Annual Report 2020
Department of Clinical Laboratories
Nobuo Kuninaka, Hiroshi Yamakawa, Masahiro Karibe, Yuko Adegawa, Noriaki Sato, Masaki Takeda, Kumi Horiuchi, Kenta Akie, Masaru Kanno, Nobuyuki Nakamura, Michiko Iida, Saki Sunohara, Yuki Takahashi, Tomofumi Tan, Keisei Nakamura, Koudai Tamaki, Akira Miyaura, Yasuharu Hashimoto, Michiteru Yamagishi, Yuichiro Yazaki, Rana Goto, Momoka Sakayori, Takuya Hanazawa, Ayaka Takahashi, Keiko Nakai, Ayumi Nakanishi, Misato Nojiri, Risako Ueda, Hitomi Nakamura, Sayuri Shibayama, Tomomi Wachi, Takuya Yamaguchi, Mari Hisano, Masaharu Tsunoda, Mitsunori Tajima, Ito Masayuki, Riko Kawarai, Yuka Tachihara, Mika Narikiyo, Takuya Aiba, Ayumi Setsuda, Takaki Kobayashi, Chiharu Eda, Kentaro Yamada, Masayuki Sukekawa, Shouta Ooishi, Yuma Furuya, Masahiro Inoue, Hikari Sekiguchi, Aiko Kimura, Yumiko Ito, Ayumi Iwaya, Aya Koike, Mika Sasanuma, Tomoko Ikeda, Tomoe Katakura, Kaori Toyama, Ayaka Ohashi, Yoriko Furusawa, Izumi Suzuki, Haruka Nozaki, Megumi Yamaguchi, Mariko Kinoshita, Yasuko Nakazeko, Chiho Furuya, Noriko Sato, Mana Shimamura, Kanako Nakayama, Yumiko Follett
Introduction
Our laboratory testing operations, including urinalysis and other general testing, hematology, biochemistry, and immunology, have been shifted in-house from being outsourced, and the entire laboratory has functioned independently since October 2018. In FY2020, our clinical laboratory was launched independent of the department of pathology and clinical laboratory. Our clinical laboratory performs eight testing types, namely hematology/urinalysis and other general testing, biochemistry and immunology, blood transfusion, bacteriology, physiology, pathology and genetic testing, and supporting laboratory testing in clinical studies. We will continue to perform clinical testing operations, including pathology in cooperation with the department of pathology and clinical laboratory.
The Team and What We Do
With the spread of the novel coronavirus (SARS-CoV-2) infection in 2020, we promptly initiated PCR screening tests for high-risk surgical patients in April. With the introduction of LUMIPULSE®G600, we initiated quantitative antigen tests for all inpatients and PCR tests for clinical trial patients in September. Consequently, we processed a large number of specimens and with 1,316 PCR tests and 7,068 quantitative antigen tests performed we contributed to the preventive measures against the spread of hospital infections (Figure 1).
The number of annual clinical tests performed in our laboratory has increased, with a total of 4,112,632 clinical tests, amounting to a 2% increase from the previous year (Table 1). In addition, the number of clinical trial tests in each field showed an increasing trend, with an increase from the previous year of 7% for pharmacokinetics (PK), 3% for electrocardiogram, 26% for fundus examination, and 18% for the preparation of pathological specimens (Table 2).
Figure 1. Number of PCR tests and quantitative antigen tests performed in each month of FY2020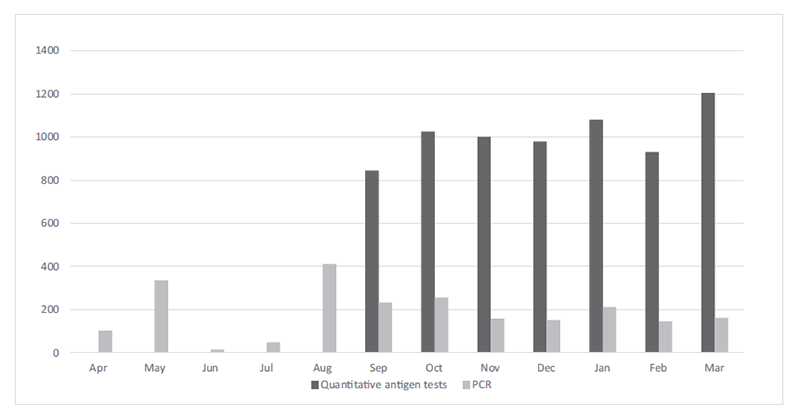

Table 1. Number of clinical tests performed in FY2020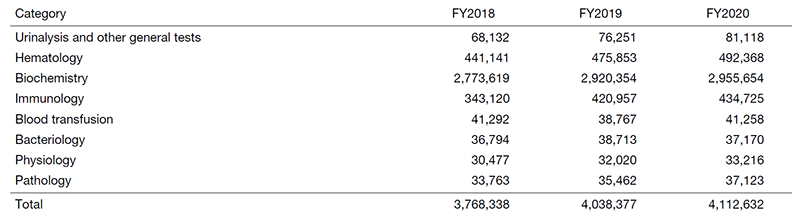

Table 2. Number of clinical trial tests performed in FY2020 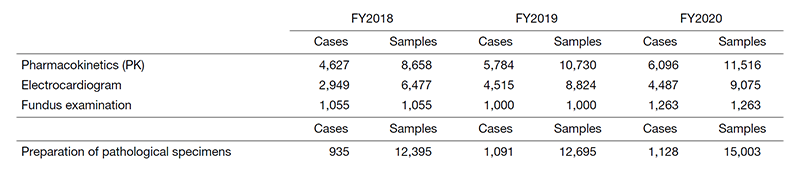

To perform quality control and to ensure the technological capability of the laboratory, we have established a system to carry out all testing operations, including laboratory tests, physiological function tests, clinical research tests (clinical trial), and pathological tests, in accordance with the international standard ISO15189.
Research activities
- Usage of liquid-based cytology (LBC) instead of the conventional method: Based on our observations, fine-needle aspiration cytology performed in the Department of Head and Neck Surgery is entirely by LBC, except for suspected malignant lymphoma, and has become a daily operation.
- Introduction of rapid testing by Ultrafast Papanicolaou (UFP) staining and the introduction of LBC into the endoscopic ultrasound-fine needle aspiration (EUS-FNA) area: The conventional Diff-Quick staining has been replaced by UFP, and we have introduced UFP for rapid testing. In addition, specimens can now be collected in vials at the clinical site, which has led to the introduction of LBC.
- Consideration of the conditions for immunostaining on cytological specimens (particularly the head and neck area): Immunostaining of cytological specimens is needed for cases in which tissue biopsy is clinically difficult.
Education
We have established an education and training system based on the requirements of the ISO15189 (medical laboratories) certification of the Japan Accreditation Board (JAB). We have had study sessions, workshops, and lectures once a month to improve the academic knowledge, skills, and ethical conduct of our staff. Moreover, our laboratory made efforts to improve staff skills by designing an educational program tailored to each skill level. As we improve our skills, the proficiency level is determined by two approaches: self-evaluation and evaluation by evaluators, using an operation comprehension check sheet.
In FY2020, we gave 10 presentations at academic conferences and submitted a paper. We worked with commitment for our participation in conferences, from the preparation of abstracts to presentation rehearsals, and we received an academic society award at the 48th conference of the Japanese Association for National Hospital Medical Technologists, Kanshin Branch (September 2020), which recognized the effort that we invest in our daily activities. Our laboratory personnel are fully aware of the role of the national center and have been working to improve their skills and knowledge. We will continue to raise the standards of our clinical laboratory through the three pillars of daily operations, education, and research.
Furthermore, we actively accepted clinical trainees. Paying sufficient attention to infection control measures during the coronavirus (COVID-19) crisis, we accepted four trainees from Tsukuba International University and one from Daito Bunka University and have been working on the betterment of the next generation.
Future Prospects
The clinical laboratory forms the basis of not only daily medical care, but also of medical research. By maintaining and continuously adhering to the international standard certification of ISO15189, we guarantee the quality of medical care, clinical trials, and research at the National Cancer Center Hospital East (NCCHE). Moreover, with the aim of obtaining CAP (College of American Pathologists) certification in August 2022, we have started various programs. We will strengthen our cooperation with other departments of the NCCHE and the Exploratory Oncology Research & Clinical Trial Center, and will actively participate in the development of advanced tests, new drugs, and devices in the future. Our efforts will contribute to the achievement of the vision of NCCHE “Providing the world’s best cancer care and creating novel world-class cancer care.”
