Annual Report 2023
Department of Hepatobiliary and Pancreatic Surgery
Naoto Gotohda, Shin Kobayashi, Motokazu Sugimoto, Masashi Kudo, Masaru Konishi, Yusuke Abe, Kimimasa Sasaki, Yu Igata, Yu Shibahara, Norikazu Une, Taiki Sunakawa, Keisiro Aoki, Masaaki Kagoura, Tsuyoshi Terada, Hirokazu Okamoto, Shota Kami
Introduction
The Department of Hepatobiliary and Pancreatic Surgery consists of five staff surgeons, four chief residents and seven residents. Our department is responsible for the surgical treatment of patients with hepatic, biliary, pancreatic and duodenal cancer or low-grade malignant tumors. We conduct multidisciplinary treatment in cooperation with the Department of Hepatobiliary and Pancreatic Oncology, the Department of Diagnostic Radiology, and the Department of Radiation Oncology. In addition to conventional open surgery, we perform minimally invasive surgery including laparoscopic and robot-assisted surgery for patients with liver and pancreatic tumors.
The Team and What We Do
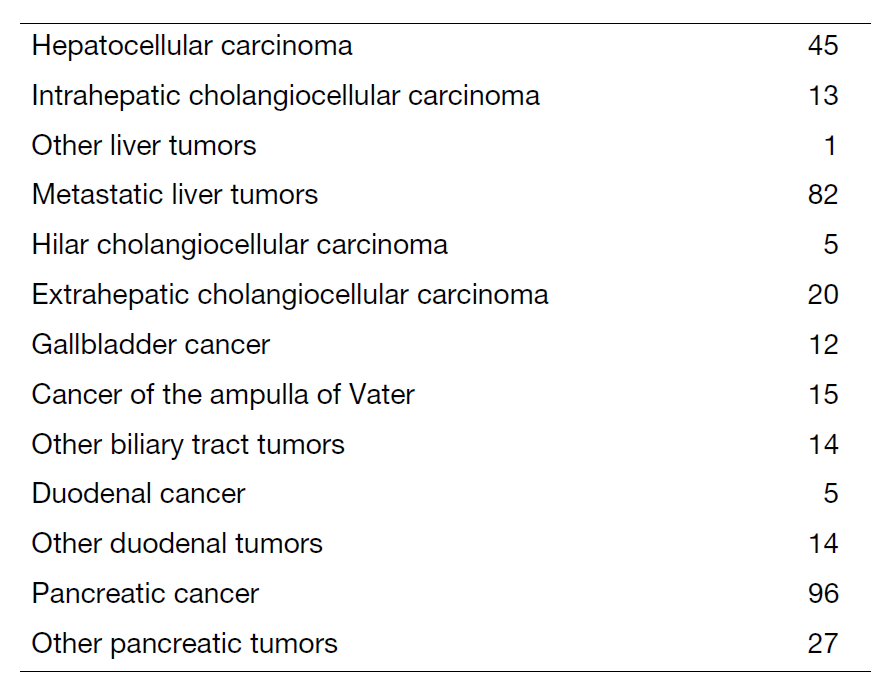
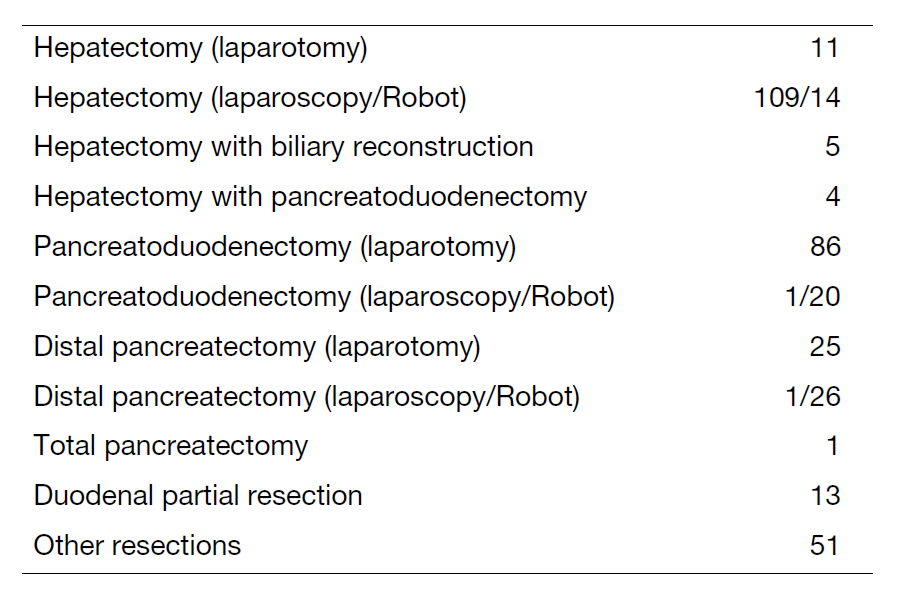
We work with outpatients five days a week and have approximately 25 inpatients. Staff meetings in which we discuss the treatment strategy or the key points of surgery for patients are held with all members of the department every morning. A Cancer Board is held in cooperation with radiologists and medical oncologists every Tuesday. A pathology conference is held monthly with pathologists. In 2023, 398 patients with hepatobiliary and pancreatic diseases underwent surgical treatment. The main diseases are listed in Table 1. We performed a lot of minimally invasive surgeries for liver and pancreatic tumors (Table 2). In 2023, we also introduced robot-assisted pancreaticoduodenectomy.
Research Activities
1. Conversion surgery for pancreatic cancer
Currently, the treatment outcomes of pancreatic cancer patients are improving remarkably with the chemotherapy regimen Gem+nab-PTX or FOLFIRINOX. We attempt conversion surgery for selected patients with borderline resectable or unresectable pancreatic cancer who received chemotherapy. We aim to determine the most appropriate duration of chemotherapy and identify good indications for conversion surgery.
2. Function-preserving surgery
Pancreas-sparing duodenectomy (PSD) represents an alternative procedure to pancreaticoduodenectomy (PD) for patients with duodenal neoplasms. PSD is a function-preserving surgery technique and has the advantage over PD of preservation of the pancreas. We are attempting to establish a safe procedure for PSD.
3. Evaluation of liver function
Postoperative liver failure is one of the fatal complications after major hepatectomy. In our practice, we assess liver function using the indocyanine green retention rate at 15 min (ICG15) test. We are developing an alternative method to evaluate liver function employing liver-specific magnetic resonance imaging (MRI) with a gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid (Gd-EOB-DTPA) contrast agent instead of using the ICG15 test.
4. Cancer genome screening
We conducted clinical studies for cancer genome screening. For patients with resectable HCC, analysis by blood screening (COSMOS-HCC-01) was started in 2020. For patients with resectable biliary tract cancer, analysis by blood screening (COSMOS-BTC-01) was started in 2023.
“A multicenter proof of concept study for personalized perioperative therapy based on genetic alteration status for resectable oligometastases from colorectal cancer” (PRECISION study) (UMIN000042490): This is a multicenter prospective observational study to investigate the clinical utility of pre-treatment ctDNA analysis in patients undergoing surgery for colorectal oligometastases, conducted with a grant from AMED (20ck0106629h0001). The main eligibility criterion is patients with previously untreated resectable colorectal oligometastases, and the primary endpoint is the detection rate of pre-treatment ctDNA and the positive rate of BRAF V600E in tissue. The period of accrual and follow-up is two years each, and the study started in March 2021.
Clinical Trials
1. A non-randomized controlled study comparing proton beam therapy and hepatectomy for resectable hepatocellular carcinoma (JCOG1315C). Recruitment started in 2016.
2. A randomized phase III trial of neoadjuvant gemcitabine and cisplatin and S-1 (GCS) versus surgery first for resectable biliary tract cancer (JCOG1920). Recruitment was started in 2021.
3. A randomized phase III trial of laparoscopic versus open distal pancreatectomy for pancreatic cancer (JCOG2202). Recruitment was started in 2023.
Education
“Board-certified expert surgeons” have a high level of skill in the field of hepatobiliary-pancreatic surgery. To qualify as one, surgeons must perform a specified number of operations under the guidance of a board-certified instructor. The residents in our department are being trained to receive the certification by the end of the chief resident course.
Future Prospects
Our goal is the establishment of multidisciplinary treatment for patients with refractory hepatobiliary and pancreatic cancer and the establishment of minimally invasive surgery for patients with pancreatic cancer and liver cancer. We are conducting robot assisted surgeries in our field.
List of papers published in 2023
Journal
1. Kobayashi S, Nakachi K, Ikeda M, Konishi M, Ogawa G, Sugiura T, Yanagimoto H, Morinaga S, Wada H, Shimada K, Takahashi Y, Nakagohri T, Kamata K, Shimizu Y, Ajiki T, Hirano S, Gotohda N, Ueno M, Okusaka T, Furuse J. Feasibility of S-1 adjuvant chemotherapy after major hepatectomy for biliary tract cancers: An exploratory subset analysis of JCOG1202. European journal of surgical oncology, 50:107324, 2024
2. Ikenaga N, Hashimoto T, Mizusawa J, Kitabayashi R, Sano Y, Fukuda H, Nakata K, Shibuya K, Kitahata Y, Takada M, Kamei K, Kurahara H, Ban D, Kobayashi S, Nagano H, Imamura H, Unno M, Takahashi A, Yagi S, Wada H, Shirakawa H, Yamamoto N, Hirono S, Gotohda N, Hatano E, Nakamura M, Ueno M. A multi-institutional randomized phase III study comparing minimally invasive distal pancreatectomy versus open distal pancreatectomy for pancreatic cancer; Japan Clinical Oncology Group study JCOG2202 (LAPAN study). BMC cancer, 24:231, 2024
3. Une N, Kobayashi S, Kitaguchi D, Sunakawa T, Sasaki K, Ogane T, Hayashi K, Kosugi N, Kudo M, Sugimoto M, Hasegawa H, Takeshita N, Gotohda N, Ito M. Intraoperative artificial intelligence system identifying liver vessels in laparoscopic liver resection: a retrospective experimental study. Surgical endoscopy, 38:1088-1095, 2024
4. Wu X, Iwatsuki M, Takaki M, Saito T, Hayashi T, Kondo M, Sakai Y, Gotohda N, Tanaka E, Nishida T, Baba H. FBXW7 regulates the sensitivity of imatinib in gastrointestinal stromal tumors by targeting MCL1. Gastric cancer, 27:235-247, 2024
5. Kagoura M, Kobayashi S, Kojima M, Kudo M, Sugimoto M, Konishi M, Gotohda N. Survival outcome of patient with pT1N0 biliary tract cancer treated with surgery alone. European journal of surgical oncology, 50:107980, 2024
6. Kobayashi S, Bando H, Taketomi A, Takamoto T, Shinozaki E, Shiozawa M, Hara H, Yamazaki K, Komori K, Matsuhashi N, Kato T, Kagawa Y, Yokota M, Oki E, Komine K, Takahashi S, Wakabayashi M, Yoshino T. NEXUS trial: a multicenter phase II clinical study evaluating the efficacy and safety of the perioperative use of encorafenib, binimetinib, and cetuximab in patients with previously untreated surgically resectable BRAF V600E mutant colorectal oligometastases. BMC cancer, 23:779, 2023
7. Nakachi K, Gotohda N, Hatano E, Nara S, Takahashi S, Kawamoto Y, Ueno M. Adjuvant and neoadjuvant chemotherapy for biliary tract cancer: a review of randomized controlled trials. Japanese journal of clinical oncology, 53:1019-1026, 2023
8. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. ASO Author Reflections: Characteristics of Surgery for Biliary Tract Cancer that Should be Considered During the Design of Future Clinical Trials. Annals of surgical oncology, 30:7384-7385, 2023
9. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. A Multicenter Survey on Eligibility for a Randomized Phase III Trial of Adjuvant Chemotherapy for Resected Biliary Tract Cancer (JCOG1202, ASCOT). Annals of surgical oncology, 30:7331-7337, 2023
10. Sasaki S, Kitaguchi D, Takenaka S, Nakajima K, Sasaki K, Ogane T, Takeshita N, Gotohda N, Ito M. Machine learning-based Automatic Evaluation of Tissue Handling Skills in Laparoscopic Colorectal Surgery: A Retrospective Experimental Study. Annals of surgery, 278:e250-e255, 2023
11. Sakamoto K, Beppu T, Honda G, Kotake K, Yamamoto M, Takahashi K, Endo I, Hasegawa K, Itabashi M, Hashiguchi Y, Kotera Y, Kobayashi S, Yamaguchi T, Natsume S, Tabuchi K, Kobayashi H, Yamaguchi K, Tani K, Morita S, Miyazaki M, Sugihara K. Comprehensive data of 4502 patients newly diagnosed with colorectal liver metastasis between 2015 and 2017, and prognostic data of 2427 patients newly diagnosed with colorectal liver metastasis in 2013 and 2014: Third report of a nationwide survey in Japan. Journal of hepato-biliary-pancreatic sciences, 30:570-590, 2023
12. Beppu T, Yamamura K, Sakamoto K, Honda G, Kobayashi S, Endo I, Hasegawa K, Kotake K, Itabashi M, Hashiguchi Y, Kotera Y, Yamaguchi T, Natsume S, Tabuchi K, Kobayashi H, Yamaguchi K, Morita S, Kikuchi K, Miyazaki M, Sugihara K, Yamamoto M, Takahashi K. Validation study of the JSHBPS nomogram for patients with colorectal liver metastases who underwent hepatic resection in the recent era - a nationwide survey in Japan. Journal of hepato-biliary-pancreatic sciences, 30:591-601, 2023
13. Yajima S, Nakanishi Y, Sugimoto M, Kobayashi S, Kudo M, Yasujima R, Hirose K, Sekiya K, Umino Y, Okubo N, Kataoka M, Gotohda N, Masuda H. A novel predictive model for postoperative delirium using multiple geriatric screening factors. Journal of surgical oncology, 127:1071-1078, 2023
14. Igata Y, Kojima M, Suzuki T, Ishii G, Morisue R, Suzuki T, Kudo M, Sugimoto M, Kobayashi S, Martin JD, Stylianopoulos T, Cabral H, Kano MR, Konishi M, Gotohda N. Relationships between physical and immunological tumor microenvironment in pancreatic ductal adenocarcinoma. Cancer science, 114:3783-3792, 2023
15. Kudo M, Gotohda N, Sugimoto M, Kobayashi S, Konishi M, Kobayashi T. Liver functional assessment using time-associated change in the liver-to-spleen signal intensity ratio on enhanced magnetic resonance imaging: a retrospective study. BMC surgery, 23:179, 2023
16. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. Correction: A Multicenter Survey on Eligibility for a Randomized Phase III Trial of Adjuvant Chemotherapy for Resected Biliary Tract Cancer (JCOG1202, ASCOT). Annals of surgical oncology, 30:6651, 2023
17. Morisue R, Kojima M, Suzuki T, Watanabe R, Sakamoto N, Sakashita S, Harada K, Nakai T, Ishii G, Nakatsura T, Gotohda N, Ishikawa S. Common clinicopathological and immunological features of sarcomatoid carcinoma across organs: A histomorphology-based cross-organ study. International journal of cancer, 153:1997-2010, 2023
18. Gotohda N, Nishida T, Sato S, Ozaka M, Nakahara Y, Komatsu Y, Kondo M, Cho H, Kurokawa Y, Kitagawa Y. Re-appraisal of the universal definition of tumor rupture among patients with high-risk gastrointestinal stromal tumors. Annals of gastroenterological surgery, 7:1021-1031, 2023
