) system in NCC was discussed in the CRAS and updated to the NCC headquarters.
(Future prospects)
The NCC embarked on the new era under the leadership of a newly appointed President and Directors of the NCC Research Institute (NCCRI) and both hospitals (NCCH, NCCHE) on April 1, 2016. The Vision and high-priority research targets have been redefined, and reorganization is in progress in various parts of the NCC. However, the core concept of the CRAS stays unchanged to contribute to the NCC mission by promoting organic unity of the NCC as a whole. It is particularly important to respect differences in roles and characteristics of each NCC campus, Tsukiji and Kashiwa, to make the most of their strong points to maximize the research output of the NCC. Collaborations with the Clinical Research Support Offices of both hospitals remain crucial, and the CRAS aims to contribute to bringing the various sections together in the NCC.
Activities and Future prospects of each Division/Section
1. Research administration Division
1) Research Administration Section
The Research Administration Section is a central office in charge of various administrative work related to research funding including application and reporting. The major external funding sources of the NCC are competitive grants from the government and governmentsupported agencies, such as the Ministry of Health, Labour and Welfare (MHLW), the Japan Science and Technology Agency (JST), and the Japan Agency for Medical Research and Development (AMED).
This section also serves as an administrative office for the NCC Research and Development Fund, which is provided directly from the government to the NCC for fulfillment of its mission as the national core institute of cancer control. This section organized seminars regarding research funding and its rules to prevent financial misconduct.
(Future prospects)
The Guidelines for Managing and Auditing Public Research Funds at Research Institutes was updated by the Ministry of Education, Culture, Sports, Science and Technology (MEXT) in February 2014 and adopted by the MHLW in March 2014. This section serves as a compliance promotion office of the Guidelines and has established a new system for research fund administration, which is fully compatible with the new Guidelines.
2) Research Alliance Section and Intellectual Property Section
To make the NCC research outcomes clinically useful products available to cancer patients, the Research Alliance Section promotes collaborative research arrangements with the private sector. The number of collaborative research projects and their funds have been increasing each year (Figure 1). In 2018, the number of collaborations was 378, and their research funds amount to approximately 4.5 billion yen, which exceeds that of 2017. The NCC has developed the comprehensive collaboration research system with companies or academic institutions. With addition of a new collaborative framework this year, NCC has 11 comprehensive collaborative alliances (Figure 2). The section supported a nationwide genomic screening project with the participation of major pharmaceutical companies and institutions across Japan (SCRUM-Japan "Cancer Genome Screening Project for Individualized Medicine in Japan"), which launched in March 2016. Currently, 16 pharmaceutical companies have registered (Figure 3).
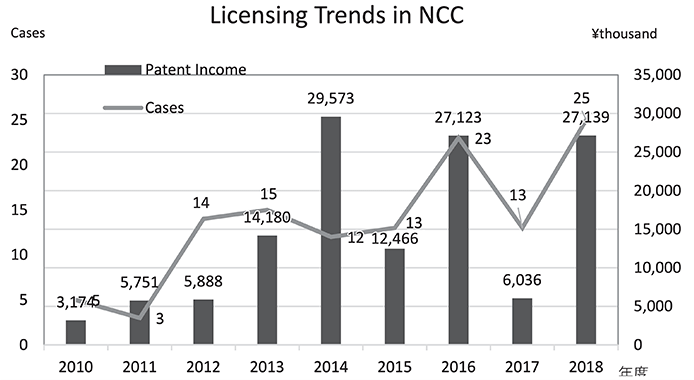
The Intellectual Property (IP) Section constantly reviews IPs and dismisses those which cannot find a business sponsor within a certain period, so the limited budget can be focused on IPs which are commercially viable. The number of patent licenses in recent years is shown in Figure 4.
Staff members actively participate in seminars with regard to IP laws and regulations to update their knowledge and elevate their skills to promote academic-industrial alliances. Their competency in problem solution gained through OJT and effective consultation with experts will enable them to face new challenges in innovative affairs.
(Future prospects)
In the trend of Open Innovation, the Section will keep supporting creation of systemic and effective collaborative research frameworks. It also foresees the possibility of new laboratory setups where research is being performed by researchers from both industry and the NCC, and will lead to more functional collaboration.
As to IP management, the NCC employs patent strategies to protect the potential value of the invention for industry, through which the translation of academic science and technology goes to the patient's bedside. The IP section plays an important role in assisting NCC's comprehensive decision making, taking various aspects into consideration such as incubation of innovative technology, cost and effect balance, and risk management.
3) Research Administrators
Research administrators (RA) helped the Director of the NCCRI to review the research achievements in 2018, to develop plans for 2019 and to revise 6-year research plans. As part of Tsukiji TR Board activity, RA has visited 23 domestic and global pharmaceutical companies to introduce the bioresources, technologies and TR/rTR supporting system in NCC to promote co-clinical studies in cooperation with companies. RA has also supported the promotion of the commercial viability of research outcomes based upon a comprehensive alliance with companies. Candidate compounds for clinical development have been provided by collaboration with business enterprises.
(Future prospects)
RA promotes the acquisition of large competitive research funds through the reinforcement of collaboration between organizations in the NCC, and also promotes the translation of innovative research in the NCC to clinical diagnostic and therapeutic development and to patient care through four major mechanisms: the NCC Seeds Selection Committee, comprehensive alliances with leading companies, participation in the academic Drug Discovery Network, and establishment of NCC spin-off venture companies.
2. Biostatistics Division
The Biostatistics Division has a role of responsibility in study design, analysis, interpretation and publication, especially in JCOG and EPOC clinical trials and the investigator-initiated clinical trials which are led by investigators in the NCC Hospitals. We have also committed to establish an infrastructure to support clinical trials in the NCC Hospitals. Furthermore, we have been actively involved in collaborative relationships with the NCC Research Institute and the Center for Public Health Sciences.
We provided 14 introductory Biostatistics lectures for investigators in the NCC to learn and review the elementary aspects of Biostatistics. We newly provided lectures about the implementation of statistical analysis with analysis software. We had a cumulative total of 496 participants. In addition, we hosted four Biostatistics lectures to cover the important biostatistical side of various application fields and a cumulative total of 159 investigators participated. The lectures are open to any applicants from outside institutes.
Furthermore, we have provided biostatistical consultation and expertise, which supports NCC investigators working on basic, translational, clinical and epidemiological research. We offered advice for about 87 problems (49 in Tsukiji campus and 38 in Kashiwa campus) for which biostatistical consultation was requested from April 2018 to March 2019.
In addition, we provided an internship program for 11 graduate students from the two graduate schools of the University of Tokyo, i.e., the Biostatistics and bioinformatics course, Graduate School of Interdisciplinary Information Studies, and the School of Public Health, Graduate School of Medicine.
(Future Prospects)
The NCC has a critical role for providing clinical service, education, conducting research and making policy recommendations/proposals, which are all required to make a decision on the basis of solid and scientific evidence from reliable data and information. The mission of the Biostatistics Division is to contribute to providing best evidence and the improvement of clinical practice and public health through the development and application of statistical methods. The Biostatistics Division is expanding on its independent and collaborative research with a range of areas, including prevention and policy recommendations/proposals, as well as treatment development. This year, we have made a particular effort to build effective relationships with the investigators at the NCC Research Institute and the Center for Public Health Sciences. Collaborative projects have been launched, including the evaluation and implementation of medical technology with artificial intelligence. Collaboration on epidemiologic studies have motivated us to develop statistical methods to get deeper insight of etiological mechanisms or public health impacts. We are working on promoting a cooperative framework with outside experts in statistics/biostatistics. We are opening up a new methodological research area in which a mathematical approach will serve as a solid basis.
3. Bioethics Division
The Bioethics Division has a mission of providing information, education and training programs into research ethics including human subject protection, conflict of interests (COIs) management on clinical research, and the administration of institutional research ethics reviews boards (IRBs). In addition to this mission, each of the following three sections under the Division has its specific responsible roles described as below.
1) Bioethics and Healthcare Law Section
The Bioethics and Healthcare Law Section (BHLS) provides research ethics consultation services (RECS) to researchers, research supporters, IRB members, and other concerned parties or groups of people. The RECS have an advisory function, which does not hold any compulsory or binding force and therefore differs from the roles of the IRBs, so as to give instructions and advice in terms of ethics on how to handle and solve ethical and sometimes legal issues arising in each human research study. The RECS therefore sometimes go beyond regulatory compliance. In FY2018, the BHLS held 153 consultations (95 at Tsukiji campus, and 58 at Kashiwa campus), which involved issues on the Clinical Trial Act (17), on governmental ethical guidelines (132), and others (4). In addition, the BHLS held several educational seminars for both NCC and non-NCC researchers and/or IRB staff.
(Future prospects)
The BHLS will continue providing and promoting the RECS as well as offering educational materials of research ethics, so as to enhance the ethical conduct of human subject research. In FY2019 in particular, the BHLS will aim to build a collaborative consultation system among the full-time BHLS staff between the two campuses.
2) Human Research Protection Section
The Human Research Protection Section (HRPS) serves as the administrative offices for the various types of IRBs for human subject research, including the certified review committees defined by the Clinical Trials Act (Act No. 16 of April 14, 2017).
Accomplishments of the reviews by the IRBs at the NCC in FY2018 included 1,701 active research projects and 429 new research plans by NCC faculty members; while a total of 29 new research plans by researchers outside of the NCC were requested for review.
(Future prospects)
The HRPS will continue working, in cooperation with the NCCH's and the NCCHE's Ethical Review Support Sections, to ameliorate all of the NCC's ethics review processes for more appropriate and efficient ways to achieve goals. Particularly in FY2019, the HRPS is planning standardization of the operation of the IRBs.
3) COI Management Section
The COI Management Section (COIMS) is the administrative office of the COI Review Committee on Clinical Research at the NCC, in addition to responding to COI-related inquiries from researchers. The targets of the COI reviews are individual researchers and research projects using public research funds, but also each member of various committees at the NCC undergoes COI reviews.
In FY2018, the COIMS managed 2,744 researchers' COI reviews in 249 research projects; and we provided consultations for 11 inquiries from other departments at the NCC.
(Future prospects)
The COIMS will continue to strive for efficient, high-quality management of COIs, as well as providing the latest information on the COI standards for researchers and committee members.
Figure 1. Collavorative Research Trends in NCC
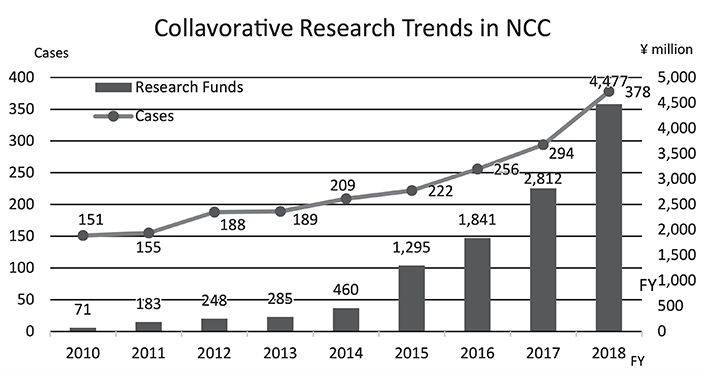

Figure 2. Major Industry Partners of the NCC
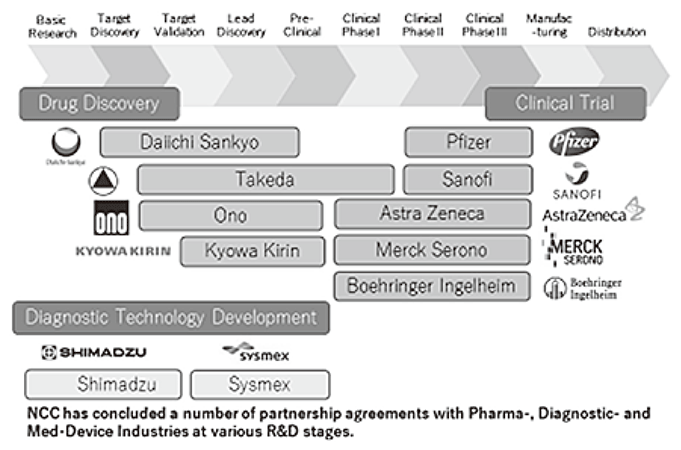

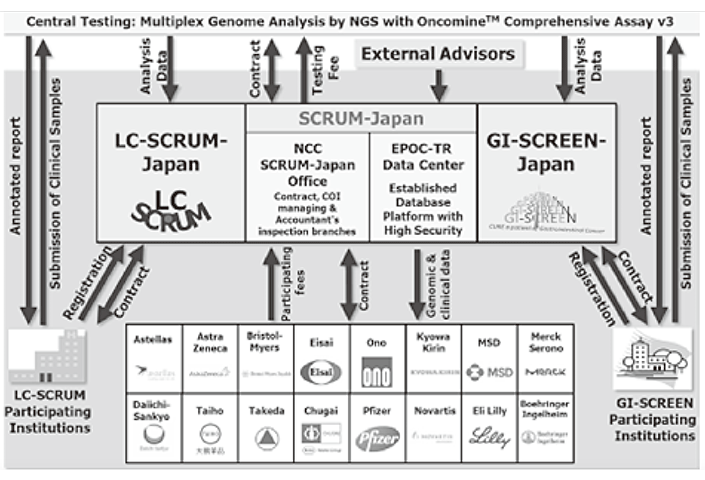
Figure 4. Licensing Trends in NCC