Annual Report 2020
Department of Medical Oncology
Toru Mukohara, Ako Hosono, Yoichi Naito, Nobuaki Matsubara, Hirofumi Mukai, Kenichi Harano, Chihiro Kondoh, Takahiro Kogawa, Yoko Fukasawa, Chikako Funasaka, Shota Kusuhara, Yoriko Hasegawa, Yumi Fujimoto
Introduction
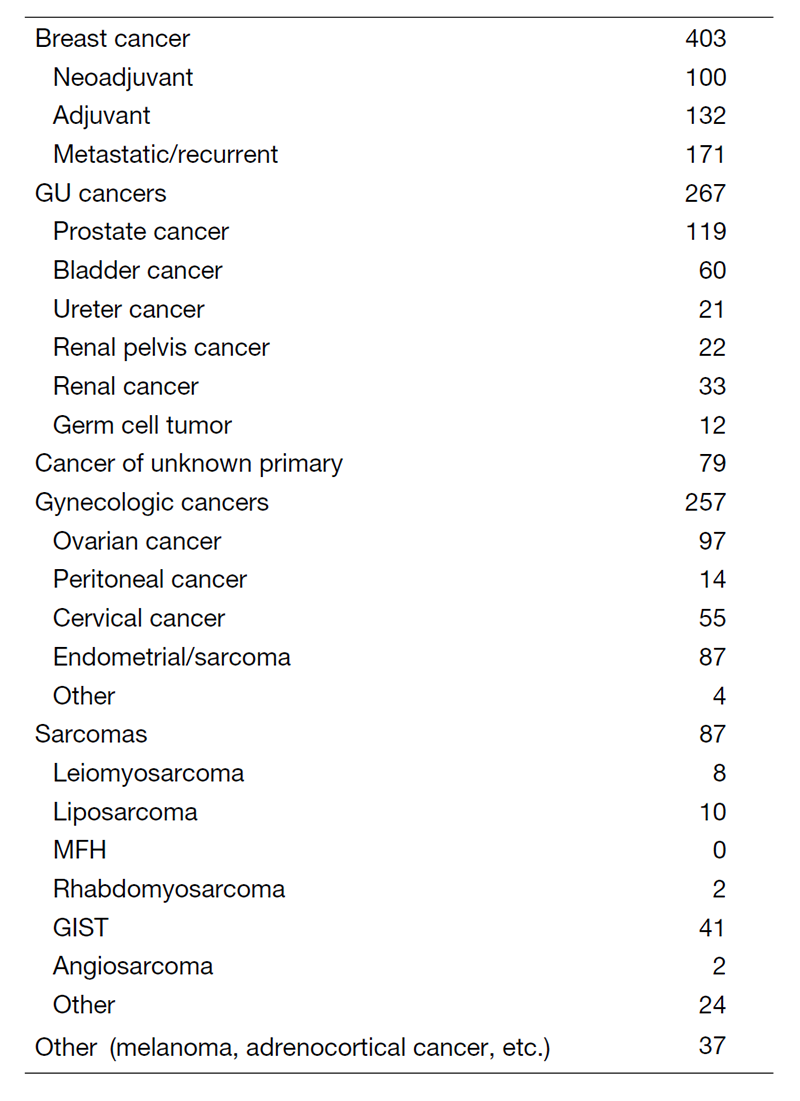
In the clinic, we care for patients with breast, urologic, and gynecologic cancers, as well as sarcoma. We also provide the best available treatment to patients with cancers of unknown primary, multiple primary cancers, or other rare cancers that are difficult to treat in other hospitals.
We also played a central role in implementing multi-gene panel testing, which has come to insurance coverage in 2019. Our department chief, Dr. Mukohara, serves as Head of the expert panel (molecular tumor board). We managed 208 out of 430 total cases that were discussed in our expert panel, which contributed to genomic medicine in several regions.
We also contributed to the operation of the “Lady’s Center” which opened in 2018 as a core of multi-disciplinary care for women with cancer. Further, we run the Hereditary Breast and Ovarian Cancer (HBOC) syndrome clinic to provide proper information to patients carrying a high risk of the syndrome. We also developed an institutional system for upcoming insurance coverage of BRCA genetic testing and associated Risk Reducing Mastectomy (RRM) and Risk Reducing Salpingo-Oophorectomy (RRSO).
This year, we launched an Investigator-initiated trial (IIT) of olaparib +/- pembrolizumab as neoadjuvant therapy for advanced ovarian cancer (PI, Kenichi Harano). Adjunct translational research utilizing single-cell RNA sequence technology is conducted in collaboration with the Division of Genome TR. We are currently preparing for another IIT of limb-cooling as a means to mitigate paclitaxel-induced sensory neuropathy. We are also conducting collaborative studies with a technology company to explore the clinical utility of their system for detecting circulating tumor cells (CTCs).
Research activities
We are participating in many company-sponsored trials and other collaborative studies of clinical trial groups, such as JCOG, JBCRG, and WJOG.
We are also implementing clinical and translational researches with funds obtained from Grant-in-Aid for Scientific Research (“3D co-culture of cancer cells in malignant effusion/CTC with adipose stem cell”; PI, Mukohara T) (“New sub-classification of triple-negative breast cancer to evaluate immunogenicity”; PI, Kogawa T).
This year, we published a preclinical study about combination therapy for HER2-positive breast cancer in collaboration with the Division of Genome TR. We are now proposing an IIT based on the preclinical findings.
Education
Our goal in education is to foster “genuine” medical oncologists. Graduates from our resident/chief resident programs are expected to provide not only standard therapies regardless of cancer types but also multi-disciplinary care in cooperation with other medical professionals. Obtaining skills for palliative care and dealing with oncological emergencies are also expected. Further, we require and help them to implement clinical research to address clinical questions they develop themselves and report the results in international journals. This is because we aim to develop scientific clinicians who can focus on issues scientifically and create evidence for themselves. As an achievement, this year, one of our residents earned a Diplomate, Subspecialty Board of Medical Oncology, JSMO.
Further, we earned an educational grant with the theme “Comprehensive educational program to develop medical professionals and peer supporters to empower Adolescent and Young Adult (AYA) breast cancer patients”. We held a multidisciplinary AYA conference (mAYAcon) to deliver optimal care to AYA cancer patients and to conduct educational programs for medical professionals.
Future Prospects
In the clinic, we will provide clinical care with high patient satisfaction through a multi-disciplinary team approach. In education, we will foster medical oncologists with skills and knowledge for cross-organ oncology care and scientific acumen. In research, we will initiate multi-center clinical trials and Phase I to III developmental trials of investigational drugs. We will also expand our research activity to preclinical research for response prediction and overcoming resistance to anti-cancer drugs. Multiple research themes are currently under discussion with industries and other academic organizations.
List of papers published in 2020
Journal
1. Umeda K, Miyamura T, Yamada K, Sano H, Hosono A, Sumi M, Okita H, Kumamoto T, Kawai A, Hirayama J, Jyoko R, Sawada A, Nakayama H, Hosoya Y, Maeda N, Yamamoto N, Imai C, Hasegawa D, Chin M, Ozaki T. Clinical outcome of patients with recurrent or refractory localized Ewing's sarcoma family of tumors: A retrospective report from the Japan Ewing Sarcoma Study Group. Cancer Rep (Hoboken), e1329, 2021
2. Doi T, Fujiwara Y, Shitara K, Shimizu T, Yonemori K, Matsubara N, Ohno I, Kogawa T, Naito Y, Leopold L, Munteanu M, Yatsuzuka N, Han SR, Samkari A, Yamamoto N. The safety and tolerability of epacadostat alone and in combination with pembrolizumab in patients with advanced solid tumors: results from a first-in-Japanese phase I study (KEYNOTE-434). Invest New Drugs, 39:152-162, 2021
3. Sunami K, Naito Y, Aimono E, Amano T, Ennishi D, Kage H, Kanai M, Komine K, Koyama T, Maeda T, Morita S, Sakai D, Kohsaka S, Tsuchihara K, Yoshino T. The initial assessment of expert panel performance in core hospitals for cancer genomic medicine in Japan. Int J Clin Oncol, 26:443-449, 2021
4. Naito Y, Aburatani H, Amano T, Baba E, Furukawa T, Hayashida T, Hiyama E, Ikeda S, Kanai M, Kato M, Kinoshita I, Kiyota N, Kohno T, Kohsaka S, Komine K, Matsumura I, Miura Y, Nakamura Y, Natsume A, Nishio K, Oda K, Oda N, Okita N, Oseto K, Sunami K, Takahashi H, Takeda M, Tashiro S, Toyooka S, Ueno H, Yachida S, Yoshino T, Tsuchihara K. Clinical practice guidance for next-generation sequencing in cancer diagnosis and treatment (edition 2.1). Int J Clin Oncol, 26:233-283, 2021
5. Hasegawa Y, Matsubara N, Kogawa T, Naito Y, Harano K, Hosono A, Onishi T, Hojo T, Shimokawa M, Mukohara T. Neo-Bioscore in Guiding Post-surgical Therapy in Patients With Triple-negative Breast Cancer Who Received Neoadjuvant Chemotherapy. In Vivo, 35:1041-1049, 2021
6. Powles T, Rosenberg JE, Sonpavde GP, Loriot Y, Durán I, Lee JL, Matsubara N, Vulsteke C, Castellano D, Wu C, Campbell M, Matsangou M, Petrylak DP. Enfortumab Vedotin in Previously Treated Advanced Urothelial Carcinoma. N Engl J Med, 384:1125-1135, 2021
7. Okamoto A, Kondo E, Nakamura T, Yanagida S, Hamanishi J, Harano K, Hasegawa K, Hirasawa T, Hori K, Komiyama S, Matsuura M, Nakai H, Nakamura H, Sakata J, Tabata T, Takehara K, Takekuma M, Yokoyama Y, Kase Y, Sumino S, Soeda J, Suri A, Aoki D, Sugiyama T. Phase 2 single-arm study on the efficacy and safety of niraparib in Japanese patients with heavily pretreated, homologous recombination-deficient ovarian cancer. J Gynecol Oncol, 32:e16, 2021
8. Umeda K, Miyamura T, Yamada K, Sano H, Hosono A, Sumi M, Okita H, Kamio T, Maeda N, Fujisaki H, Jyoko R, Watanabe A, Hosoya Y, Hasegawa D, Takenaka S, Nakagawa S, Chin M, Ozaki T. Prognostic and therapeutic factors influencing the clinical outcome of metastatic Ewing sarcoma family of tumors: A retrospective report from the Japan Ewing Sarcoma Study Group. Pediatr Blood Cancer, 68:e28844, 2021
9. Kijima T, Yamamoto H, Saito K, Kusuhara S, Yoshida S, Yokoyama M, Matsuoka Y, Numao N, Sakai Y, Matsubara N, Yuasa T, Masuda H, Yonese J, Kageyama Y, Fujii Y. Early C-reactive protein kinetics predict survival of patients with advanced urothelial cancer treated with pembrolizumab. Cancer Immunol Immunother, 70:657-665, 2021
10. Uemura H, Matsushima H, Kobayashi K, Mizusawa H, Nishimatsu H, Fizazi K, Smith M, Shore N, Tammela T, Tabata KI, Matsubara N, Iinuma M, Uemura H, Oya M, Momma T, Kawakita M, Fukasawa S, Kobayashi T, Kuss I, Le Berre MA, Snapir A, Sarapohja T, Suzuki K. Efficacy and safety of darolutamide in Japanese patients with nonmetastatic castration-resistant prostate cancer: a sub-group analysis of the phase III ARAMIS trial. Int J Clin Oncol, 26:578-590, 2021
11. Yamaguchi T, Hozumi Y, Sagara Y, Takahashi M, Yoneyama K, Fujisawa T, Osumi S, Akabane H, Nishimura R, Mieno MN, Mukai H. The impact of neoadjuvant systemic therapy on breast conservation rates in patients with HER2-positive breast cancer: Surgical results from a phase II randomized controlled trial. Surg Oncol, 36:51-55, 2021
12. Masuda N, Mukai H, Inoue K, Rai Y, Ohno S, Ohtani S, Shimizu C, Hashigaki S, Muramatsu Y, Umeyama Y, Iwata H, Toi M. Analysis of subsequent therapy in Japanese patients with hormone receptor?positive/human epidermal growth factor receptor 2?negative advanced breast cancer who received palbociclib plus endocrine therapy in PALOMA-2 and -3. Breast Cancer, 28:335-345, 2021
13. Takashima T, Hara F, Iwamoto T, Uemura Y, Ohsumi S, Yotsumoto D, Hozumi Y, Watanabe T, Saito T, Watanabe KI, Tsurutani J, Toyama T, Akabane H, Nishimura R, Taira N, Ohashi Y, Mukai H. A Correlation Analysis Between Metabolism-related Genes and Treatment Response to S-1 as First-line Chemotherapy for Metastatic Breast Cancer: The SELECT BC-EURECA Study. Clin Breast Cancer, 2021
14. Minami H, Doi T, Toyoda M, Imamura Y, Kiyota N, Mitsuma A, Shimokata T, Naito Y, Matsubara N, Tajima T, Tokushige K, Ishihara K, Cameron S, Ando Y. Phase I study of the antiprogrammed cell death-1 Ab spartalizumab (PDR001) in Japanese patients with advanced malignancies. Cancer Sci, 112:725-733, 2021
15. Mukohara T, Hosono A, Mimaki S, Nakayama A, Kusuhara S, Funasaka C, Nakao T, Fukasawa Y, Kondoh C, Harano K, Naito Y, Matsubara N, Tsuchihara K, Kuwata T. Effects of Ado-Trastuzumab Emtansine and Fam-Trastuzumab Deruxtecan on Metastatic Breast Cancer Harboring HER2 Amplification and the L755S Mutation. Oncologist, 2021
16. Tsurutani J, Hara F, Kitada M, Takahashi M, Kikawa Y, Kato H, Sakata E, Naito Y, Hasegawa Y, Saito T, Iwasa T, Taira N, Takashima T, Kashiwabara K, Aihara T, Mukai H. Randomized phase II study to determine the optimal dose of 3-week cycle nab-paclitaxel in patients with metastatic breast cancer. Breast, 55:63-68, 2021
17. Yamamoto M, Yoshida M, Furuse J, Sano K, Ohtsuka M, Yamashita S, Beppu T, Iwashita Y, Wada K, Nakajima TE, Sakamoto K, Hayano K, Mori Y, Asai K, Matsuyama R, Hirashita T, Hibi T, Sakai N, Tabata T, Kawakami H, Takeda H, Mizukami T, Ozaka M, Ueno M, Naito Y, Okano N, Ueno T, Hijioka S, Shikata S, Ukai T, Strasberg S, Sarr MG, Jagannath P, Hwang TL, Han HS, Yoon YS, Wang HJ, Luo SC, Adam R, Gimenez M, Scatton O, Oh DY, Takada T. Clinical practice guidelines for the management of liver metastases from extrahepatic primary cancers 2021. J Hepatobiliary Pancreat Sci, 28:1-25, 2021
18. Toi M, Imoto S, Ishida T, Ito Y, Iwata H, Masuda N, Mukai H, Saji S, Shimizu A, Ikeda T, Haga H, Saeki T, Aogi K, Sugie T, Ueno T, Kinoshita T, Kai Y, Kitada M, Sato Y, Jimbo K, Sato N, Ishiguro H, Takada M, Ohashi Y, Ohno S. Adjuvant S-1 plus endocrine therapy for oestrogen receptor-positive, HER2-negative, primary breast cancer: a multicentre, open-label, randomised, controlled, phase 3 trial. Lancet Oncol, 22:74-84, 2021
19. Nakatsukasa K, Niikura N, Kashiwabara K, Amemiya T, Watanabe KI, Hata H, Kikawa Y, Taniike N, Yamanaka T, Mitsunaga S, Nakagami K, Adachi M, Kondo N, Shibuya Y, Hayashi N, Naito M, Yamashita T, Umeda M, Mukai H, Ota Y. Secondary endpoints analysis in patients with estrogen receptor-positive metastatic breast cancer treated with everolimus and exemestane enrolled in Oral Care-BC. BMC Cancer, 21:34, 2021
20. Shimoi T, Nagai SE, Yoshinami T, Takahashi M, Arioka H, Ishihara M, Kikawa Y, Koizumi K, Kondo N, Sagara Y, Takada M, Takano T, Tsurutani J, Naito Y, Nakamura R, Hattori M, Hara F, Hayashi N, Mizuno T, Miyashita M, Yamashita N, Yamanaka T, Saji S, Iwata H, Toyama T. The Japanese Breast Cancer Society Clinical Practice Guidelines for systemic treatment of breast cancer, 2018 edition. Breast Cancer, 27:322-331, 2020
21. Ohashi Y, Ikeda M, Kunitoh H, Sasako M, Okusaka T, Mukai H, Fujiwara K, Nakamura M, Oba MS, Kimura T, Ibusuki K, Sakon M. Venous thromboembolism in cancer patients: report of baseline data from the multicentre, prospective Cancer-VTE Registry. Jpn J Clin Oncol, 50:1246-1253, 2020
22. Doi T, Matsubara N, Kawai A, Naka N, Takahashi S, Uemura H, Yamamoto N. Phase I study of TAS-115, a novel oral multi-kinase inhibitor, in patients with advanced solid tumors. Invest New Drugs, 38:1175-1185, 2020
23. Shingaki S, Kogawa T, Shimokawa M, Harano K, Naito Y, Kusuhara S, Fujimoto Y, Matsubara N, Hosono A, Mukai H, Onishi T, Hojo T, Mukohara T. Use of eribulin as an earlier-line chemotherapy for patients with HER2-negative metastatic breast cancer. J Cancer, 11:4099-4105, 2020
24. Nakayama K, Osaka W, Matsubara N, Takeuchi T, Toyoda M, Ohtake N, Uemura H. Shared decision making, physicians' explanations, and treatment satisfaction: a cross-sectional survey of prostate cancer patients. BMC Med Inform Decis Mak, 20:334, 2020
25. Cortes J, Cescon DW, Rugo HS, Nowecki Z, Im SA, Yusof MM, Gallardo C, Lipatov O, Barrios CH, Holgado E, Iwata H, Masuda N, Otero MT, Gokmen E, Loi S, Guo Z, Zhao J, Aktan G, Karantza V, Schmid P. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): a randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet, 396:1817-1828, 2020
26. Fujimoto Y, Morita TY, Ohashi A, Haeno H, Hakozaki Y, Fujii M, Kashima Y, Kobayashi SS, Mukohara T. Combination treatment with a PI3K/Akt/mTOR pathway inhibitor overcomes resistance to anti-HER2 therapy in PIK3CA-mutant HER2-positive breast cancer cells. Sci Rep, 10:21762, 2020
27. Hussain M, Mateo J, Fizazi K, Saad F, Shore N, Sandhu S, Chi KN, Sartor O, Agarwal N, Olmos D, Thiery-Vuillemin A, Twardowski P, Roubaud G, ?zg?ro?lu M, Kang J, Burgents J, Gresty C, Corcoran C, Adelman CA, de Bono J. Survival with Olaparib in Metastatic Castration-Resistant Prostate Cancer. N Engl J Med, 383:2345-2357, 2020
28. Necchi A, Nishiyama H, Matsubara N, Lee JL, Petrylak DP, de Wit R, Drakaki A, Liepa AM, Mao H, Bell-McGuinn K, Powles T. Health-related quality of life in the randomized phase 3 study of ramucirumab plus docetaxel versus placebo plus docetaxel in platinum-refractory advanced urothelial carcinoma (RANGE). BMC Urol, 20:181, 2020
29. Demetri GD, Antonescu CR, Bjerkehagen B, Bov?e JVMG, Boye K, Chac?n M, Dei Tos AP, Desai J, Fletcher JA, Gelderblom H, George S, Gronchi A, Haas RL, Hindi N, Hohenberger P, Joensuu H, Jones RL, Judson I, Kang YK, Kawai A, Lazar AJ, Le Cesne A, Maestro R, Maki RG, Mart?n J, Patel S, Penault-Llorca F, Premanand Raut C, Rutkowski P, Safwat A, Sbaraglia M, Schaefer IM, Shen L, Serrano C, Sch?ffski P, Stacchiotti S, Sundby Hall K, Tap WD, Thomas DM, Trent J, Valverde C, van der Graaf WTA, von Mehren M, Wagner A, Wardelmann E, Naito Y, Zalcberg J, Blay JY. Diagnosis and management of tropomyosin receptor kinase (TRK) fusion sarcomas: expert recommendations from the World Sarcoma Network. Ann Oncol, 31:1506-1517, 2020
30. Fukushima H, Kijima T, Fukuda S, Moriyama S, Uehara S, Yasuda Y, Tanaka H, Yoshida S, Yokoyama M, Matsuoka Y, Saito K, Matsubara N, Numao N, Sakai Y, Yuasa T, Masuda H, Yonese J, Kageyama Y, Fujii Y. Impact of radiotherapy to the primary tumor on the efficacy of pembrolizumab for patients with advanced urothelial cancer: A preliminary study. Cancer Med, 9:8355-8363, 2020
31. Sawaki M, Taira N, Uemura Y, Saito T, Baba S, Kobayashi K, Kawashima H, Tsuneizumi M, Sagawa N, Bando H, Takahashi M, Yamaguchi M, Takashima T, Nakayama T, Kashiwaba M, Mizuno T, Yamamoto Y, Iwata H, Kawahara T, Ohashi Y, Mukai H. Randomized Controlled Trial of Trastuzumab With or Without Chemotherapy for HER2-Positive Early Breast Cancer in Older Patients. J Clin Oncol, 38:3743-3752, 2020
32. Ohashi Y, Ikeda M, Kunitoh H, Sasako M, Okusaka T, Mukai H, Fujiwara K, Nakamura M, Oba MS, Kimura T, Ibusuki K, Sakon M. Corrigendum to: Venous thromboembolism in cancer patients: report of baseline data from the multicentre, prospective Cancer-VTE Registry. Jpn J Clin Oncol, 50:1346, 2020
33. Inoue K, Takahashi M, Mukai H, Yamanaka T, Egawa C, Sakata Y, Ikezawa H, Matsuoka T, Tsurutani J. Effectiveness and safety of eribulin in Japanese patients with HER2-negative, advanced breast cancer: a 2-year post-marketing observational study in a real-world setting. Invest New Drugs, 38:1540-1549, 2020
34. Yamashita S, Hattori N, Fujii S, Yamaguchi T, Takahashi M, Hozumi Y, Kogawa T, El-Omar O, Liu YY, Arai N, Mori A, Higashimoto H, Ushijima T, Mukai H. Multi-omics analyses identify HSD17B4 methylation-silencing as a predictive and response marker of HER2-positive breast cancer to HER2-directed therapy. Sci Rep, 10:15530, 2020
35. Iwamoto T, Fujisawa T, Shien T, Araki K, Sakamaki K, Sangai T, Kikawa Y, Takao S, Nishimura R, Takahashi M, Aihara T, Mukai H, Taira N. The efficacy of sequential second-line endocrine therapies (ETs) in postmenopausal estrogen receptor-positive and HER2-negative metastatic breast cancer patients with lower sensitivity to initial ETs. Breast Cancer, 27:973-981, 2020
36. Fizazi K, Shore N, Tammela TL, Ulys A, Vjaters E, Polyakov S, Jievaltas M, Luz M, Alekseev B, Kuss I, Le Berre MA, Petrenciuc O, Snapir A, Sarapohja T, Smith MR. Nonmetastatic, Castration-Resistant Prostate Cancer and Survival with Darolutamide. N Engl J Med, 383:1040-1049, 2020
37. Matsubara N, Suzuki K, Kazama H, Tsukube S, Seto T, Matsuyama H. Cabazitaxel in patients aged ≧80 years with castration-resistant prostate cancer: Results of a post-marketing surveillance study in Japan. J Geriatr Oncol, 11:1067-1073, 2020
38. Shitara K, Yamazaki K, Tsushima T, Naito T, Matsubara N, Watanabe M, Sarholz B, Johne A, Doi T. Phase I trial of the MET inhibitor tepotinib in Japanese patients with solid tumors. Jpn J Clin Oncol, 50:859-866, 2020
39. Ohsumi S, Mukai H, Takahashi M, Hozumi Y, Akabane H, Park Y, Tokunaga E, Takashima T, Watanabe T, Sagara Y, Kaneko T, Ohashi Y. Factors affecting enrollment in randomized controlled trials conducted for patients with metastatic breast cancer. Jpn J Clin Oncol, 50:873-881, 2020
40. Matsuyama H, Matsubara N, Kazama H, Seto T, Tsukube S, Suzuki K. Real-world efficacy and safety of two doses of cabazitaxel (20 or 25 mg/m(2)) in patients with castration-resistant prostate cancer: results of a Japanese post-marketing surveillance study. BMC Cancer, 20:649, 2020
41. Tsuda H, Tsugawa K, Akiyama F, Horii R, Kurosumi M, Moriya T, Takano T, Takei H, Nakayama T, Miyagi Y, Yamauchi C, Yamashita T, Aogi K, Mukai H, Sugie T, Iwata H, Masuda S. Correction to: Histological classification of breast tumors in the General Rules for Clinical and Pathological Recording of Breast Cancer (18th edition). Breast Cancer, 27:792, 2020
42. Yoshino T, Pentheroudakis G, Mishima S, Overman MJ, Yeh KH, Baba E, Naito Y, Calvo F, Saxena A, Chen LT, Takeda M, Cervantes A, Taniguchi H, Yoshida K, Kodera Y, Kitagawa Y, Tabernero J, Burris H, Douillard JY. JSCO-ESMO-ASCO-JSMO-TOS: international expert consensus recommendations for tumour-agnostic treatments in patients with solid tumours with microsatellite instability or NTRK fusions. Ann Oncol, 31:861-872, 2020
43. Kijima T, Fukushima H, Kusuhara S, Tanaka H, Yoshida S, Yokoyama M, Ishioka J, Matsuoka Y, Numao N, Sakai Y, Saito K, Matsubara N, Yuasa T, Masuda H, Yonese J, Kageyama Y, Fujii Y. Association Between the Occurrence and Spectrum of Immune-Related Adverse Events and Efficacy of Pembrolizumab in Asian Patients With Advanced Urothelial Cancer: Multicenter Retrospective Analyses and Systematic Literature Review. Clin Genitourin Cancer, 2020
44. Yamaguchi T, Inoue K, Tsunoda H, Uematsu T, Shinohara N, Mukai H. A deep learning-based automated diagnostic system for classifying mammographic lesions. Medicine (Baltimore), 99:e20977, 2020
45. Iwamoto T, Hara F, Uemura Y, Mukai H, Watanabe T, Ohashi Y. NSAS-BC02 substudy of chemotherapy-induced amenorrhea (CIA) in premenopausal patients who received either taxane alone or doxorubicin(A) cyclophosphamide(C) followed by taxane as postoperative chemotherapy. Breast Cancer Res Treat, 182:325-332, 2020
46. Saito T, Makiura D, Inoue J, Doi H, Yakushijin K, Okamura A, Matsuoka H, Mukohara T, Saura R, Sakai Y, Ono R. Comparison between quantitative and subjective assessments of chemotherapy-induced peripheral neuropathy in cancer patients: A prospective cohort study. Phys Ther Res, 23:166-171, 2020
47. Suzuki H, Shin T, Fukasawa S, Hashine K, Kitani S, Ohtake N, Shibayama K, Tran N, Mundle S, Fizazi K, Matsubara N. Efficacy and safety of abiraterone acetate plus prednisone in Japanese patients with newly diagnosed, metastatic hormone-naive prostate cancer: final subgroup analysis of LATITUDE, a randomized, double-blind, placebo-controlled, phase 3 study. Jpn J Clin Oncol, 50:810-820, 2020
48. Sasaki A, Harano K, Kogawa T, Matsubara N, Naito Y, Hosono A, Mukai H, Yoshino T, Mukohara T. Intestinal Perforation due to Neutropenic Enterocolitis in a Patient Treated with Bevacizumab for Ovarian Cancer. Case Rep Oncol Med, 2020:7231358, 2020
49. Mukai H, Yamaguchi T, Takahashi M, Hozumi Y, Fujisawa T, Ohsumi S, Akabane H, Nishimura R, Takashima T, Park Y, Sagara Y, Toyama T, Imoto S, Mizuno T, Yamashita S, Fujii S, Uemura Y. Ki-67 response-guided preoperative chemotherapy for HER2-positive breast cancer: results of a randomised Phase 2 study. Br J Cancer, 122:1747-1753, 2020
50. Iwase T, Harano K, Masuda H, Kida K, Hess KR, Wang Y, Dirix L, Van Laere SJ, Lucci A, Krishnamurthy S, Woodward WA, Layman RM, Bertucci F, Ueno NT. Quantitative hormone receptor (HR) expression and gene expression analysis in HR+ inflammatory breast cancer (IBC) vs non-IBC. BMC Cancer, 20:430, 2020
51. Kawazoe A, Kuboki Y, Bando H, Fukuoka S, Kojima T, Naito Y, Iino S, Yodo Y, Doi T, Shitara K, Yoshino T. Phase 1 study of napabucasin, a cancer stemness inhibitor, in patients with advanced solid tumors. Cancer Chemother Pharmacol, 85:855-862, 2020
52. Chin M, Yokoyama R, Sumi M, Okita H, Kawai A, Hosono A, Koga Y, Sano H, Watanabe H, Ozaki T, Mugishima H. Multimodal treatment including standard chemotherapy with vincristine, doxorubicin, cyclophosphamide, ifosfamide, and etoposide for the Ewing sarcoma family of tumors in Japan: Results of the Japan Ewing Sarcoma Study 04. Pediatr Blood Cancer, 67:e28194, 2020
53. Matsumoto K, Takahashi M, Sato K, Osaki A, Takano T, Naito Y, Matsuura K, Aogi K, Fujiwara K, Tamura K, Baba M, Tokunaga S, Hirano G, Imoto S, Miyazaki C, Yanagihara K, Imamura CK, Chiba Y, Saeki T. A double-blind, randomized, multicenter phase 3 study of palonosetron vs granisetron combined with dexamethasone and fosaprepitant to prevent chemotherapy-induced nausea and vomiting in patients with breast cancer receiving anthracycline and cyclophosphamide. Cancer Med, 9:3319-3327, 2020
54. Tsuda H. Histological classification of breast tumors in the General Rules for Clinical and Pathological Recording of Breast Cancer (18th edition). Breast Cancer, 27:309-321, 2020
55. Matsubara N, Chi KN, ?zg?ro?lu M, Rodriguez-Antolin A, Feyerabend S, Fein L, Alekseev BY, Sulur G, Protheroe A, Li S, Mundle S, De Porre P, Tran N, Fizazi K. Correlation of Prostate-specific Antigen Kinetics with Overall Survival and Radiological Progression-free Survival in Metastatic Castration-sensitive Prostate Cancer Treated with Abiraterone Acetate plus Prednisone or Placebos Added to Androgen Deprivation Therapy: Post Hoc Analysis of Phase 3 LATITUDE Study. Eur Urol, 77:494-500, 2020
56. Matsubara N, Kimura G, Uemura H, Uemura H, Nakamura M, Nagamori S, Mizokami A, Kikukawa H, Hosono M, Kinuya S, Krissel H, Siegel J, Kakehi Y. A randomized, double-blind, comparison of radium-223 and placebo, in combination with abiraterone acetate and prednisolone, in castration-resistant metastatic prostate cancer: subgroup analysis of Japanese patients in the ERA 223 study. Int J Clin Oncol, 25:720-731, 2020
57. Takehara K, Yamashita N, Watanabe R, Teramoto N, Tsuda H, Motohashi T, Harano K, Nakanishi T, Tokunaga H, Susumu N, Ueda Y, Yokoyama Y, Saito T. Clinical status and prognostic factors in Japanese patients with uterine leiomyosarcoma. Gynecol Oncol, 157:115-120, 2020
58. Tap WD, Wagner AJ, Sch?ffski P, Martin-Broto J, Krarup-Hansen A, Ganjoo KN, Yen CC, Abdul Razak AR, Spira A, Kawai A, Le Cesne A, Van Tine BA, Naito Y, Park SH, Fedenko A, P?pai Z, Soldatenkova V, Shahir A, Mo G, Wright J, Jones RL. Effect of Doxorubicin Plus Olaratumab vs Doxorubicin Plus Placebo on Survival in Patients With Advanced Soft Tissue Sarcomas: The ANNOUNCE Randomized Clinical Trial. JAMA, 323:1266-1276, 2020
59. Inoue M, Naito Y, Kogawa T, Kusuhara S, Fukasawa Y, Fukasawa Y, Harano K Matsubara N, Hosono A, Mukohara T. Safety and Efficacy of Palbociclib in Male Metastatic Breast Cancer: A Report of Two Cases. Ann Case Report, 14:416, 2020
