Annual Report 2021
Knowledge Integration Section
Yuichi Shiraishi, Ai Okada, Kenichi Chiba, Naoko Iida
Introduction
The Section of Knowledge Integration is constructing the Cancer Knowledge Database (CKDB) to control quality and advance the genomic diagnosis of cancer. The section coordinates with the information repository system developed by the Section of Cancer Genomics Repository and optimizes the CKDB for Japan to support the expert panels at core hospitals for cancer genomic medicine.
The Team and What We Do
-
Development of the Cancer Knowledge Database
The CKDB was developed and maintained to improve the quality of cancer genome testing. A curator team consisting of 32 clinical oncologists and bioinformaticians was organized to maintain the system, updating data from clinical trials, medicines and marker evidence twice a month. -
Providing C-CAT findings
We have provided expert panel C-CAT findings, a report summary of clinical trials, medicines and marker evidence to annotate the clinical significance of variants found by cancer panel testing performed by clinical laboratories. We have also established and maintained a management system to control the quality of C-CAT findings by standardizing the review process by expert reviewers, who are equivalent to M.D. or Ph.D. level.
Research activities
The CKDB was developed by collecting, standardizing and integrating the information of marker evidence, anti-cancer therapies and clinical trials. Using the CKDB, we constructed a system to generate C-CAT findings. We also developed the CKDB-portal to search, query and curate the knowledge stored in the CKDB. As of March 2022, information on 822 clinical trials, 1,366 medicines, and 23,874 items of marker evidence are stored in the CKDB. We signed non-disclosure agreements with six pharmaceutical companies to receive detailed information from 74 clinical trials between April 2021 and March 2022. A total of 16,058 C-CAT findings were provided to expert panels of core hospitals between April 2021 and March 2022.
Future prospects
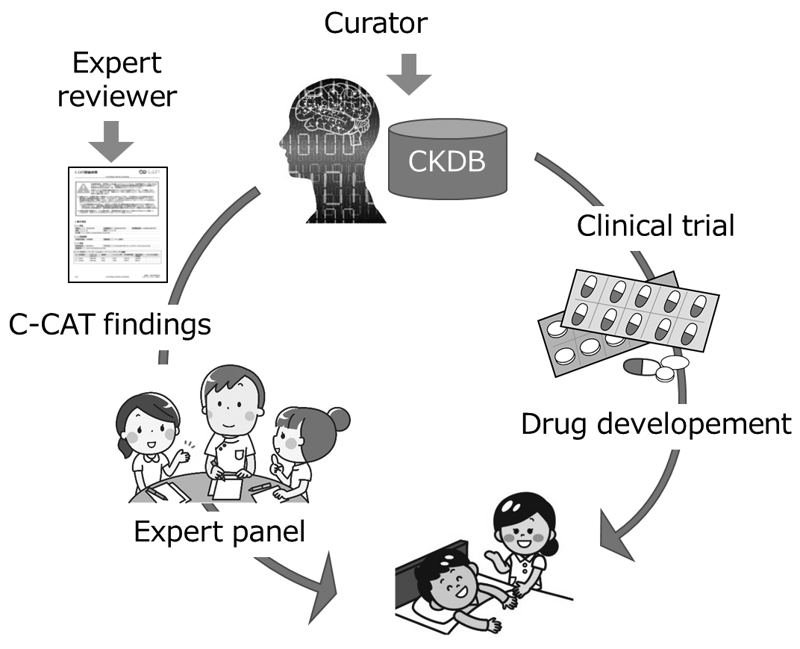
C-CAT findings support the expert panels with information on the newest evidence, anti-cancer therapies and clinical trials. As a result, genome medicine will be uniformly and efficiently promoted. Furthermore, the promotion of clinical trials will activate new drug development in Japan, leading to the discovery of evidence and contributing significantly to progress in cancer medicine (Figure 1).
Figure 1. Development of the CKDB to improve the quality of
genome medicines