Annual Report 2023
Department of Hepatobiliary and Pancreatic Oncology
Masafumi Ikeda, Shuichi Mitsunaga, Hiroshi Imaoka, Mitsuhito Sasaki, Kazuo Watanabe, Tomoyuki Satake, Taro Shibuki, Kanae Inoue, Tomonao Taira, Shota Yamaguchi, Koh Fukushi
Introduction
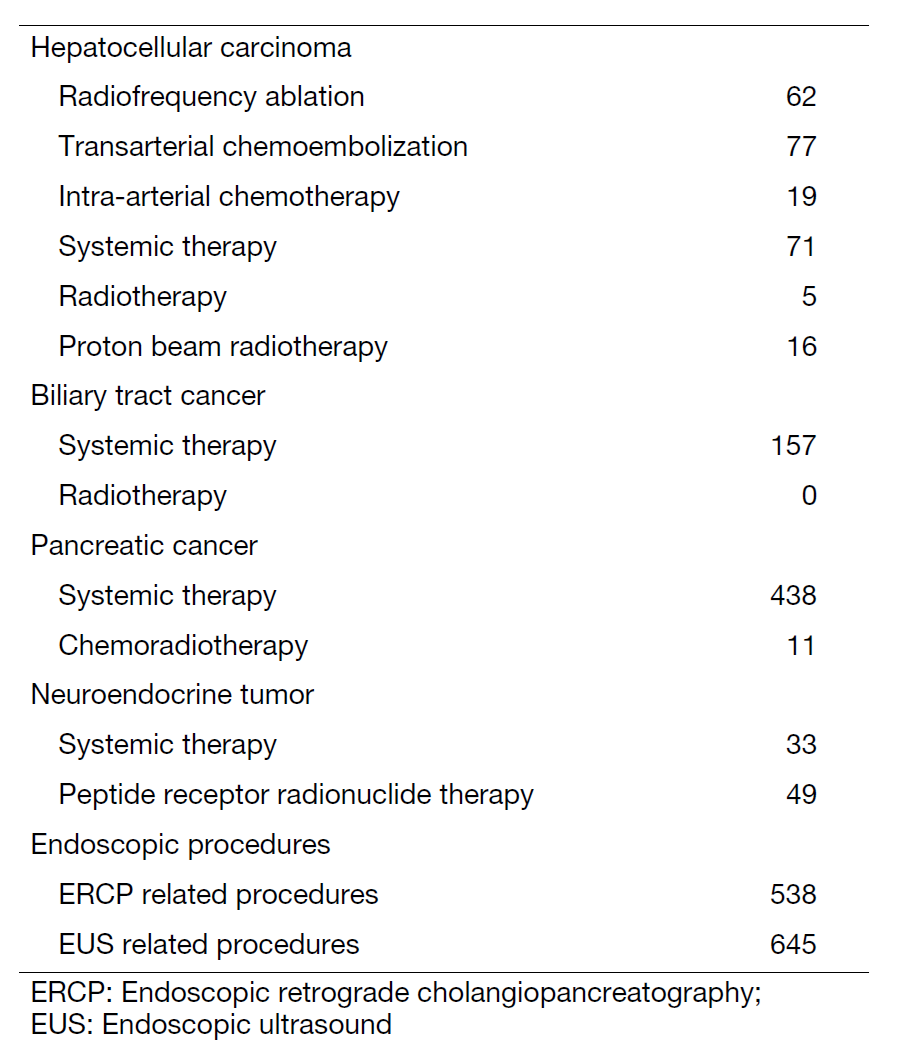
The Department of Hepatobiliary and Pancreatic Oncology is responsible for the diagnosis and treatment of patients with hepatic, biliary, pancreatic cancers and neuroendocrine neoplasms as well as interventional management by endoscopic or percutaneous procedures (Table 1). Our goal is to provide high-quality cancer treatment with adequate palliative care, and to develop novel and effective treatments and procedures through well-designed clinical trials and research.
The Team and What We Do
Our department consists of 7 staff oncologists and 4 residents, with an average of 38 beds in the hospital. These doctors are grouped into two teams and each team determines daily treatment plans for each admitted patient, and all doctors discuss the overall treatment strategy for all admitted patients in our department once a week. The major treatment strategies for individual patients are discussed in weekly tumor board conferences attended by medical oncologists, surgeons, radiologists, radiation oncologists, and pharmacists. We are also responsible for endoscopic abdominal ultrasonographic examinations, endoscopic or percutaneous ultrasound-guided biopsies of abdominal masses, local ablative therapy for liver tumors, endoscopic or percutaneous biliary and abscess drainage, and obstructive jaundice stenting, etc (Table 2).

Research Activities
Hepatocellular carcinoma (HCC)
Systemic therapy is one of the most important treatment modalities for advanced hepatocellular carcinoma (HCC). Combined immunotherapies are firstly selected when patients have no contraindications to immunotherapy, such as autoimmune diseases. Molecular-targeted therapy is selected when immunotherapy is not indicated or proves ineffective. It is also necessary to select the appropriate treatment taking into account the patient’s clinical condition, expected treatment efficacy, and adverse effects of the treatment. To clarify the real-world data in clinical practice in Japan, a prospective observational study of systemic therapy for HCC in Japan: PRISM study is ongoing. At present, over 1500 patients have been enrolled in this study. It is becoming clear how it will be used in daily practice. The results of the multicenter phase II trial of lenvatinib plus hepatic intra-arterial infusion chemotherapy (HAIC) with cisplatin for advanced HCC (LEOPARD) have been published.
Biliary tract cancer (BTC)
For unresectable BTC, combination therapy of gemcitabine plus cisplatin (GC) and anti-PD-L1/PD-1 antibody has been established as the standard treatment and is widely used in clinical practice. Several unique genomic alterations have been identified for unresectable BTC, and precision medicine is actively being applied. We are conducting some research on the treatment outcomes of GC with durvalumab, the prognostic factors in patients receiving systemic therapy, and the usefulness of HER2-targeted therapy for HER2-amplified BTC. We are also investigating biliary stenting methods for the treatment of obstructive jaundice.
Pancreatic cancer (PC)
For unresectable PC, gemcitabine plus nab-paclitaxel (GnP) and FOLFIRINOX for first-line chemotherapy, nano-liposomal irinotecan plus 5-fluorouracil/leucovorin (nalIRI+5-FULV) for second-line chemotherapy have been approved in Japan. We are conducting some research on the treatment outcomes of GnP for PC with malignant ascites, rechallenge of tocilizumab plus GnP for GnP refractory PC, and treatment outcomes for a rare fraction of undifferentiated carcinoma. Furthermore, we investigated the effectiveness of gastrointestinal stents and radiotherapy for tumor bleeding due to PC invasion into the gastrointestinal tract.
Others
We conducted the multicenter validation study of a treatment selection MAP for pancreatic neuroendocrine tumors, atezolizumab plus bevacizumab for combined hepatocellular-cholangiocarcinoma, and pathogenic germline variants in BRCA1/2 and p53 in Asian patients.
Clinical Trials
One hundred and six clinical trials (sponsored: 48 trials, investigator-initiated: 58 trials) are ongoing, and 10 clinical trials (sponsored: 3 trials, investigator-initiated: 7 trials) are being planned for the upcoming year.
HCC
The developments of new regimen are actively ongoing by triplet and quatro regimens as well as doublet regimens, such as combination therapy of VEGFR-targeted multi-kinase inhibitors or antibodies, and immune checkpoint inhibitors. Some clinical trials of new agents of anti-LAG-3 antibody (relatlimab), TGF-beta inhibitor (livmoniolimab), anti TIGIT antibody (tiragolumab), anti-Glypican3/CD3 bispecific antibody (ERY974), coformulated pembrolizumab/quavonlimab (MK-1308A), beta-catenin modulator (E7386), anti-DLK-1 antibody (CBA-1205), and some new combination therapy for durvalumab plus tremelimumab plus lenvatinib as combined therapy with transarterial chemoembolization for intermediate -stage HCC, and lenvatinib plus HAIC with cisplatin for borderline resectable HCC are underway. In addition, a prospective observational study, PRISM, was conducted to collect real-world data in Japan and establish real-world evidence.
BTC
Several trials are being conducted to add other immune checkpoint inhibitors and radiotherapy to GC plus durvalumab. Some clinical trials of novel molecular targeted drugs for specific genomic alterations are also underway: Niraparib for BRCA mutation, E7090 or futibatinib for FGFR2 fusion, LY3410738 for IDH1 mutation, DS-8201a for HER2 positive, and BI 907828 for MDM2 amplified TP53 wild-type. For resected BTC, a phase III trial of rilvegostomig plus chemotherapy vs. placebo plus chemotherapy as adjuvant therapy is ongoing.
PC
Several sponsored trials are underway to develop novel agents to be added to the standard of care, GnP or mFOLFIRINOX: a monoclonal antibody targeting TGF-β (NIS793), zolbetuximab for CLDN18.2 positive, prostaglandin receptor (EP4) antagonist (ONO-4578), a monoclonal antibody against CD47 (ONO-7913), AXL inhibitor (ONO-7475), etc. And binimetinib for BRAF fusion, ponsegromab for elevated GDF-15 levels are underway. Some investigator-initiated trials were ongoing, including a phase II/III trial of neoadjuvant S-1 and concurrent radiotherapy vs. GnP for borderline resectable PC (GABARNANCE), a phase III trial of concurrent S-1 and radiotherapy combined with nivolumab vs. S-1 and radiotherapy alone for locally advanced or borderline resectable PC (JCOG 1908E), and a phase I/II trial of nanoliposomal irinotecan plus S-1.
Neuroendocrine tumor
A phase III trial of everolimus plus lanreotide vs. everolimus (investigator-initiated trial) for gastroenterol pancreatic neuroendocrine tumor, a phase III trial of carboplatin plus etoposide with/without durvalumab (investigator-initiated trial) and a phase II trial of T-cell engager, BI 764532 for gastroenterol pancreatic neuroendocrine carcinoma are ongoing.
Others
Some investigator-initiated studies using circulating tumor DNA are ongoing for hepatobiliary and pancreatic cancer.
Education
Our trainees are provided with daily training with group discussions on the daily practice of inpatient and outpatient management. They can learn the indications, administration and the management of adverse events from loco-regional treatments to systemic therapy for patients with hepatic, biliary, and pancreatic cancer and the accompanying procedures to undertake diagnosis and interventional management, and provide outpatient care. Trainees conduct some retrospective clinical researches and prospective clinical trials to resolve some clinical questions arising from daily practice. Further, they can make presentations of their research in domestic and overseas meetings and publish papers in English under the instruction of staff physicians.
Future Prospects
Immunotherapy has become the standard treatment for HCC and BTC. The number of patients with HCC in Japan is on the decline and the number of patients with BTC has plateaued. However, the numbers of the annual incidence of these cancers remain higher as compared to the other countries. Cytotoxic agents in PC remain the main treatment, and the efficacy of standard treatments is limited. However, the number of patients with PC is rapidly increasing. Developing new agents is an urgent need in the field of hepatobiliary and pancreatic cancer, and Japanese oncologists are expected to play a crucial role in new agent development. Therefore, we actively participate in drug development from the early phases, lead domestic clinical trials, and plan international clinical trials collaborating with overseas investigators. In addition, it is necessary to develop biomarker research and endoscopic management alongside cancer treatment.
List of papers published in 2023
Journal
1. Okano N, Morizane C, Okusaka T, Sadachi R, Kataoka T, Kobayashi S, Ikeda M, Ozaka M, Mizutani T, Sugimori K, Todaka A, Shimizu S, Mizuno N, Yamamoto T, Sano K, Tobimatsu K, Katanuma A, Gotoh K, Yamaguchi H, Ishii H, Ohba A, Furuse J, Ueno M. Early Tumor Shrinkage and Depth of Response as Predictors of Survival for Advanced Biliary Tract Cancer: An Exploratory Analysis of JCOG1113. The oncologist, 29:e97-e107, 2024
2. Ikeda M, Yamashita T, Ogasawara S, Kudo M, Inaba Y, Morimoto M, Tsuchiya K, Shimizu S, Kojima Y, Hiraoka A, Nouso K, Aikata H, Numata K, Sato T, Okusaka T, Furuse J. Multicenter Phase II Trial of Lenvatinib plus Hepatic Intra-Arterial Infusion Chemotherapy with Cisplatin for Advanced Hepatocellular Carcinoma: LEOPARD. Liver cancer, 13:193-202, 2024
3. Piscaglia F, Ikeda K, Cheng AL, Kudo M, Ikeda M, Breder V, Ryoo BY, Mody K, Ren M, Ramji Z, Sung MW. Association between treatment-emergent hypertension and survival with lenvatinib treatment for patients with hepatocellular carcinoma in the REFLECT study. Cancer, 130:1281-1291, 2024
4. Kudo M, Ueshima K, Saeki I, Ishikawa T, Inaba Y, Morimoto N, Aikata H, Tanabe N, Wada Y, Kondo Y, Tsuda M, Nakao K, Ito T, Hosaka T, Kawamura Y, Kuzuya T, Nojiri S, Ogawa C, Koga H, Hino K, Ikeda M, Moriguchi M, Hisai T, Yoshimura K, Furuse J, Arai Y. A Phase 2, Prospective, Multicenter, Single-Arm Trial of Transarterial Chemoembolization Therapy in Combination Strategy with Lenvatinib in Patients with Unresectable Intermediate-Stage Hepatocellular Carcinoma: TACTICS-L Trial. Liver cancer, 13:99-112, 2024
5. Inoue K, Fukushi K, Yamaguchi S, Taira T, Shibuki T, Satake T, Watanabe K, Sasaki M, Imaoka H, Mitsunaga S, Ikeda M. Treatment outcomes of gemcitabine plus nab-paclitaxel in pancreatic cancer patients with malignant ascites. Pancreatology, 24:616-623, 2024
6. Shibuki T, Fukushi K, Inoue K, Taira T, Satake T, Watanabe K, Sasaki M, Imaoka H, Mitsunaga S, Ikeda M. Covered self-expandable metallic stent placement for tumor bleeding from duodenal invasion in patients with unresectable pancreatic cancer. DEN open, 4:e361, 2024
7. Mizuno N, Ioka T, Ogawa G, Nakamura S, Hiraoka N, Ito Y, Katayama H, Takada R, Kobayashi S, Ikeda M, Miwa H, Okano N, Kuramochi H, Sekimoto M, Okusaka T, Ozaka M, Todaka A, Gotoh K, Tobimatsu K, Yamaguchi H, Nakagohri T, Kajiura S, Sudo K, Okamura K, Shimizu S, Shirakawa H, Kato N, Sano K, Iwai T, Fujimori N, Ueno M, Ishii H, Furuse J. Effect of systemic inflammatory response on induction chemotherapy followed by chemoradiotherapy for locally advanced pancreatic cancer: an exploratory subgroup analysis on systemic inflammatory response in JCOG1106. Japanese journal of clinical oncology, 53:704-713, 2023
8. Imaoka H, Ikeda M, Nomura S, Morizane C, Okusaka T, Ozaka M, Shimizu S, Yamazaki K, Okano N, Sugimori K, Shirakawa H, Mizuno N, Satoi S, Yamaguchi H, Sugimoto R, Gotoh K, Sano K, Asagi A, Nakamura K, Ueno M. Development of a nomogram to predict survival in advanced biliary tract cancer. Scientific reports, 13:21548, 2023
9. Satake T, Kondo S, Tanabe N, Mizuno T, Katsuya Y, Sato J, Koyama T, Yoshida T, Hirata M, Yamamoto N. Pathogenic Germline Variants in BRCA1/2 and p53 Identified by Real-world Comprehensive Cancer Genome Profiling Tests in Asian Patients. Cancer research communications, 3:2302-2311, 2023
10. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. ASO Author Reflections: Characteristics of Surgery for Biliary Tract Cancer that Should be Considered During the Design of Future Clinical Trials. Annals of surgical oncology, 30:7384-7385, 2023
11. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. A Multicenter Survey on Eligibility for a Randomized Phase III Trial of Adjuvant Chemotherapy for Resected Biliary Tract Cancer (JCOG1202, ASCOT). Annals of surgical oncology, 30:7331-7337, 2023
12. Okusaka T, Saiura A, Shimada K, Ikeda M, Ioka T, Kimura T, Hosokawa J, Takita A, Oba MS. Incidence and risk factors for venous thromboembolism in the Cancer-VTE Registry pancreatic cancer subcohort. Journal of gastroenterology, 58:1261-1271, 2023
13. Oh DY, He AR, Qin S, Chen LT, Okusaka T, Vogel A, Kim JW, Suksombooncharoen T, Lee MA, Kitano M, Burris H, Bouattour M, Tanasanvimon S, McNamara MG, Zaucha R, Avallone A, Tan B, Cundom J, Lee CK, Takahashi H, Ikeda M, Chen JS, Wang J, Makowsky M, Rokutanda N, Żotkiewicz M, Kurland JF, Cohen G, Valle JW. Plain language summary of the TOPAZ-1 study: durvalumab and chemotherapy for advanced biliary tract cancer. Future oncology (London, England), 19:2277-2289, 2023
14. Honma Y, Ikeda M, Hijioka S, Matsumoto S, Ito T, Aoki T, Furuse J. Optimal first-line treatment strategies of systemic therapy for unresectable gastrointestinal neuroendocrine tumors based on the opinions of Japanese experts. Investigational new drugs, 41:777-786, 2023
15. Llovet JM, Kudo M, Merle P, Meyer T, Qin S, Ikeda M, Xu R, Edeline J, Ryoo BY, Ren Z, Masi G, Kwiatkowski M, Lim HY, Kim JH, Breder V, Kumada H, Cheng AL, Galle PR, Kaneko S, Wang A, Mody K, Dutcus C, Dubrovsky L, Siegel AB, Finn RS. Lenvatinib plus pembrolizumab versus lenvatinib plus placebo for advanced hepatocellular carcinoma (LEAP-002): a randomised, double-blind, phase 3 trial. The Lancet. Oncology, 24:1399-1410, 2023
16. Naito Y, Mishima S, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of tropomyosin receptor kinase inhibitors in adult and pediatric patients with neurotrophic receptor tyrosine kinase fusion-positive advanced solid tumors. International journal of clinical oncology, 28:827-840, 2023
17. Mishima S, Naito Y, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of immunotherapy in patients with high tumor mutational burden tumors. International journal of clinical oncology, 28:941-955, 2023
18. Mishima S, Naito Y, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of immunotherapy in patients with DNA mismatch repair deficient (dMMR) tumors, third edition. International journal of clinical oncology, 28:1237-1258, 2023
19. Habu T, Kumanishi R, Ogata T, Fujisawa T, Mishima S, Kotani D, Kadowaki S, Nakamura M, Hojo H, Fujiwara H, Kumagai S, Koyama S, Fujita T, Kinoshita T, Nishikawa H, Yano T, Tajika M, Muro K, Mitsunaga S, Kojima T, Bando H. Complete response to definitive chemoradiotherapy in unresectable locally advanced esophageal squamous cell carcinoma. Esophagus, 20:533-540, 2023
20. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. Correction: A Multicenter Survey on Eligibility for a Randomized Phase III Trial of Adjuvant Chemotherapy for Resected Biliary Tract Cancer (JCOG1202, ASCOT). Annals of surgical oncology, 30:6651, 2023
21. Kato N, Kudo M, Tsuchiya K, Hagihara A, Numata K, Aikata H, Inaba Y, Kondo S, Motomura K, Okano N, Ikeda M, Morimoto M, Kuroda S, Kimura A. Cabozantinib in Japanese patients with advanced hepatocellular carcinoma: Final results of a multicenter phase II study. Hepatology research, 53:409-416, 2023
22. Hasegawa K, Takemura N, Yamashita T, Watadani T, Kaibori M, Kubo S, Shimada M, Nagano H, Hatano E, Aikata H, Iijima H, Ueshima K, Ohkawa K, Genda T, Tsuchiya K, Torimura T, Ikeda M, Furuse J, Akahane M, Kobayashi S, Sakurai H, Takeda A, Murakami T, Motosugi U, Matsuyama Y, Kudo M, Tateishi R. Clinical Practice Guidelines for Hepatocellular Carcinoma: The Japan Society of Hepatology 2021 version (5th JSH-HCC Guidelines). Hepatology research, 53:383-390, 2023
23. Shibuki T, Okumura K, Sekine M, Kobori I, Miyagaki A, Sasaki Y, Takano Y, Hashimoto Y. Covered self-expandable metallic stents versus plastic stents for endoscopic ultrasound-guided hepaticogastrostomy in patients with malignant biliary obstruction. Clinical endoscopy, 56:802-811, 2023
24. Imaoka H, Ikeda M, Umemoto K, Sunakawa Y, Ueno M, Ueno H, Ozaka M, Kuwahara T, Okano N, Kanai M, Hisano T, Suzuki Y, Asagi A, Shioji K, Todaka A, Tsuji K, Ikezawa K, Miki I, Komatsu Y, Akutsu N, Yamashita T, Okuyama H, Furuse J, Nagano H. Comprehensive review of undifferentiated carcinoma of the pancreas: from epidemiology to treatment. Japanese journal of clinical oncology, 53:764-773, 2023
25. Satake T, Shibuki T, Watanabe K, Sasaki M, Imaoka H, Mitsunaga S, Kojima M, Ikeda M. Case Report: Atezolizumab plus bevacizumab for combined hepatocellular-cholangiocarcinoma. Frontiers in oncology, 13:1234113, 2023
26. Mitsunaga S, Ikeda M, Imaoka H, Sasaki M, Watanabe K, Sato A, Aoki K, Ochiai A, Makikawa M, Nishidate M, Yamaguchi K, Terao K, Sawada N, Fujitomo T, Fujii E, Kato A, Tsunoda H. Fibroblast inhibition by tocilizumab enabled gemcitabine/nab-paclitaxel rechallenge for pancreatic cancer. Cancer science, 114:4006-4019, 2023
27. Kudo M, Finn RS, Galle PR, Zhu AX, Ducreux M, Cheng AL, Ikeda M, Tsuchiya K, Aoki KI, Jia J, Lencioni R. IMbrave150: Efficacy and Safety of Atezolizumab plus Bevacizumab versus Sorafenib in Patients with Barcelona Clinic Liver Cancer Stage B Unresectable Hepatocellular Carcinoma: An Exploratory Analysis of the Phase III Study. Liver cancer, 12:238-250, 2023
28. Kobayashi K, Ogasawara S, Maruta S, Okubo T, Itokawa N, Haga Y, Seko Y, Moriguchi M, Watanabe S, Shiko Y, Takatsuka H, Kanzaki H, Koroki K, Inoue M, Nakamura M, Kiyono S, Kanogawa N, Kondo T, Suzuki E, Ooka Y, Nakamoto S, Inaba Y, Ikeda M, Okabe S, Morimoto N, Itoh Y, Nakamura K, Ito K, Azemoto R, Atsukawa M, Itobayashi E, Kato N. A Prospective Study Exploring the Safety and Efficacy of Lenvatinib for Patients with Advanced Hepatocellular Carcinoma and High Tumor Burden: The LAUNCH Study. Clinical cancer research, 29:4760-4769, 2023
29. Shibuki T, Sasaki M, Yamaguchi S, Inoue K, Taira T, Satake T, Watanabe K, Imaoka H, Mitsunaga S, Fujisawa T, Tomizawa K, Oyoshi H, Nakamura M, Hojo H, Ikeda M. Palliative radiotherapy for tumor bleeding in patients with unresectable pancreatic cancer: a single-center retrospective study. Radiation oncology (London, England), 18:178, 2023
30. Kudo M, Tsuchiya K, Shao Y, Finn RS, Galle PR, Ducreux M, Cheng A, Yamashita T, Koga H, Take R, Yamada K, Asakawa T, Nakagawa Y, Ikeda M. Impact of Bevacizumab Being Skipped due to Adverse Events of Special Interest for Bevacizumab in Patients with Unresectable Hepatocellular Carcinoma Treated with Atezolizumab plus Bevacizumab: An Exploratory Analysis of the Phase III IMbrave150 Study. Liver Cancer, 2023
