Annual Report 2023
Department of Medical Oncology
Toru Mukohara, Ako Hosono, Yoichi Naito, Nobuaki Matsubara, Kenichi Harano, Chihiro Kondoh, Chikako Funasaka, Hiromichi Nakajima, Hirofumi Mukai, Takehiro Nakao, Nobuyuki Takahashi, Misao Fukuda, Akira Hirota, Mai Shimura, Mao Uematsu
Introduction
Our department is the only department in the hospital that provides care for cross-organ cancers, mainly breast cancer, urologic cancer, gynecologic cancer, and bone and soft tissue sarcoma. We also provide appropriate treatment for malignancies that are difficult to treat at other facilities, such as cancers of unknown primary, multiple, and rare cancers, utilizing our abundant experience as an oncologist. We also play a central role in conferences that include surgical departments and radiation therapy departments, such as the "Urologic Tumor Conference," "Bone and Soft Tissue Sarcoma Conference," "Gynecologic Tumor Conference," and "Breast Tumor Conference," and implement multidisciplinary treatment.
The Team and What We Do
In terms of medical care, the number of new patients was comparable to the record high in FY2021, and the average number of patients treated per day was the highest ever recorded. In addition, we were deeply involved in the operation of the cancer gene profiling test under the insurance reimbursement system as in the previous fiscal year. Dr. Mukohara, our department's chief, served as the head of the expert panel. In total, 286 (41%) of the 704 cases discussed by the expert panel in FY2023 were handled by our department. In addition, 189 cases were from collaborating hospitals, contributing to the implementation of genomic medicine in our hospital and other regions. In terms of research, we actively enrolled cases in company-initiated clinical trials, presented at international conferences, and had opportunities to be the first author and co-author of English-language journals.
Research Activities
We are conducting both retrospective research and prospective clinical trials, new drug development trials, and related translational research (TR). The randomized, double-blind, investigator-initiated clinical trial of limb cooling therapy for the prevention of peripheral neuropathy in breast cancer patients receiving paclitaxel therapy, has been completed. Enrollment in an investigator-initiated trial evaluating olaparib plus pembrolizumab as preoperative chemotherapy for HRD-positive advanced ovarian cancer has also been completed. The clinical trial results and its associated translational research applying single-cell RNA sequencing will be published soon. In addition, registration for the investigator-initiated clinical trial of niraparib + pimitespib and joint research with a company on circulating tumor cells (CTCs) are also underway. In addition, in collaboration with basic research laboratories, we are conducting in vitro drug sensitivity tests for a β-catenin inhibitor and a study to establish a platform for ex vivo drug efficacy study using malignant pleural fluid or ascites. Further, we are conducting a drug efficacy study of ADCs using a brain metastasis model of breast cancer.
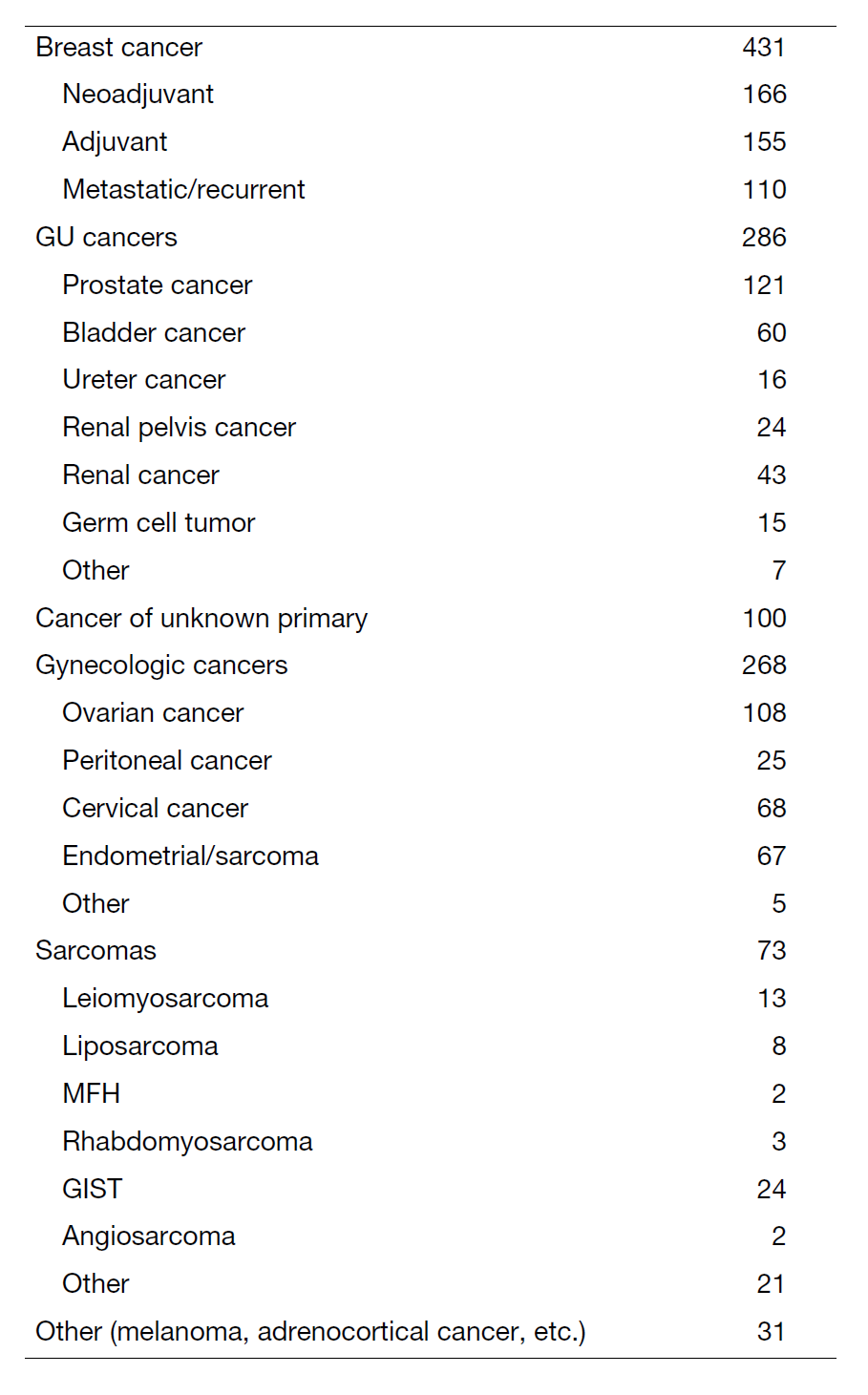
Clinical Trials
IITs we are leading:
- Phase I Trial of Niraparib and Pimitespib in Patients with Solid Tumors (PI, Naito Y): jRCT2031220179
- A randomized Control trial to Evaluate mitigation of CIPN by limb-cooling apparatus in breast cancer patients who undergo weekly paclitaxel (CECILIA) (PI, Mukohara T): jRCT2032210115
- Phase I clinical study to evaluate the safety and tolerability of intraperitoneal administration of anti-GPC3-CAR expressing iPS cell-derived ILC/NK cells in patients with inoperable advanced recurrent ovarian clear cell carcinoma expressing GPC3 and with peritoneal dissemination (PI, Harano K): jRCT2033200431
- Double-blind, placebo-controlled, randomized, phase III trial of paclitaxel plus carboplatin plus atezolizumab in patients with advanced/ recurrent uterine cancer (PI, Harano K): jRCT2031190013
- Olaparib Monotherapy and Olaparib + Pembrolizumab Combination Therapy for Ovarian Cancer (OLAPem) (PI, Harano K): NCT04417192
Education
The goal of our training and education is to develop true medical oncologists. Residents who graduate from our department are expected to be able to provide the standard of care for all types of cancer and to work collaboratively with other medical staff in the daily practice of medicine. They must also conduct research and write papers to address clinical questions that they identify during their residency. In FY2023, two papers were published in English with the residents as first authors under the supervision of our staff. In addition, four residents were given the opportunity to make presentations at the Japanese Society of Clinical Oncology and other conferences
Future Prospects
In the clinic, we will provide medical care with high patient satisfaction through multidisciplinary team medicine. In education, we will promote the training of medical oncologists who can treat patients across organ systems and scientific knowledge. In research, we will continue to lead development trials of new drugs from Phase I to Phase III. In addition, we will conduct preclinical research with the Genome TR laboratory on predicting drug efficacy and overcoming drug resistance, and aim to conduct investigator-initiated clinical trials based on the findings obtained. We will also continue to promote joint research with companies and other academic institutions.
List of papers published in 2023
Journal
1. Sunami K, Naito Y, Saigusa Y, Amano T, Ennishi D, Imai M, Kage H, Kanai M, Kenmotsu H, Komine K, Koyama T, Maeda T, Morita S, Sakai D, Hirata M, Ito M, Kozuki T, Sakashita H, Horinouchi H, Okuma Y, Takashima A, Kubo T, Hironaka S, Segawa Y, Yakushijin Y, Bando H, Makiyama A, Suzuki T, Kinoshita I, Kohsaka S, Ohe Y, Ishioka C, Yamamoto K, Tsuchihara K, Yoshino T. A Learning Program for Treatment Recommendations by Molecular Tumor Boards and Artificial Intelligence. JAMA oncology, 10:95-102, 2024
2. Doi T, Takahashi S, Aoki D, Yonemori K, Hara H, Hasegawa K, Takehara K, Harano K, Yunokawa M, Nomura H, Shimoi T, Horie K, Ogasawara A, Okame S. A first-in-human phase I study of TAS-117, an allosteric AKT inhibitor, in patients with advanced solid tumors. Cancer chemotherapy and pharmacology, 93:605-616, 2024
3. Matsubara N, de Wit R, Balar AV, Siefker-Radtke AO, Zolnierek J, Csoszi T, Shin SJ, Park SH, Atduev V, Gumus M, Su YL, Karaca SB, Cutuli HJ, Sendur MAN, Shen L, O'Hara K, Okpara CE, Franco S, Moreno BH, Grivas P, Loriot Y. Pembrolizumab with or Without Lenvatinib as First-line Therapy for Patients with Advanced Urothelial Carcinoma (LEAP-011): A Phase 3, Randomized, Double-Blind Trial. European urology, 85:229-238, 2024
4. Matsubara N, Matsuyama H, Kazama H, Seto T, Sunaga Y, Suzuki K. Baseline characteristics may impact treatment duration of cabazitaxel in patients with mCRPC: a subanalysis of data from a post-marketing surveillance. Japanese journal of clinical oncology, 54:97-102, 2024
5. Kelly WK, Danila DC, Lin CC, Lee JL, Matsubara N, Ward PJ, Armstrong AJ, Pook D, Kim M, Dorff TB, Fischer S, Lin YC, Horvath LG, Sumey C, Yang Z, Jurida G, Smith KM, Connarn JN, Penny HL, Stieglmaier J, Appleman LJ. Xaluritamig, a STEAP1 × CD3 XmAb 2+1 Immune Therapy for Metastatic Castration-Resistant Prostate Cancer: Results from Dose Exploration in a First-in-Human Study. Cancer discovery, 14:76-89, 2024
6. Yanagita M, Muto S, Nishiyama H, Ando Y, Hirata S, Doi K, Fujiwara Y, Hanafusa N, Hatta T, Hoshino J, Ichioka S, Inoue T, Ishikura K, Kato T, Kitamura H, Kobayashi Y, Koizumi Y, Kondoh C, Matsubara T, Matsubara K, Matsumoto K, Okuda Y, Okumura Y, Sakaida E, Shibagaki Y, Shimodaira H, Takano N, Uchida A, Yakushijin K, Yamamoto T, Yamamoto K, Yasuda Y, Oya M, Okada H, Nangaku M, Kashihara N. Clinical questions and good practice statements of clinical practice guidelines for management of kidney injury during anticancer drug therapy 2022. Clinical and experimental nephrology, 28:85-122, 2024
7. Agarwal N, Saad F, Azad AA, Mateo J, Matsubara N, Shore ND, Chakrabarti J, Chen HC, Lanzalone S, Niyazov A, Fizazi K. TALAPRO-3 clinical trial protocol: phase III study of talazoparib plus enzalutamide in metastatic castration-sensitive prostate cancer. Future oncology (London, England), 20:493-505, 2024
8. Arend RC, Monk BJ, Shapira-Frommer R, Haggerty AF, Alvarez EA, Amit A, Alvarez Secord A, Muller C, Casado Herraez A, Herzog TJ, Tewari KS, Cohen JG, Huang M, Yachnin A, Holeman LL, Ledermann JA, Rachmilewitz Minei T, Buyse M, Fain Shmueli S, Lavi M, Harats D, Penson RT. Ofranergene Obadenovec (Ofra-Vec, VB-111) With Weekly Paclitaxel for Platinum-Resistant Ovarian Cancer: Randomized Controlled Phase III Trial (OVAL Study/GOG 3018). Journal of clinical oncology, 42:170-179, 2024
9. Mateo J, de Bono JS, Fizazi K, Saad F, Shore N, Sandhu S, Chi KN, Agarwal N, Olmos D, Thiery-Vuillemin A, Özgüroğlu M, Mehra N, Matsubara N, Young Joung J, Padua C, Korbenfeld E, Kang J, Marshall H, Lai Z, Barnicle A, Poehlein C, Lukashchuk N, Hussain M. Olaparib for the Treatment of Patients With Metastatic Castration-Resistant Prostate Cancer and Alterations in BRCA1 and/or BRCA2 in the PROfound Trial. Journal of clinical oncology, 42:571-583, 2024
10. Kikuchi Y, Shimada H, Hatanaka Y, Kinoshita I, Ikarashi D, Nakatsura T, Kitano S, Naito Y, Tanaka T, Yamashita K, Oshima Y, Nanami T. Clinical practice guidelines for molecular tumor markers, 2nd edition review part 1. International journal of clinical oncology, 29:1-19, 2024
11. Fizazi K, Azad AA, Matsubara N, Carles J, Fay AP, De Giorgi U, Joung JY, Fong PCC, Voog E, Jones RJ, Shore ND, Dunshee C, Zschäbitz S, Oldenburg J, Ye D, Lin X, Healy CG, Di Santo N, Laird AD, Zohren F, Agarwal N. First-line talazoparib with enzalutamide in HRR-deficient metastatic castration-resistant prostate cancer: the phase 3 TALAPRO-2 trial. Nature medicine, 30:257-264, 2024
12. Nishiyama H, Yonese J, Kawahara T, Matsumoto R, Miyake H, Matsubara N, Uemura H, Eto M, Azuma H, Obara W, Terai A, Fukasawa S, Suekane S. TAS0313 plus Pembrolizumab for Post-Chemotherapy Immune Checkpoint Inhibitor-Naïve Locally Advanced or Metastatic Urothelial Carcinoma. Molecular cancer therapeutics, 23:532-540, 2024
13. Nishida T, Naito Y, Takahashi T, Saito T, Hisamori S, Manaka D, Ogawa K, Hirota S, Ichikawa H. Molecular and clinicopathological features of KIT/PDGFRA wild-type gastrointestinal stromal tumors. Cancer science, 115:894-904, 2024
14. Fizazi K, Azad AA, Matsubara N, Carles J, Fay AP, De Giorgi U, Joung JY, Fong PCC, Voog E, Jones RJ, Shore ND, Dunshee C, Zschäbitz S, Oldenburg J, Ye D, Lin X, Healy CG, Di Santo N, Laird AD, Zohren F, Agarwal N. Publisher Correction: First-line talazoparib with enzalutamide in HRR-deficient metastatic castration-resistant prostate cancer: the phase 3 TALAPRO-2 trial. Nature medicine, 2024
15. Dorff T, Horvath LG, Autio K, Bernard-Tessier A, Rettig MB, Machiels JP, Bilen MA, Lolkema MP, Adra N, Rottey S, Greil R, Matsubara N, Tan DSW, Wong A, Uemura H, Lemech C, Meran J, Yu Y, Minocha M, McComb M, Penny HL, Gupta V, Hu X, Jurida G, Kouros-Mehr H, Janát-Amsbury MM, Eggert T, Tran B. A Phase I Study of Acapatamab, a Half-life Extended, PSMA-Targeting Bispecific T-cell Engager for Metastatic Castration-Resistant Prostate Cancer. Clinical cancer research, 30:1488-1500, 2024
16. Takahashi K, Uozumi R, Mukohara T, Hayashida T, Iwabe M, Iihara H, Kusuhara-Mamishin K, Kitagawa Y, Tsuchiya M, Kitahora M, Nagayama A, Kosaka S, Asano-Niwa Y, Seki T, Ohnuki K, Suzuki A, Ono F, Futamura M, Kawazoe H, Nakamura T. Proton Pump Inhibitors and Cyclin-Dependent Kinase 4/6 Inhibitors in Patients With Breast Cancer. The oncologist, 29:e741-e749, 2024
17. Rosenberg JE, Mamtani R, Sonpavde GP, Loriot Y, Duran I, Lee JL, Matsubara N, Vulsteke C, Castellano D, Sridhar SS, Pappot H, Gurney H, Bedke J, van der Heijden MS, Galli L, Keam B, Masumori N, Meran J, O'Donnell PH, Park SH, Grande E, Sengeløv L, Uemura H, Skaltsa K, Campbell M, Matsangou M, Wu C, Hepp Z, McKay C, Powles T, Petrylak DP. Health-related Quality of Life in Patients with Previously Treated Advanced Urothelial Carcinoma from EV-301: A Phase 3 Trial of Enfortumab Vedotin Versus Chemotherapy. European urology, 85:574-585, 2024
18. Powles T, Valderrama BP, Gupta S, Bedke J, Kikuchi E, Hoffman-Censits J, Iyer G, Vulsteke C, Park SH, Shin SJ, Castellano D, Fornarini G, Li JR, Gümüş M, Mar N, Loriot Y, Fléchon A, Duran I, Drakaki A, Narayanan S, Yu X, Gorla S, Homet Moreno B, van der Heijden MS. Enfortumab Vedotin and Pembrolizumab in Untreated Advanced Urothelial Cancer. The New England journal of medicine, 390:875-888, 2024
19. Funasaka C, Hanai A, Zenda S, Mori K, Fukui M, Hirano N, Shinohara R, Fuse N, Wakabayashi M, Itagaki M, Tomioka Y, Nishina M, Arai Y, Kogawa T, Ozaki Y, Nishimura M, Kobayashi T, Hara F, Takano T, Mukohara T. Mitigation of paclitaxel-induced peripheral neuropathy in breast cancer patients using limb-cooling apparatus: a study protocol for a randomized controlled trial. Frontiers in oncology, 13:1216813, 2023
20. Sasada S, Kondo N, Hashimoto H, Takahashi Y, Terata K, Kida K, Sagara Y, Ueno T, Anan K, Suto A, Kanbayashi C, Takahashi M, Nakamura R, Ishiba T, Tsuneizumi M, Nishimura S, Naito Y, Hara F, Shien T, Iwata H. Prognostic impact of adjuvant endocrine therapy for estrogen receptor-positive and HER2-negative T1a/bN0M0 breast cancer. Breast cancer research and treatment, 202:473-483, 2023
21. Izawa N, Masuishi T, Takahashi N, Shoji H, Yamamoto Y, Matsumoto T, Sugiyama K, Kajiwara T, Kawakami K, Aomatsu N, Kondoh C, Kawakami H, Takegawa N, Esaki T, Shimokawa M, Nishio K, Narita Y, Hara H, Sunakawa Y, Boku N, Moriwaki T, Eguchi Nakajima T, Muro K. A Phase II Trial of Trifluridine/Tipiracil in Combination with Cetuximab Rechallenge in Patients with RAS Wild-Type mCRC Refractory to Prior Anti-EGFR Antibodies: WJOG8916G Trial. Targeted oncology, 18:369-381, 2023
22. Naito Y, Mishima S, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of tropomyosin receptor kinase inhibitors in adult and pediatric patients with neurotrophic receptor tyrosine kinase fusion-positive advanced solid tumors. International journal of clinical oncology, 28:827-840, 2023
23. Mishima S, Naito Y, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of immunotherapy in patients with high tumor mutational burden tumors. International journal of clinical oncology, 28:941-955, 2023
24. Mishima S, Naito Y, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of immunotherapy in patients with DNA mismatch repair deficient (dMMR) tumors, third edition. International journal of clinical oncology, 28:1237-1258, 2023
25. Tamura K, Mukohara T, Yonemori K, Kawabata Y, Nicolas X, Tanaka T, Iwata H. Phase 1 study of oral selective estrogen receptor degrader (SERD) amcenestrant (SAR439859), in Japanese women with ER-positive and HER2-negative advanced breast cancer (AMEERA-2). Breast cancer (Tokyo, Japan), 30:506-517, 2023
26. Krop IE, Masuda N, Mukohara T, Takahashi S, Nakayama T, Inoue K, Iwata H, Yamamoto Y, Alvarez RH, Toyama T, Takahashi M, Osaki A, Saji S, Sagara Y, O'Shaughnessy J, Ohwada S, Koyama K, Inoue T, Li L, Patel P, Mostillo J, Tanaka Y, Sternberg DW, Sellami D, Yonemori K. Patritumab Deruxtecan (HER3-DXd), a Human Epidermal Growth Factor Receptor 3-Directed Antibody-Drug Conjugate, in Patients With Previously Treated Human Epidermal Growth Factor Receptor 3-Expressing Metastatic Breast Cancer: A Multicenter, Phase I/II Trial. Journal of clinical oncology, 41:5550-5560, 2023
27. Matsubara N, de Bono J, Sweeney C, Chi KN, Olmos D, Sandhu S, Massard C, Garcia J, Chen G, Harris A, Schenkel F, Sane R, Hinton H, Bracarda S, Sternberg CN. Safety Profile of Ipatasertib Plus Abiraterone vs Placebo Plus Abiraterone in Metastatic Castration-resistant Prostate Cancer. Clinical genitourinary cancer, 21:230-237.e1, 2023
28. Pongor LS, Schultz CW, Rinaldi L, Wangsa D, Redon CE, Takahashi N, Fialkoff G, Desai P, Zhang Y, Burkett S, Hermoni N, Vilk N, Gutin J, Gergely R, Zhao Y, Nichols S, Vilimas R, Sciuto L, Graham C, Caravaca JM, Turan S, Tsai-Wei S, Rajapakse VN, Kumar R, Upadhyay D, Kumar S, Kim YS, Roper N, Tran B, Hewitt SM, Kleiner DE, Aladjem MI, Friedman N, Hager GL, Pommier Y, Ried T, Thomas A. Extrachromosomal DNA Amplification Contributes to Small Cell Lung Cancer Heterogeneity and Is Associated with Worse Outcomes. Cancer discovery, 13:928-949, 2023
29. Naito Y, Nishida T, Doi T. Current status of and future prospects for the treatment of unresectable or metastatic gastrointestinal stromal tumours. Gastric cancer, 26:339-351, 2023
30. Baba K, Kawamoto M, Mamishin K, Uematsu M, Kiyohara H, Hirota A, Takahashi N, Fukuda M, Kusuhara S, Nakajima H, Funasaka C, Nakao T, Kondoh C, Harano K, Matsubara N, Naito Y, Hosono A, Kawasaki T, Mukohara T. The impact of the COVID-19 pandemic on perioperative chemotherapy for breast cancer. Cancer medicine, 12:12095-12105, 2023
31. Anthracycline-containing and taxane-containing chemotherapy for early-stage operable breast cancer: a patient-level meta-analysis of 100 000 women from 86 randomised trials. Lancet (London, England), 401:1277-1292, 2023
32. Ohsumi S, Watanabe K, Kondo N, Kosaka Y, Ishikawa T, Kitahara M, Kubo S, Oba MS, Kimura T, Takita A, Mukai H. Venous thromboembolism in Japanese patients with breast cancer: subgroup analysis of the Cancer-VTE Registry. Breast cancer (Tokyo, Japan), 30:607-616, 2023
33. Iwase T, Saji S, Iijima K, Higaki K, Ohtani S, Sato Y, Hozumi Y, Hasegawa Y, Yanagita Y, Takei H, Tanaka M, Masuoka H, Tanabe M, Egawa C, Komoike Y, Nakamura T, Ohtsu H, Mukai H. Postoperative Adjuvant Anastrozole for 10 or 5 Years in Patients With Hormone Receptor-Positive Breast Cancer: AERAS, a Randomized Multicenter Open-Label Phase III Trial. Journal of clinical oncology, 41:3329-3338, 2023
34. Doi T, Matsubara N, Naito Y, Kuboki Y, Harano K, Ono M, Urasaki T, Ohmoto A, Kawanai T, Hisai T, Ikezawa H, Shiba S, Ito K, Semba T, Asano O, Takahashi S. First-in-human study of E7130 (a tumor microenvironment-ameliorating microtubule inhibitor) in patients with advanced solid tumors: Primary results of the dose-escalation part. Cancer, 129:2348-2359, 2023
35. Abel ML, Takahashi N, Peer C, Redon CE, Nichols S, Vilimas R, Lee MJ, Lee S, Shelat M, Kattappuram R, Sciuto L, Pinkiert D, Graham C, Butcher D, Karim B, Sharma AK, Malin J, Kumar R, Schultz CW, Goyal S, Del Rivero J, Krishnamurthy M, Upadhyay D, Schroeder B, Sissung T, Tyagi M, Kim J, Pommier Y, Aladjem M, Raffeld M, Figg WD, Trepel J, Xi L, Desai P, Thomas A. Targeting Replication Stress and Chemotherapy Resistance with a Combination of Sacituzumab Govitecan and Berzosertib: A Phase I Clinical Trial. Clinical cancer research, 29:3603-3611, 2023
36. Agarwal N, Azad AA, Carles J, Fay AP, Matsubara N, Heinrich D, Szczylik C, De Giorgi U, Young Joung J, Fong PCC, Voog E, Jones RJ, Shore ND, Dunshee C, Zschäbitz S, Oldenburg J, Lin X, Healy CG, Di Santo N, Zohren F, Fizazi K. Talazoparib plus enzalutamide in men with first-line metastatic castration-resistant prostate cancer (TALAPRO-2): a randomised, placebo-controlled, phase 3 trial. Lancet (London, England), 402:291-303, 2023
37. Matsuyama H, Matsubara N, Kazama H, Seto T, Sunaga Y, Suzuki K. Real-world effectiveness of third-line cabazitaxel in patients with metastatic castration-resistant prostate cancer: CARD-like analysis of data from a post-marketing surveillance in Japan. BMC cancer, 23:538, 2023
38. George S, Heinrich MC, Somaiah N, Oppelt P, McLeod R, Nishioka S, Kundu MG, Qian X, Kumar P, Laadem A, Lau Y, Tran BP, Fallon M, Dosunmu O, Shi J, Naito Y. A Phase I, Multicenter, Open-Label, First-in-Human Study of DS-6157a in Patients with Advanced Gastrointestinal Stromal Tumor. Clinical cancer research, 29:3659-3667, 2023
39. Arai N, Hattori N, Yamashita S, Liu YY, Ebata T, Takeuchi C, Takeshima H, Fujii S, Kondo H, Mukai H, Ushijima T. HSD17B4 methylation enhances glucose dependence of BT-474 breast cancer cells and increases lapatinib sensitivity. Breast cancer research and treatment, 201:317-328, 2023
40. Ishikura K, Omae K, Sasaki S, Shibagaki Y, Ichioka S, Okuda Y, Koitabashi K, Suyama K, Mizukami T, Kondoh C, Hirata S, Matsubara T, Hoshino J, Yanagita M. Chapter 4: CKD treatment in cancer survivors, from Clinical Practice Guidelines for the Management of Kidney Injury During Anticancer Drug Therapy 2022. International journal of clinical oncology, 28:1333-1342, 2023
41. Ohba K, Monji K, Osawa T, Yamana K, Yasuda Y, Tanaka H, Nakagawa Y, Fukuyama T, Matsubara N, Uemura H, Sakai H, Eto M. Comprehensive investigation of clinicopathological and immunological features to determine prognostic impact in metastatic renal cell carcinoma: The JEWEL study. International journal of urology, 30:977-984, 2023
42. Schultz CW, Zhang Y, Elmeskini R, Zimmermann A, Fu H, Murai Y, Wangsa D, Kumar S, Takahashi N, Atkinson D, Saha LK, Lee CF, Elenbaas B, Desai P, Sebastian R, Sharma AK, Abel M, Schroeder B, Krishnamurthy M, Kumar R, Roper N, Aladjem M, Zenke FT, Ohler ZW, Pommier Y, Thomas A. ATR inhibition augments the efficacy of lurbinectedin in small-cell lung cancer. EMBO molecular medicine, 15:e17313, 2023
43. Takada M, Imoto S, Ishida T, Ito Y, Iwata H, Masuda N, Mukai H, Saji S, Ikeda T, Haga H, Saeki T, Aogi K, Sugie T, Ueno T, Ohno S, Ishiguro H, Kanbayashi C, Miyamoto T, Hagiwara Y, Toi M. A risk-based subgroup analysis of the effect of adjuvant S-1 in estrogen receptor-positive, HER2-negative early breast cancer. Breast cancer research and treatment, 202:485-496, 2023
44. Uematsu M, Nakajima H, Hosono A, Kiyohara H, Hirota A, Takahashi N, Fukuda M, Kusuhara S, Nakao T, Funasaka C, Kondoh C, Harano K, Matsubara N, Naito Y, Akimoto T, Mukohara T. Safety of immune checkpoint inhibitors after proton beam therapy in head and neck mucosal melanoma: a case series. Melanoma research, 33:547-552, 2023
45. Masuda J, Sakai H, Tsurutani J, Tanabe Y, Masuda N, Iwasa T, Takahashi M, Futamura M, Matsumoto K, Aogi K, Iwata H, Hosonaga M, Mukohara T, Yoshimura K, Imamura CK, Miura S, Yamochi T, Kawabata H, Yasojima H, Tomioka N, Yoshimura K, Takano T. Efficacy, safety, and biomarker analysis of nivolumab in combination with abemaciclib plus endocrine therapy in patients with HR-positive HER2-negative metastatic breast cancer: a phase II study (WJOG11418B NEWFLAME trial). Journal for immunotherapy of cancer, 11:e007126, 2023
46. Terada M, Ito A, Kikawa Y, Koizumi K, Naito Y, Shimoi T, Ishihara M, Yamanaka T, Ozaki Y, Hara F, Nakamura R, Hattori M, Miyashita M, Kondo N, Yoshinami T, Takada M, Matsumoto K, Narui K, Sasada S, Iwamoto T, Hosoda M, Takano Y, Oba T, Sakai H, Murakami A, Higuchi T, Tsuchida J, Tanabe Y, Shigechi T, Tokuda E, Harao M, Kashiwagi S, Mase J, Watanabe J, Nagai SE, Yamauchi C, Yamamoto Y, Iwata H, Saji S, Toyama T. The Japanese Breast Cancer Society Clinical Practice Guidelines for systemic treatment of breast cancer, 2022 edition. Breast cancer (Tokyo, Japan), 30:872-884, 2023
47. Takahashi N, Hao Z, Villaruz LC, Zhang J, Ruiz J, Petty WJ, Mamdani H, Riess JW, Nieva J, Pachecho JM, Fuld AD, Shum E, Chauhan A, Nichols S, Shimellis H, McGlone J, Sciuto L, Pinkiert D, Graham C, Shelat M, Kattappuram R, Abel M, Schroeder B, Upadhyay D, Krishnamurthy M, Sharma AK, Kumar R, Malin J, Schultz CW, Goyal S, Redon CE, Pommier Y, Aladjem MI, Gore SD, Steinberg SM, Vilimas R, Desai P, Thomas A. Berzosertib Plus Topotecan vs Topotecan Alone in Patients With Relapsed Small Cell Lung Cancer: A Randomized Clinical Trial. JAMA oncology, 9:1669-1677, 2023
48. Loriot Y, Matsubara N, Park SH, Huddart RA, Burgess EF, Houede N, Banek S, Guadalupi V, Ku JH, Valderrama BP, Tran B, Triantos S, Kean Y, Akapame S, Deprince K, Mukhopadhyay S, Stone NL, Siefker-Radtke AO. Erdafitinib or Chemotherapy in Advanced or Metastatic Urothelial Carcinoma. The New England journal of medicine, 389:1961-1971, 2023
49. Jones RH, Fizazi K, James ND, Tammela TL, Matsubara N, Priou F, Beuzeboc P, Lesimple T, Bono P, Kataja V, Garcia JA, Protheroe A, Shore N, Aspegren J, Joensuu H, Kuss I, Fiala-Buskies S, Vjaters E. Safety and tolerability of long-term treatment with darolutamide in patients with metastatic castration-resistant prostate cancer. Prostate cancer and prostatic diseases, 2023
50. Radiotherapy to regional nodes in early breast cancer: an individual patient data meta-analysis of 14 324 women in 16 trials. Lancet (London, England), 402:1991-2003, 2023
51. Misawa S, Denda T, Kodama S, Suzuki T, Naito Y, Kogawa T, Takada M, Suichi T, Shiosakai K, Kuwabara S. Efficacy and safety of mirogabalin for chemotherapy-induced peripheral neuropathy: a prospective single-arm trial (MiroCIP study). BMC cancer, 23:1098, 2023
52. Takahashi M, Cortés J, Dent R, Pusztai L, McArthur H, Kümmel S, Denkert C, Park YH, Im SA, Ahn JH, Mukai H, Huang CS, Chen SC, Kim MH, Jia L, Li XT, Tryfonidis K, Karantza V, Iwata H, Schmid P. Pembrolizumab Plus Chemotherapy Followed by Pembrolizumab in Patients With Early Triple-Negative Breast Cancer: A Secondary Analysis of a Randomized Clinical Trial. JAMA network open, 6:e2342107, 2023
