Annual Report 2022
Cancer Screening Center
Nozomu Kobayashi, Masau Sekiguchi, Keiko Nakamura, Yasuo Kakugawa, Minori Matsumoto, Eriko Tsuruki, Masayoshi Yamada, Hiroyuki Takamaru, Takaaki Tsuchida, Masahiko Kusumoto, Gen Iinuma, Nachiko Uchiyama, Mari Kikuchi, Kimiteru Ito, Hiroaki Kurihara, Miyuki Sone, Yasunori Mizuguchi, Hirokazu Watanabe, Mototaka Miyake, Shunsuke Sugawara, Yuko Kubo, Chihiro Ito, Nao Kikkawa, Shintaro Kimura, Sawako Kaku, Mizuki Ozawa, Tomoyasu Kato, Mitsuya Ishikawa, Masaya Uno, Yasuhito Tanase, Mayumi Kato, Kenichi Nakamura, Hiroshi Katayama, Junko Eba, (Visiting Researcher) Takahisa Matsuda, Shuji Yamamoto, Hidetsugu Yamagishi, (Special Research Assistant) Mika Mori
Introduction
In the Cancer Screening Center (former name: Research Center for Cancer Prevention and Screening; RCCPS), we have provided broad opportunistic cancer screening using newly developed modalities since 2004. Most of the staff doctors hold two positions concurrently in both the Cancer Screening Center and their own specialized department. Our department is in charge of multiphasic cancer screening using several imaging modalities to develop new cancer screening systems and to assess new screening tests.
The Team and What We Do
1. Cancer screening courses: The basic plan for males consists of screening for cancer of the lungs, esophagus, stomach, colorectum, liver, gall bladder, pancreas, kidneys, and prostate. The basic plan for females consists of screening for cancer of the breast, uterus, and ovaries, in addition to the plan for males, excluding the prostate. In addition, PET (positron emission tomography) is provided as an option. In addition to multiphasic programs, an independent cancer screening program has been prepared for cancers of the lungs and female genitalia, including cancer of the uterus, ovaries, breast, and gastrointestinal tract. Blood samples are also obtained for biochemistry and tumor markers such as CA19-9, CEA, CA125, and PSA, as well as for genetic analysis.
2. Cancer screening methods: On the first day, the multiphasic cancer screening programs (comprehensive cancer screening program) perform CT for lung cancer, abdominal US for cancer of the liver, gall bladder, pancreas, and kidneys, gynecological examinations with pap-smear and HPV test for uterine cancer, and MMG/tomosynthesis and US for breast cancer. On the following day, gastroscopy for cancer of the esophagus and stomach, and total colonoscopy for cancer of the colon and rectum are conducted. Moreover, from the beginning of December 2010, CT-colonography (CTC) has been provided as an option for cancer screening. FDG-PET/CT is offered on the first day as an option, if the participants wish to undergo the examination. Furthermore, FDG-PET/MRI has been provided as an optional examination since 2018.
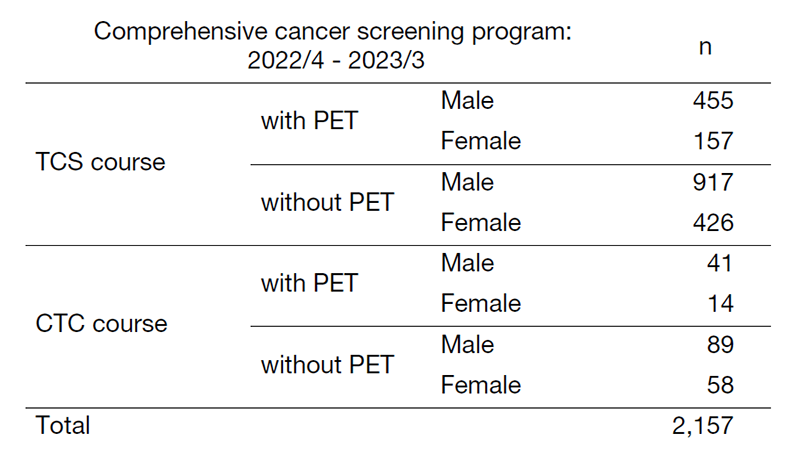
3. Number of cancer screening participants: The number of cancer screening participants between April 2022 and March 2023 is shown in this report (Table 1). Due to the influence of COVID-19, the number of participants decreased remarkably. A total of 2,386 people underwent cancer screening at the Cancer Screening Center during this period. Most of the participants (90%; n=2,157) chose the comprehensive cancer screening course. Regarding the cancer detection rate data in each modality, we will report them in the near future.
Research Activities
1. Amino-index (AICS) cancer screening accuracy evaluation study
Since the start of the research in July 2012, we have received plasma samples from 8,111 screening examinees and are proceeding with comprehensive amino acid analysis. This year, we confirmed the results of AICS in the patients with gastric cancer, using data of 6,532 individuals. Among 33 patients with gastric cancer, only five were classified as rank C, the highest risk group. The sensitivity and specificity of AICS for gastric cancer were 15% and 90%, respectively.
2. Study using colorectal cancer screening data from colonoscopy (CS)
The AP-Colon SL study is a prospective study of colorectal serrated lesions in patients undergoing colorectal cancer screening at multicenter sites in the Asia-Pacific region, aiming to determine the true prevalence of serrated lesions and to identify the risk factors for colorectal serrated lesions. For the total colonoscopy, the protocol stipulates that all patients will undergo right-sided colonoscopy with dye endoscopy using indigocarmine to more reliably detect serrated colorectal lesions and to determine their true prevalence. The trial is ongoing with a planned enrollment of 1,000 patients.
Clinical Trials
We are conducting ongoing research based on the study protocol titled “Evaluation of effectiveness of cancer screening modality at the National Cancer Center”. The target modalities are as follows: 1) upper gastrointestinal endoscopy, 2) lower gastrointestinal endoscopy, 3) CT colonography, 4) chest computed tomography (CT), 5) sputum cytology, 6) mammography, 7) breast ultrasonography, 8) FDG-positron emission tomography (PET), 9) abdominal ultrasonography, and 10) serum tumor markers.
Future Prospects
Based on cancer screening data such as examination results, medical institution findings, follow-up findings, and the questionnaire survey concerning lifestyles for 10 years, we commenced with assessment them with the support of the National Cancer Center Research and Development Fund.
List of papers published in 2022
Journal
1. Seki K, Sakamoto T, Makiguchi ME, Toyoshima N, Takamaru H, Sekiguchi M, Yamada M, Sekine S, Kanemitsu Y, Saito Y. Unexpected adhesive bowel obstruction after endoscopic submucosal dissection of early sigmoid colon cancer. DEN open, 3:e194, 2023
2. Suzuki H, Ono H, Hirasawa T, Takeuchi Y, Ishido K, Hoteya S, Yano T, Tanaka S, Toya Y, Nakagawa M, Toyonaga T, Takemura K, Hirasawa K, Matsuda M, Yamamoto H, Tsuji Y, Hashimoto S, Yuki M, Oyama T, Takenaka R, Yamamoto Y, Naito Y, Yamamoto K, Kobayashi N, Kawahara Y, Hirano M, Koizumi S, Hori S, Tajika M, Hikichi T, Yao K, Yokoi C, Ohnita K, Hisanaga Y, Sumiyoshi T, Kitamura S, Tanaka H, Shimoda R, Shimazu T, Takizawa K, Tanabe S, Kondo H, Iishi H, Ninomiya M, Oda I. Long-term Survival After Endoscopic Resection For Gastric Cancer: Real-world Evidence From a Multicenter Prospective Cohort. Clinical gastroenterology and hepatology, 21:307-318.e2, 2023
3. Hirata S, Toyoshima N, Takamaru H, Yamada M, Kobayashi N, Kozu T, Saito Y. Underwater endoscopic mucosal resection with submucosal injection. Endoscopy, 55:E70-E71, 2023
4. Hirai Y, Abe S, Makiguchi ME, Sekiguchi M, Nonaka S, Suzuki H, Yoshinaga S, Saito Y. Endoscopic Resection of Undifferentiated Early Gastric Cancer. Journal of gastric cancer, 23:146-158, 2023
5. Sekiguchi M, Westerberg M, Ekbom A, Hultcrantz R, Forsberg A. Endoscopist Characteristics and Polyp Detection in Colonoscopy: Cross-Sectional Analyses of Screening of Swedish Colons. Gastroenterology, 164:293-295.e4, 2023
6. Ishikawa H, Yamada M, Sato Y, Tanaka S, Akiko C, Tajika M, Doyama H, Takayama T, Ohda Y, Horimatsu T, Sano Y, Tanakaya K, Ikematsu H, Saida Y, Ishida H, Takeuchi Y, Kashida H, Kiriyama S, Hori S, Lee K, Tashiro J, Kobayashi N, Nakajima T, Suzuki S, Mutoh M. Intensive endoscopic resection for downstaging of polyp burden in patients with familial adenomatous polyposis (J-FAPP Study III): a multicenter prospective interventional study. Endoscopy, 55:344-352, 2023
7. Kajiwara Y, Oka S, Tanaka S, Nakamura T, Saito S, Fukunaga Y, Takamatsu M, Kawachi H, Hotta K, Ikematsu H, Kojima M, Saito Y, Yamada M, Kanemitsu Y, Sekine S, Nagata S, Yamada K, Kobayashi N, Ishihara S, Saitoh Y, Matsuda K, Togashi K, Komori K, Ishiguro M, Kuwai T, Okuyama T, Ohuchi A, Ohnuma S, Sakamoto K, Sugai T, Katsumata K, Matsushita HO, Yamano HO, Eda H, Uraoka T, Akimoto N, Kobayashi H, Ajioka Y, Sugihara K, Ueno H. Nomogram as a novel predictive tool for lymph node metastasis in T1 colorectal cancer treated with endoscopic resection: a nationwide, multicenter study. Gastrointestinal endoscopy, 97:1119-1128.e5, 2023
8. Ohata K, Kobayashi N, Saito Y. Reply. Gastroenterology, 164:1342-1343, 2023
9. Sekiguchi M, Igarashi A, Toyoshima N, Takamaru H, Yamada M, Esaki M, Kobayashi N, Saito Y. Cost-effectiveness analysis of computer-aided detection systems for colonoscopy in Japan. Digestive endoscopy, 2023
10. Lui RN, Wong SH, Ding NS, Sekiguchi M, Yu J, Ang TL, Yeoh KG, Chiu HM, Sung JJY. Is this the end of colonoscopy screening for colorectal cancer? An Asia-Pacific perspective. Journal of gastroenterology and hepatology, 38:671-677, 2023
11. Koyama Y, Fukuzawa M, Kono S, Madarame A, Morise T, Uchida K, Yamaguchi H, Sugimoto A, Nagata N, Kawai T, Takamaru H, Sekiguchi M, Yamada M, Sakamoto T, Matsuda T, Saito Y, Itoi T. Diagnostic efficacy of the Japan NBI Expert Team classification with dual-focus magnification for colorectal tumors. Surgical endoscopy, 36:5032-5040, 2022
12. Kobayashi N, Takeuchi Y, Ohata K, Igarashi M, Yamada M, Kodashima S, Hotta K, Harada K, Ikematsu H, Uraoka T, Sakamoto N, Doyama H, Abe T, Katagiri A, Hori S, Michida T, Yamaguchi T, Fukuzawa M, Kiriyama S, Fukase K, Murakami Y, Ishikawa H, Saito Y. Outcomes of endoscopic submucosal dissection for colorectal neoplasms: Prospective, multicenter, cohort trial. Digestive endoscopy, 34:1042-1051, 2022
13. Takamaru H, Saito Y, Hammoud GM, Mizuguchi Y, Cho H, Sekiguchi M, Yamada M, Sakamoto T, Matsuda T. Comparison of postpolypectomy bleeding events between cold snare polypectomy and hot snare polypectomy for small colorectal lesions: a large-scale propensity score-matched analysis. Gastrointestinal endoscopy, 95:982-989.e6, 2022
14. Abe S, Sekiguchi M. It is time to tailor endoscopic resection for early gastric cancer: Evaluate not only lesion but also patient. Digestive endoscopy, 34:826-827, 2022
15. Ohata K, Kobayashi N, Sakai E, Takeuchi Y, Chino A, Takamaru H, Kodashima S, Hotta K, Harada K, Ikematsu H, Uraoka T, Murakami T, Tsuji S, Abe T, Katagiri A, Hori S, Michida T, Suzuki T, Fukuzawa M, Kiriyama S, Fukase K, Murakami Y, Ishikawa H, Saito Y. Long-term Outcomes After Endoscopic Submucosal Dissection for Large Colorectal Epithelial Neoplasms: A Prospective, Multicenter, Cohort Trial From Japan. Gastroenterology, 163:1423-1434.e2, 2022
16. Uozumi T, Saito Y, Toyoshima N, Takamaru H, Sekiguchi M, Yamada M, Abe S. Effective use of image-enhanced endoscopy and endoscopic submucosal dissection for multiple flat non-polypoid colorectal neoplasms. Endoscopy, 54:E684-E685, 2022
17. Koyama Y, Yamada M, Makiguchi ME, Sekiguchi M, Takamaru H, Sakamoto T, Kono S, Fukuzawa M, Sylvia Wu SY, Sugumaran A, Kawai T, Matsuda T, Itoi T, Saito Y. New scoring system to distinguish deep invasive submucosal and muscularis propria colorectal cancer during colonoscopy: a development and global multicenter external validation study (e-T2 Score). Gastrointestinal endoscopy, 96:321-329.e2, 2022
18. Sekiguchi M, Hotta K, Takeuchi Y, Tanaka S, Yamamoto H, Shinmura K, Harada K, Uraoka T, Hisabe T, Sano Y, Kondo H, Horimatsu T, Kikuchi H, Kawamura T, Nagata S, Yamamoto K, Tajika M, Tsuji S, Kusaka T, Okuyama Y, Yoshida N, Moriyama T, Hasebe A, So S, Kobara H, Kashida H, Miyanaga R, Kato S, Hayashi Y, Sada M, Fukuzawa M, Kato H, Takayama T, Konishi J, Matsushita HO, Narasaka T, Ohata K, Togashi K, Nakamura H, Moriichi K, Oda Y, Kanda N, Kuwai T, Terai S, Sanomura M, Kitamura S, Miyamoto H, Kiriyama S, Mizuno C, Saito Y, Sekine S, Ito S, Nakahira H, Oka S, Hayashi Y, Yoshimura K, Ishikawa H, Matsuda T. Characteristics of colorectal neuroendocrine tumors in patients prospectively enrolled in a Japanese multicenter study: a first report from the C-NET STUDY. Journal of gastroenterology, 57:547-558, 2022
19. Sekiguchi M, Matsuda T, Hotta K, Saito Y. Post-polypectomy surveillance: the present and the future. Clinical endoscopy, 55:489-495, 2022
20. Takamaru H, Stammers M, Yanagisawa F, Mizuguchi Y, Sekiguchi M, Yamada M, Sakamoto T, Matsuda T, Saito Y. Conditional inference tree models to perceive depth of invasion in T1 colorectal cancer. Surgical endoscopy, 36:9234-9243, 2022
21. Sung JJY, Chiu HM, Lieberman D, Kuipers EJ, Rutter MD, Macrae F, Yeoh KG, Ang TL, Chong VH, John S, Li J, Wu K, Ng SSM, Makharia GK, Abdullah M, Kobayashi N, Sekiguchi M, Byeon JS, Kim HS, Parry S, Cabral-Prodigalidad PAI, Wu DC, Khomvilai S, Lui RN, Wong S, Lin YM, Dekker E. Third Asia-Pacific consensus recommendations on colorectal cancer screening and postpolypectomy surveillance. Gut, 71:2152-2166, 2022
22. Sekiguchi M, Westerberg M, Ekbom A, Hultcrantz R, Forsberg A. Detection rates of colorectal neoplasia during colonoscopies and their associated factors in the SCREESCO study. Journal of gastroenterology and hepatology, 37:2120-2130, 2022
23. Kawamura T, Sekiguchi M, Takamaru H, Mizuguchi Y, Horiguchi G, Kato M, Kobayashi K, Sada M, Oda Y, Yokoyama A, Utsumi T, Tsuji Y, Ohki D, Takeuchi Y, Shichijo S, Ikematsu H, Matsuda K, Teramukai S, Kobayashi N, Matsuda T, Saito Y, Tanaka K. "Endoscopic" adenoma detection rate as a quality indicator of colonoscopy: First report from the J-SCOUT study. Digestive endoscopy, 2022
24. Sekiguchi M, Oda I, Morita S, Katai H, Yano T, Terashima M, Kataoka T, Muto M. Management of elderly patients with early gastric cancer in Japan. Japanese journal of clinical oncology, 52:425-432, 2022
