Annual Report 2023
Department of Head and Neck Medical Oncology
Makoto Tahara, Susumu Okano, Tomohiro Enokida, Takao Fujisawa, Ryutaro Onaga, Nobukazu Tanaka, Yuta Hoshi, Takuma Kishida
Introduction
The Department of Head and Neck Medical Oncology is engaged in the clinical management of patients with head and neck cancer (HNC), and research into anticancer drugs for the treatment of HNC.
Our missions are to: 1) provide the best evidence-based treatment; 2) promote the importance of supportive care in the treatment of patients with HNC; 3) facilitate the timely approval of new drugs by active participation in global clinical trials to eliminate the drug lag; 4) develop cutting-edge treatments; and 5) train experts in head and neck medical oncology.
The Team and What We Do
Our department consists of four physicians, two senior residents and three residents. We manage the treatment of patients with HNC who receive anticancer drugs. An estimated 60% of patients with HNC require a multidisciplinary approach, including surgery, radiotherapy, and chemotherapy. Given the increasing complexity of HNC management, we decide on the recommended treatment for patients who are referred to our institution at the weekly tumor board attended by a multidisciplinary team.
A total of 271 patients were referred to our department from April 2023 to March 2024 (Tables 1 and 2). The outpatient service of our department is available from Monday to Friday. We carefully follow patients during and after treatment and provide palliative chemotherapy as an outpatient service.
Table 1. Number of patients by site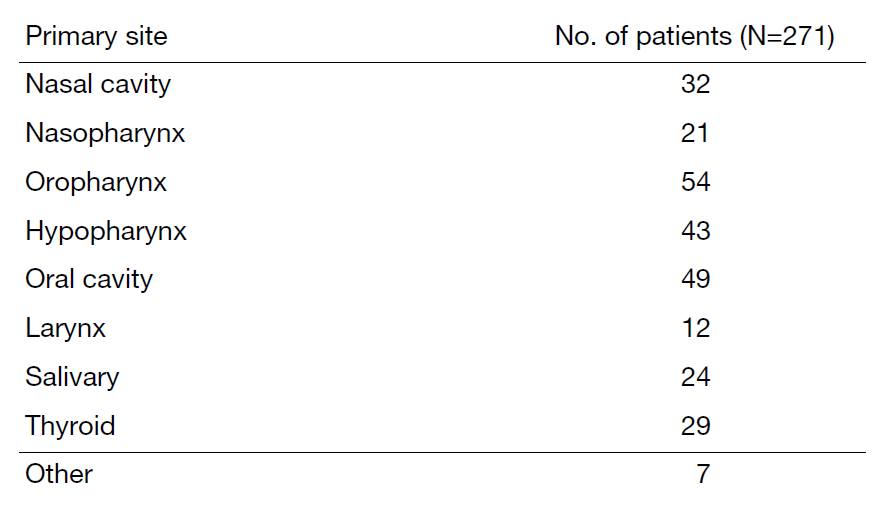

Table 2. Number of patients by procedure

Research Activities
Our research activity has focused on three areas, the development of new treatments in clinical trials for HNC, biomarker analysis in HNC and retrospective analysis of HNC treatment.
The retrospective review of 59 patients with recurrent or metastatic Squamous cell carcinoma of the head and neck (R/M SCCHN) treated with paclitaxel+cetuximab (PE)-based chemotherapy following disease progression after immune checkpoint inhibitor (Tanaka, Front Oncol, 2023) revealed promising clinical outcomes. The PE-based chemotherapy demonstrated an objective response rate (ORR) of 62.7%, a median progression-free survival (PFS) of 4.6 months, and a median overall survival (OS) of 17.1 months. Notably, the occurrence of grade 3 or worse adverse events was 40.7%, but no treatment-related death was observed. These results suggest that PE-based chemotherapy following immune checkpoint inhibitor is encouraging for its robust antitumor efficacy in R/M SCCHN, providing a potential treatment option for these patients.
Our retrospective assessment of 37 patients with R/M SCCHN from the oral cavity, oropharynx, hypopharynx, and larynx who received paclitaxel+CBDCA+cetuximab (PCE) as 1st-line treatment followed by nivolumab as 2nd-line at our institute between December 2016 and July 2021 (Takeshita N, Oral Oncol 2023) revealed promising results. The PCE regimen demonstrated an ORR of 48.6% and a median PFS of 4.4 months. In the nivolumab phase, the ORR was 10.8%, and the median PFS2, PFS3, and OS were 6.8, 11.6, and 19.5 months, respectively. These findings suggest that PCE followed by nivolumab shows a favorable survival outcome, offering hope for rapid tumor response with PCE and extension of OS by the addition of nivolumab regardless of combined positive score.
Our retrospective review of patients with salivary gland carcinoma treated with a taxane and platinum, which contained docetaxel plus cisplatin, or paclitaxel plus carboplatin, between January 2000 and September 2021 (Onaga, Front Oncol 2023) revealed reassuring results. of the 40 patients identified, 29 were treated with docetaxel plus cisplatin and 11 with paclitaxel plus carboplatin. The total population showed an ORR and median PFS of 37.5% and 5.4 months, respectively. On subgroup analysis, docetaxel plus cisplatin provided favorable efficacy compared with paclitaxel plus carboplatin (ORR: 46.5% vs. 20.0%, median PFS: 7.2 vs. 2.8 months). These findings suggest that the combination of taxane and platinum, particularly docetaxel plus cisplatin, is generally effective and well-tolerated for recurrent or metastatic salivary gland carcinoma, instilling confidence in its use.
Clinical Trials
The following investigator-initiated clinical trials are ongoing; 1) a phase 2 study of combination with nivolumab plus lenvatinib for unresectable anaplastic thyroid cancer and 2) a phase 2 study of darolutamide for androgen receptor positive recurrent or metastatic salivary gland carcinoma.
To facilitate the timely approval of new drugs and eliminate the drug lag, we have also participated in the global phase trials including immune-checkpoint inhibitors.
Education
We educate not only medical staff in our institute but also outside of our institute by conducting the education program, Seminar of the Japan Society of Supportive Care for patients with HNC. Furthermore, our department is accepting trainees at all time.
Future Prospects
We hope that ongoing or planned clinical trials will change the standard of care for HNC. Our biomarker analysis, a result of our collective efforts, will lead to the development of new treatment strategy. The success of our education program, aiming to increase the number of medical oncologists involved in HNC treatment, will result in improved patient survival quality.
List of papers published in 2023
Journal
1. Fujisawa T, Motegi A, Hirata H, Zenda S, Hojo H, Nakamura M, Oyoshi H, Tomizawa K, Zhou Y, Fukushi K, Kageyama SI, Enokida T, Okano S, Tahara M, Shinozaki T, Hayashi R, Matsuura K, Akimoto T. Long-term outcomes of patients with oral cavity cancer receiving postoperative radiotherapy after salvage neck dissection for cervical lymph node recurrence. Head & neck, 46:541-551, 2024
2. Uozumi S, Enokida T, Suzuki S, Nishizawa A, Kamata H, Okano T, Kawasaki T, Fujisawa T, Ueda Y, Okano S, Tahara M, Yamaguchi M. Efficacy and safety of adapalene gel as a reactive treatment for cetuximab-induced skin toxicity in recurrent or metastatic squamous cell carcinoma of the head and neck: A historical cohort comparison study. Journal of oncology pharmacy practice, 30:295-303, 2024
3. Hirai T, Naito Y, Koyama S, Nakanishi Y, Masuhiro K, Izumi M, Kuge T, Naito M, Mizuno Y, Yamaguchi Y, Kang S, Yaga M, Futami Y, Nojima S, Nishide M, Morita T, Kato Y, Tsuda T, Takemoto N, Kinugasa-Katayama Y, Aoshi T, Villa JK, Yamashita K, Enokida T, Hoshi Y, Matsuura K, Tahara M, Takamatsu H, Takeda Y, Inohara H, Kumanogoh A. Sema6D forward signaling impairs T cell activation and proliferation in head and neck cancer. JCI insight, 9:e166349, 2024
4. Hashimoto T, Nakamura Y, Mishima S, Nakayama I, Kotani D, Kawazoe A, Kuboki Y, Bando H, Kojima T, Iida N, Shibuki T, Imai M, Fujisawa T, Nagamine M, Sakamoto N, Kuwata T, Yoshino T, Shitara K. Whole-transcriptome sequencing in advanced gastric or gastroesophageal cancer: A deep dive into its clinical potential. Cancer science, 115:1622-1633, 2024
5. Sato M, Enokida T, Wada A, Okano S, Tanaka H, Fujisawa T, Ueda Y, Motegi A, Shinozaki T, Takeshita N, Tanaka N, Zenda S, Matsuura K, Akimoto T, Hayashi R, Tahara M. Potential efficacy of local therapy for progressive lesions after nivolumab in patients with recurrent or metastatic squamous cell carcinoma of the head and neck. International journal of clinical oncology, 28:1023-1032, 2023
6. Tanaka H, Enokida T, Okano S, Fujisawa T, Tanaka N, Takeshita N, Onaga R, Hoshi Y, Wada A, Sato M, Ueda Y, Tahara M. Subsequent chemotherapy with paclitaxel plus cetuximab-based chemotherapy following immune checkpoint inhibitor in recurrent or metastatic squamous cell carcinoma of the head and neck. Frontiers in oncology, 13:1221352, 2023
7. Udagawa H, Takahashi S, Hirao M, Tahara M, Iwasa S, Sato Y, Hamakawa T, Shitara K, Horinouchi H, Chin K, Masuda N, Suzuki T, Okumura S, Takase T, Nagai R, Yonemori K. Liposomal eribulin for advanced adenoid cystic carcinoma, gastric cancer, esophageal cancer, and small cell lung cancer. Cancer medicine, 12:1269-1278, 2023
8. Harrington KJ, Burtness B, Greil R, Soulières D, Tahara M, de Castro G Jr, Psyrri A, Brana I, Basté N, Neupane P, Bratland Å, Fuereder T, Hughes BGM, Mesia R, Ngamphaiboon N, Rordorf T, Wan Ishak WZ, Lin J, Gumuscu B, Swaby RF, Rischin D. Pembrolizumab With or Without Chemotherapy in Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma: Updated Results of the Phase III KEYNOTE-048 Study. Journal of clinical oncology, 41:790-802, 2023
9. Haddad RI, Harrington K, Tahara M, Ferris RL, Gillison M, Fayette J, Daste A, Koralewski P, Zurawski B, Taberna M, Saba NF, Mak M, Kawecki A, Girotto G, Alvarez Avitia MA, Even C, Toledo JGR, Guminski A, Müller-Richter U, Kiyota N, Roberts M, Khan TA, Miller-Moslin K, Wei L, Argiris A. Nivolumab Plus Ipilimumab Versus EXTREME Regimen as First-Line Treatment for Recurrent/Metastatic Squamous Cell Carcinoma of the Head and Neck: The Final Results of CheckMate 651. Journal of clinical oncology, 41:2166-2180, 2023
10. Ferris RL, Harrington K, Schoenfeld JD, Tahara M, Esdar C, Salmio S, Schroeder A, Bourhis J. Inhibiting the inhibitors: Development of the IAP inhibitor xevinapant for the treatment of locally advanced squamous cell carcinoma of the head and neck. Cancer treatment reviews, 113:102492, 2023
11. Wada A, Enokida T, Okano S, Sato M, Tanaka H, Ueda Y, Fujisawa T, Takeshita N, Tanaka N, Tahara M. Proton pump inhibitors and antibiotics adversely effect the efficacy of nivolumab in patients with recurrent or metastatic squamous cell carcinoma of the head and neck. European journal of cancer (Oxford, England : 1990), 184:30-38, 2023
12. Shibutani Y, Suzuki S, Sagara A, Enokida T, Okano S, Fujisawa T, Sato F, Yumoto T, Sano M, Kawasaki T, Tahara M. Impact of lenvatinib-induced proteinuria and renal dysfunction in patients with thyroid cancer. Frontiers in oncology, 13:1154771, 2023
13. Chatterjee S, Kiyota N, Vaish R, Sharma A, Tahara M, Noronha V, Prabhash K, D'Cruz A. Weekly versus 3-weekly cisplatin along with radiotherapy for locoregionally advanced non-nasopharyngeal head and neck cancers: Is the equipoise in literature addressed yet? Head & neck, 45:1594-1603, 2023
14. Harrington KJ, Ferris RL, Gillison M, Tahara M, Argiris A, Fayette J, Schenker M, Bratland Å, Walker JWT, Grell P, Even C, Chung CH, Redman R, Coutte A, Salas S, Grant C, de Azevedo S, Soulières D, Hansen AR, Wei L, Khan TA, Miller-Moslin K, Roberts M, Haddad R. Efficacy and Safety of Nivolumab Plus Ipilimumab vs Nivolumab Alone for Treatment of Recurrent or Metastatic Squamous Cell Carcinoma of the Head and Neck: The Phase 2 CheckMate 714 Randomized Clinical Trial. JAMA oncology, 9:779-789, 2023
15. Haddad RI, Harrington K, Tahara M, Szturz P, Le Tourneau C, Salmio S, Bajars M, Lee NY. Managing cisplatin-ineligible patients with resected, high-risk, locally advanced squamous cell carcinoma of the head and neck: Is there a standard of care? Cancer treatment reviews, 119:102585, 2023
16. Onaga R, Enokida T, Ito K, Ueda Y, Okano S, Fujisawa T, Wada A, Sato M, Tanaka H, Takeshita N, Tanaka N, Hoshi Y, Tahara M. Combination chemotherapy with taxane and platinum in patients with salivary gland carcinoma: a retrospective study of docetaxel plus cisplatin and paclitaxel plus carboplatin. Frontiers in oncology, 13:1185198, 2023
17. Hoshi Y, Shirakura S, Yamada M, Sugiyama T, Koide N, Tamii S, Kamata K, Yokomura M, Osaki S, Ohno T, Yagihara K, Hara H, Beppu T. Site of distant metastasis affects the prognosis with recurrent/metastatic head and neck squamous cell carcinoma patients treated with Nivolumab. International journal of clinical oncology, 28:1139-1146, 2023
18. Kummar S, Shen L, Hong DS, McDermott R, Keedy VL, Casanova M, Demetri GD, Dowlati A, Melcón SG, Lassen UN, Leyvraz S, Liu T, Moreno V, Patel J, Patil T, Mallick AB, Sousa N, Tahara M, Ziegler DS, Norenberg R, Arvis P, Brega N, Drilon A, Tan DSW. Larotrectinib efficacy and safety in adult patients with tropomyosin receptor kinase fusion sarcomas. Cancer, 129:3772-3782, 2023
19. Horinouchi A, Enokida T, Suzuki S, Kamata H, Kaneko A, Matsuyama C, Fujisawa T, Ueda Y, Ito K, Okano S, Kawasaki T, Tahara M. A pharmacist-led opioid de-escalation program after completion of chemoradiotherapy in locally advanced head and neck cancer. Frontiers in oncology, 13:1145323, 2023
20. Hadoux J, Elisei R, Brose MS, Hoff AO, Robinson BG, Gao M, Jarzab B, Isaev P, Kopeckova K, Wadsley J, Führer D, Keam B, Bardet S, Sherman EJ, Tahara M, Hu MI, Singh R, Lin Y, Soldatenkova V, Wright J, Lin B, Maeda P, Capdevila J, Wirth LJ. Phase 3 Trial of Selpercatinib in Advanced RET-Mutant Medullary Thyroid Cancer. The New England journal of medicine, 389:1851-1861, 2023
21. Matsuyama C, Enokida T, Ueda Y, Suzuki S, Fujisawa T, Ito K, Okano S, Tahara M. Planned drug holidays during treatment with lenvatinib for radioiodine-refractory differentiated thyroid cancer: a retrospective study. Frontiers in oncology, 13:1139659, 2023
22. Shibutani Y, Tajiri K, Suzuki S, Enokida T, Sagara A, Okano S, Fujisawa T, Sato F, Yumoto T, Sano M, Kawasaki T, Tahara M. Association between baseline blood pressure and the incidence of lenvatinib-induced hypertension in patients with thyroid cancer. Cancer medicine, 12:20773-20782, 2023
23. Takeshita N, Enokida T, Okano S, Fujisawa T, Wada A, Sato M, Tanaka H, Tanaka N, Onaga R, Hoshi Y, Sakashita S, Ishii G, Tahara M. Weekly paclitaxel, carboplatin and cetuximab (PCE) combination followed by nivolumab for recurrent and/or metastatic squamous cell carcinoma of the head and neck (R/M SCCHN). Oral oncology, 147:106615, 2023
