Annual Report 2020
Department of Colorectal Surgery
Yukihide Kanemitsu, Shunsuke Tsukamoto, Konosuke Moritani, Yasuyuki Takamizawa, Manabu Inoue
Introduction
The Department of Colorectal Surgery deals with colorectal cancer and allied malignancies in the colon and rectum. Liver metastasis from colorectal cancer is treated in cooperation with the Department of Hepatobiliary and Pancreatic Surgery. Lung metastasis from colorectal cancer is also treated in cooperation with the Department of Thoracic Surgery. Although surgery is still the main treatment modality for colorectal cancer, multidisciplinary treatments including radiotherapy and chemotherapy are important in advanced cancer. We have multidisciplinary meetings with the Department of Gastrointestinal Medical Oncology, the Department of Endoscopy, the Department of Diagnostic Radiology, and the Department of Pathology and Clinical Laboratories every week, and decide the treatment strategy with a multidisciplinary team (MDT) before treatment is performed.
The Team and What We Do
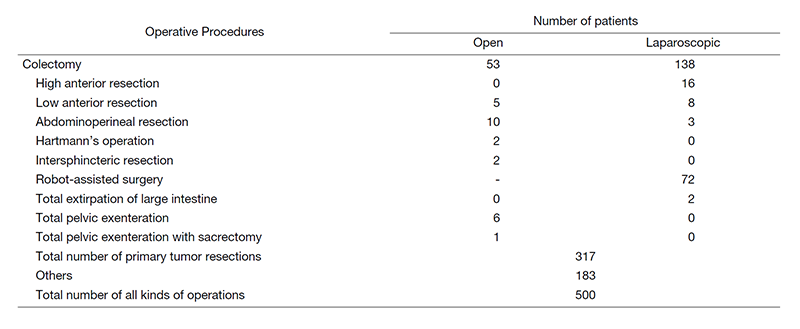
In the Department of Colorectal Surgery, we have performed more than 600 colorectal operations in a year, which is top of the class in Japan. In FY2020, we performed more than 500 operations under the COVID-19 disaster. We always aim to improve the survival rate with safe surgery associated with less complications. The length of hospital stay is as short as seven days after surgery in laparotomy, laparoscopic surgery, and robot-assisted surgery, which allows more patients to receive surgical treatment with fewer beds.
There are five staff surgeons, one chief resident, and four to seven rotating residents. Every morning (7:15-8:30), we have a morning conference and rounds in wards 15A and B. An MDT meeting is held for cancer patients as a form of institutionalized communication every Tuesday morning (7:15-8:00), in which colorectal surgeons, liver surgeons, lung surgeons, medical oncologists, radiologists, and pathologists participate to determine the best treatment plan. Every Monday morning (7:15-8:00), we hold a review meeting for cases scheduled for surgery the following week to confirm the staging of the disease and carefully simulate the surgery. Twelve to fifteen operations are performed a week in our department.
- Robot-assisted surgery
In our department, we have been performing robot-assisted rectal surgeries, in which robotic surgery technology has been applied to rectal cancer treatment since March 2014. It is considered that rectal cancer surgery using the Da Vinci surgical system enables more intricate and precise operations since the doctor performs surgery with robotic support. It also enables safer and less invasive surgery since accurate three-dimensional image data can be obtained. The field of robot-assisted surgery is expected to play an important role in next-generation surgery. As of July 2021, we have performed more than 300 robotic surgeries and have achieved excellent results with a very low complication rate.
Research activities
Patients with advanced rectal cancers are treated with conventional surgery. Adjuvant chemotherapy is being used in stage III colorectal cancer patients in a clinical setting. Although preoperative radiotherapy is not performed routinely for advanced rectal cancer, patients with T4b rectal cancers or rectal cancers with multiple lymph node metastases are treated with preoperative chemoradiotherapy and surgery. Patients with symptoms caused by unresectable tumors are treated with palliative surgery including palliative resection, bypass, and stoma before chemotherapy. To evaluate the survival benefit and safety of primary resection plus chemotherapy compared to chemotherapy alone in asymptomatic stage IV colorectal cancer with synchronous unresectable metastatic disease, a randomized controlled trial comparing resection of primary tumor plus chemotherapy with chemotherapy alone in incurable stage IV colorectal cancer is ongoing (JCOG1007, iPACS). Another randomized controlled trial is ongoing to evaluate the non-inferiority of overall survival of laparoscopic surgery to open surgery for palliative resection of primary tumor in incurable stage IV colorectal cancer (JCOG1107, ENCORE). Symptomatic, stage IV colorectal cancer patients with non-curable metastasis are pre-operatively randomized to either open or laparoscopic colorectal resection. Patients with resectable liver metastasis are treated in cooperation with the Department of Hepatobiliary and Pancreatic Surgery and adjuvant chemotherapy regimens are being evaluated in a clinical trial (JCOG0603 study). To confirm the superiority of perioperative chemotherapy, a randomized phase II/III trial started in May 2015 comparing perioperative versus postoperative chemotherapy with modified infusional fluorouracil and folinic acid with oxaliplatin (mFOLFOX6) for lower rectal cancer patients with suspected lateral pelvic node metastasis (JCOG1310). The registration for JCOG1502C (a study to examine adjuvant chemotherapy for small bowel adenocarcinoma) and JCOG1503C (a study to examine the efficacy of aspirin for stage III colorectal cancer) began in 2017 and 2018, respectively. The registration for JCOG1801 (a study to examine neoadjuvant chemoradiotherapy for locally recurrent rectal cancer) began in 2019. In addition, a translational study (JCOG1506A1) has been initiated to compare the clinical data from previous large-scale clinical trials including more than 4,000 patients and the genetic analysis data of clinical specimens such as surgical specimens and peripheral blood, in collaboration with the BioBank Japan (BBJ), toward improvement in treatment outcome and individualization of treatment. The registration for JCOG1805 (a study to examine adjuvant chemotherapy for stage II colorectal cancer patients at high risk of developing recurrence) began in 2020.
We also carry out basic research in cooperation with scientists at the National Cancer Center Research Institute (NCCRI) and the identification of a suitable treatment based on prediction is one of our important goals.
Clinical trials
Our department plays a central role in conducting multi-institutional clinical trials in Japan. Y. Kanemitsu is a representative of the Colorectal Cancer Group of the Japan Clinical Oncology Group (JCOG). Our department is participating in nine phase III JCOG studies.
1. JCOG0603: A randomized study that compares adjuvant modified FOLFOX (5-FU + l-LV + Oxaliplatin) to surgery alone after hepatic resection for liver metastasis from colorectal cancer. One hundred and seventy patients have been enrolled and recruitment continues.
2. JCOG1007: A randomized controlled trial comparing resection of primary tumor plus chemotherapy with chemotherapy alone in incurable stage IV colorectal cancer was terminated as of June 2019.
3. JCOG1018: Randomized phase III study of mFOLFOX7 or CAPOX plus bevacizumab versus 5-fluorouracil/leucovorin or capecitabine plus bevacizumab as first-line treatment in elderly patients with metastatic colorectal cancer is ongoing.
4. JCOG1107: A randomized controlled trial comparing laparoscopic surgery with open surgery in palliative resection of primary tumor in incurable stage IV colorectal cancer is ongoing.
5. JCOG1506A: Prognostic or predictive biomarker study in patients who underwent surgery with/without postoperative chemotherapy for stage II/III colorectal cancer is ongoing.
6. JCOG1502C: A Global Study to Evaluate the Potential Benefit of Adjuvant Chemotherapy for Small Bowel Adenocarcinoma is ongoing.
7. JCOG1503C: Efficacy of aspirin for stage III colorectal cancer: a randomized double-blind placebo-controlled trial is ongoing.
8. JCOG1801: A randomized controlled trial comparing surgery plus adjuvant chemotherapy with preoperative chemoradiotherapy followed by surgery plus adjuvant chemotherapy for locally recurrent rectal cancer is ongoing.
9. JCOG1805: A randomized controlled trial to examine efficacy of adjuvant chemotherapy for stage II colorectal cancer patients at high risk of developing recurrence according to T-stage and three selected pathological factors.
10. FANTASTIC study: a phase II study to evaluate the safety and efficacy of modified-FOLFOXIRI and a potential novel biomarker-driven strategy using ctDNA for CRC, after resection of oligometastases.
For JCOG1007, the results of the primary analysis were reported at the American Society of Clinical Oncology Gastrointestinal Cancer Symposium (ASCO-GI) in January 2020, and later published in the Journal of Clinical Oncology. The results of the main analysis of JCOG0603 were also reported at the American Society of Clinical Oncology (ASCO) in May 2020, and were later published in the Journal of Clinical Oncology.
Education
In our resident doctor education, under the supervision of a supervising physician, the residents are trained not only in the acquisition and improvement of skills, but also in a series of medical procedures such as judgment of the indications for surgery, selection of surgical techniques, postoperative management, experience as a primary surgeon or assistant surgeon after gaining experience, and postoperative treatment. In addition, we actively instruct resident doctors in the preparation of reports and papers for various research groups and conferences. As part of these efforts, video conferences to review surgical techniques and research conferences to check the progress of research and provide guidance are held regularly.
Future Prospects
Although Japan has been leading the world in the development of new drugs such as irinotecan and oxaliplatin, it is far behind in the development of molecular targeted drugs. While the gap is being filled by international collaborative clinical trials, there is no guarantee that innovative new drugs will continue to dramatically improve treatment outcomes of cancer patients in the future. Therefore, it is a big yet important challenge for clinical oncologists to think and act strategically. Outside of Japan, novel studies using the data from many clinical trials and the data from specimen examination have been carried out one after another. We believe that all the specialists involved in colorectal cancer treatment must work together with their wisdom and experience in order to improve treatment outcome. We will reconfirm the department’s research policy that a clinical question is reviewed by all participants and an answer is obtained from a randomized controlled trial, and continue to actively promote clinical trials in the future.
At the National Cancer Center Hospital (NCCH), a national research project working on the development of a new laparoscopic surgical system and the utilization of high definition image data using 8K Super Hi-Vision technology has been initiated. With this project, further improvement of the treatment outcome of patients with colorectal cancer is expected in the future.
List of papers published in 2020
Journal
1. Wada S, Sadahiro R, Matsuoka YJ, Uchitomi Y, Yamaguchi T, Sato T, Shimada K, Yoshimoto S, Daiko H, Kanemitsu Y, Kawai A, Kato T, Fujimoto H, Shimizu K. Yokukansan for Treatment of Preoperative Anxiety and Prevention of Postoperative Delirium in Cancer Patients Undergoing Highly Invasive Surgery. J-SUPPORT 1605 (ProD Study): A Randomized, Double-Blind, Placebo-Controlled Trial. J Pain Symptom Manage, 61:71-80, 2021
2. Hirano H, Takashima A, Hamaguchi T, Shida D, Kanemitsu Y. Current status and perspectives of immune checkpoint inhibitors for colorectal cancer. Jpn J Clin Oncol, 51:10-19, 2021
3. Mazaki J, Tsukamoto S, Miyake M, Moritani K, Sakamoto R, Shida D, Kanemitsu Y. Circumferential Resection Margin Status as a Predictive Factor for Recurrence in Preoperative MRI for Advanced Lower Rectal Cancer Without Preoperative Therapy. Dis Colon Rectum, 64:71-80, 2021
4. Tanaka K, Ogawa G, Mizusawa J, Kadota T, Nakamura K, Shimada Y, Hamaguchi T, Fujita S, Kitano S, Inomata M, Kanemitsu Y, Fukuda H. Second primary cancers and recurrence in patients after resection of colorectal cancer: An integrated analysis of trials by Japan Clinical Oncology Group: JCOG1702A. Jpn J Clin Oncol, 51:185-191, 2021
5. Yasui K, Shida D, Nakamura Y, Ahiko Y, Tsukamoto S, Kanemitsu Y. Postoperative, but not preoperative, inflammation-based prognostic markers are prognostic factors in stage III colorectal cancer patients. Br J Cancer, 124:933-941, 2021
6. Horie T, Shida D, Ahiko Y, Takamizawa Y, Inoue M, Tanabe T, Nakamura Y, Imaizumi J, Tsukamoto S, Kanemitsu Y. Laparoscopic versus Open Colectomy for Elderly Patients with Colon Cancer: A Propensity Score Analysis with the Controlling Nutritional Status (CONUT) Score. Nutr Cancer, 73:246-251, 2021
7. Tsukamoto S, Fujita S, Ota M, Mizusawa J, Shida D, Kanemitsu Y, Ito M, Shiomi A, Komori K, Ohue M, Akazai Y, Shiozawa M, Yamaguchi T, Bando H, Tsuchida A, Okamura S, Akagi Y, Takiguchi N, Saida Y, Akasu T, Moriya Y. Long-term follow-up of the randomized trial of mesorectal excision with or without lateral lymph node dissection in rectal cancer (JCOG0212). Br J Surg, 107:586-594, 2020
8. Kadota T, Ikematsu H, Sasaki T, Saito Y, Ito M, Mizutani T, Ogawa G, Shitara K, Ito Y, Kushima R, Kanemitsu Y, Muto M. Protocol for a single-arm confirmatory trial of adjuvant chemoradiation for patients with high-risk rectal submucosal invasive cancer after local resection: Japan Clinical Oncology Group Study JCOG1612 (RESCUE study). BMJ Open, 10:e034947, 2020
9. Kadota T, Tsukada Y, Ito M, Katayama H, Mizusawa J, Nakamura N, Ito Y, Bando H, Ando M, Onaya H, Fukuda H, Kanemitsu Y. A phase III randomized controlled trial comparing surgery plus adjuvant chemotherapy with preoperative chemoradiotherapy followed by surgery plus adjuvant chemotherapy for locally recurrent rectal cancer: Japan Clinical Oncology Group study JCOG1801 (RC-SURVIVE study). Jpn J Clin Oncol, 50:953-957, 2020
10. Yokota T, Saito Y, Takamaru H, Sekine S, Nakajima T, Yamada M, Sakamoto T, Taniguchi H, Kushima R, Tsukamoto S, Shida D, Kanemitsu Y, Matsuda T. Spontaneous Regression of Mismatch Repair-Deficient Colon Cancer: A Case Series. Clin Gastroenterol Hepatol, 2020
11. Kato MK, Shida D, Yoneoka Y, Yoshida H, Miyasaka N, Kanemitsu Y, Kato T. Novel classification of ovarian metastases originating from colorectal cancer by radiological imaging and macroscopic appearance. Int J Clin Oncol, 25:1663-1671, 2020
12. Miyamoto K, Wakabayashi M, Mizusawa J, Nakamura K, Katayama H, Higashi T, Inomata M, Kitano S, Fujita S, Kanemitsu Y, Fukuda H. Evaluation of the representativeness and generalizability of Japanese clinical trials for localized rectal/colon cancer: Comparing participants in the Japan Clinical Oncology Group study with patients in Japanese registries. Eur J Surg Oncol, 46:1642-1648, 2020
13. Shimoyama R, Hijioka S, Mizuno N, Ogawa G, Kataoka T, Katayama H, Machida N, Honma Y, Boku N, Hamaguchi T, Fukuda H, Terashima M, Kanemitsu Y, Furuse J. Study protocol for a multi-institutional randomized phase III study comparing combined everolimus plus lanreotide therapy and everolimus monotherapy in patients with unresectable or recurrent gastroenteropancreatic neuroendocrine tumors; Japan Clinical Oncology Group Study JCOG1901 (STARTER-NET study). Pancreatology, 20:1183-1188, 2020
14. Shida D, Inoue M, Tanabe T, Moritani K, Tsukamoto S, Yamauchi S, Sugihara K, Kanemitsu Y. Prognostic impact of primary tumor location in Stage III colorectal cancer-right-sided colon versus left-sided colon versus rectum: a nationwide multicenter retrospective study. J Gastroenterol, 55:958-968, 2020
15. Takamizawa Y, Shida D, Boku N, Nakamura Y, Ahiko Y, Yoshida T, Tanabe T, Takashima A, Kanemitsu Y. Nutritional and inflammatory measures predict survival of patients with stage IV colorectal cancer. BMC Cancer, 20:1092, 2020
16. Kanemitsu Y, Shida D, Tsukamoto S, Moritani K, Sakamoto R. Japanese Evidences on Nerve-Preserving Lateral Pelvic Lymh Node Dissection for Rectal Cancer: Major Historical Milestones and Clinical Impact: The Past, Present and Future. Clin Colon Rectal Surg, 33:349-354, 2020
17. Kanemitsu Y, Shitara K, Mizusawa J, et al: Primary tumor resection plus chemotherapy versus chemotherapy alone for colorectal cancer patients with asymptomatic, synchronous unresectable metastases (JCOG1007; iPACS): A randomized clinical trial. J Clin Oncol 39:1098-1107, 2021
