Annual Report 2023
Department of General Internal Medicine
Yoichi Naito, Yasutoshi Kuboki, Tomofumi Miura, Kensuke Shinmura, Kazuo Watanabe, Tetsuya Sakai, Saori Mishima, Hiromichi Nakajima
Introduction
We provide general management across cancer types, support for medical treatment in each department (management for complications, adverse events, etc.), and management aimed at training oncology specialists. As an educational hospital of the Japanese Society of Internal Medicine, we contribute to the training for internal medicine with the aim of nurturing general medical specialists. Based on our experience with immune-related adverse events (irAEs) associated with an increasing number of indications and use of immune checkpoint inhibitors in a wide range of diseases and our experience with managing cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) associated with chimeric antigen receptor (CAR) T-cell therapy, we work with all the departments, including the Department of Pharmacy and the Department of Nursing, to recognize the occurrence of irAEs, CRS, and ICANS and implement countermeasures. We also assist in irAE-related research.
The Team and What We Do
- Increased number of reported irAEs, CRS, and ICANS (Figure 1)
- Implementation of irAE collaboration seminar
- Revision of a manual for immune checkpoint inhibitors
- Development of manual for drug-induced liver injury and plasma exchange
- Development of manual for acute kidney injury and plasmapheresis
Figure1.Number of irAE/CRS/ICANS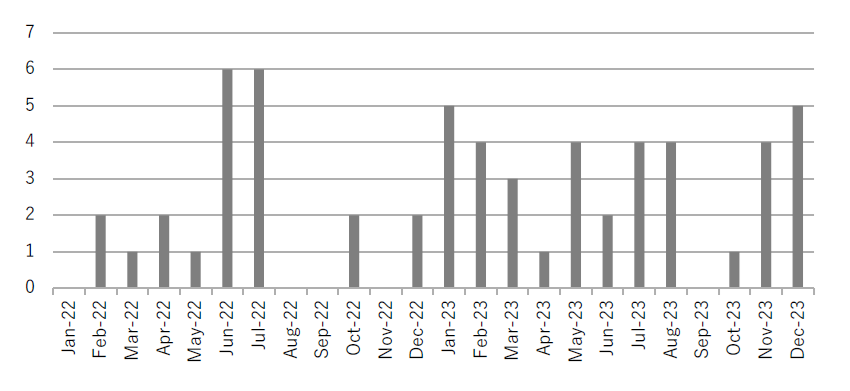

Research Activities
- Contribution to the training of internal medicine trainees
- Case presentation support for trainees at the congress of the Japanese Society of Internal Medicine or related congress
- Paper writing support
- Intensive lectures on medical oncology for new residents (updated)
- Support/guidance for new research (e.g., observational study to confirm the pharmacokinetics of mycophenolate mofetil for liver damage caused by immune checkpoint inhibitors)
- irAE cooperation seminar with regional institutions
- Conducting clinical trials related to supportive care
Clinical Trials
An observational study is currently planned in collaboration with the Department of Dermatology.
Education
We will further collaborate with regional institutions to gather outstanding trainees, and develop close connections with several academic institutions. We will revise a manual for immune checkpoint inhibitors, and develop safe and cost-effective practice. We plan to have approximately 100 case consultations on immune related AEs.
Future Prospects
We will continue to provide high-quality training. The medical treatment for cancer at our hospital is at the top level in Japan, and we will continue to improve the medical treatment system so that the valuable experience gained at this hospital can be returned to general medical treatment, and to establish a consulting system. We will continue the irAE cooperation program and promote research planning for issue identification and improvement. We will deepen our cooperation with the Department of Pharmacy and Department of Nursing, and continue our efforts to share and improve issues within the hospital.
We will further focus on management for adverse events caused by various drugs, in-hospital and out-of-hospital collaboration. We will prepare a manual on renal failure and hemodialysis, train personnel who will be a consultant for renal failure, and establish a cooperative system with collaborating facilities.
Regarding education, we will cooperate with the Human Resource Development Center to enhance internal education and will continuously cooperate with external parties. We will continuously confirm that medical issues can be shared with the risk management department.
Furthermore, we will contribute to the development of supportive care in collaboration with the Supportive and Palliative Care Research Support Office.
List of papers published in 2023
1. Takei S, Tanaka Y, Lin YT, Koyama S, Fukuoka S, Hara H, Nakamura Y, Kuboki Y, Kotani D, Kojima T, Bando H, Mishima S, Ueno T, Kojima S, Wakabayashi M, Sakamoto N, Kojima M, Kuwata T, Yoshino T, Nishikawa H, Mano H, Endo I, Shitara K, Kawazoe A. Multiomic molecular characterization of the response to combination immunotherapy in MSS/pMMR metastatic colorectal cancer. Journal for immunotherapy of cancer, 12:e008210, 2024
2. Sunami K, Naito Y, Saigusa Y, Amano T, Ennishi D, Imai M, Kage H, Kanai M, Kenmotsu H, Komine K, Koyama T, Maeda T, Morita S, Sakai D, Hirata M, Ito M, Kozuki T, Sakashita H, Horinouchi H, Okuma Y, Takashima A, Kubo T, Hironaka S, Segawa Y, Yakushijin Y, Bando H, Makiyama A, Suzuki T, Kinoshita I, Kohsaka S, Ohe Y, Ishioka C, Yamamoto K, Tsuchihara K, Yoshino T. A Learning Program for Treatment Recommendations by Molecular Tumor Boards and Artificial Intelligence. JAMA oncology, 10:95-102, 2024
3. Hotta K, Otake Y, Yamaguchi D, Shimodate Y, Hanabata N, Ikematsu H, Yabuuchi Y, Sano Y, Shimoda R, Sugimoto S, Oba M, Takamaru H, Kimura K, Kishida Y, Takada K, Ito S, Imai K, Hosotani K, Murano T, Yamada M, Shinmura K, Takezawa R, Tomonaga M, Saito Y. Comparison of the efficacy and tolerability of elobixibat plus sodium picosulfate with magnesium citrate and split-dose 2-L polyethylene glycol with ascorbic acid for bowel preparation before outpatient colonoscopy: a study protocol for the multicentre, randomised, controlled E-PLUS trial. BMC gastroenterology, 24:61, 2024
4. Hashimoto T, Nakamura Y, Mishima S, Nakayama I, Kotani D, Kawazoe A, Kuboki Y, Bando H, Kojima T, Iida N, Shibuki T, Imai M, Fujisawa T, Nagamine M, Sakamoto N, Kuwata T, Yoshino T, Shitara K. Whole-transcriptome sequencing in advanced gastric or gastroesophageal cancer: A deep dive into its clinical potential. Cancer science, 115:1622-1633, 2024
5. Kikuchi Y, Shimada H, Hatanaka Y, Kinoshita I, Ikarashi D, Nakatsura T, Kitano S, Naito Y, Tanaka T, Yamashita K, Oshima Y, Nanami T. Clinical practice guidelines for molecular tumor markers, 2nd edition review part 1. International journal of clinical oncology, 29:1-19, 2024
6. Nishida T, Naito Y, Takahashi T, Saito T, Hisamori S, Manaka D, Ogawa K, Hirota S, Ichikawa H. Molecular and clinicopathological features of KIT/PDGFRA wild-type gastrointestinal stromal tumors. Cancer science, 115:894-904, 2024
7. Kitagawa S, Zenke Y, Taki T, Aokage K, Sakai T, Shibata Y, Izumi H, Nosaki K, Umemura S, Matsumoto S, Yoh K, Sakamoto N, Sakashita S, Kojima M, Tsuboi M, Goto K, Ishii G. Prognostic value of predominant subtype in pathological stage II-III lung adenocarcinoma with epidermal growth factor receptor mutation. Lung cancer (Amsterdam, Netherlands), 188:107453, 2024
8. Okunaka M, Kotani D, Fujiwara H, Sato K, Fujiwara N, Mishima S, Sakashita S, Yoshino T, Fujita T, Kojima T. Prognosis of patients with residual pathological disease after neoadjuvant docetaxel, cisplatin, and 5-fluorouracil therapy and surgery for esophageal squamous cell carcinoma: a retrospective cohort study. Therapeutic advances in medical oncology, 16:17588359241229432, 2024
9. Koganemaru S, Fuchigami H, Yamashita H, Morizono C, Sunakawa H, Kawazoe A, Nakamura Y, Kuboki Y, Shitara K, Yano T, Doi T, Yasunaga M. Quantitative Analysis of the Concentration of Trifluridine in Tumor Hypoxic Regions Using a Novel Platform Combining Functional Endoscopy and Mass Spectrometry. Clinical pharmacology and therapeutics, 115:62-70, 2024
10. Sunakawa Y, Kuboki Y, Watanabe J, Terazawa T, Kawakami H, Yokota M, Nakamura M, Kotaka M, Sugimoto N, Ojima H, Oki E, Kajiwara T, Yamamoto Y, Tsuji Y, Denda T, Tamura T, Ishihara S, Taniguchi H, Nakajima TE, Morita S, Shirao K, Takenaka N, Ozawa D, Yoshino T. Exploratory Biomarker Analysis Using Plasma Angiogenesis-Related Factors and Cell-Free DNA in the TRUSTY Study: A Randomized, Phase II/III Study of Trifluridine/Tipiracil Plus Bevacizumab as Second-Line Treatment for Metastatic Colorectal Cancer. Targeted oncology, 19:59-69, 2024
11. Kubota Y, Kawazoe A, Mishima S, Nakamura Y, Kotani D, Kuboki Y, Bando H, Kojima T, Doi T, Yoshino T, Kuwata T, Shitara K. Corrigendum to "Comprehensive clinical and molecular characterization of claudin 18.2 expression in advanced gastric or gastroesophageal junction cancer": [ESMO Open 8 (2023) 100762]. ESMO open, 9:102232, 2024
12. Okunaka M, Kotani D, Mishima S, Nakamura M, Kawazoe A, Bando H, Yoshino T, Shitara K. Potential Efficacy of Shiunko for Anti-Epidermal Growth Factor Receptor (EGFR) Monoclonal Antibody-Induced Skin Fissure: A Single Institutional Case Series. Integrative cancer therapies, 23:15347354231225962, 2024
13. Bando H, Yamaguchi K, Mitani S, Sawada K, Mishima S, Komine K, Okugawa Y, Hosoda W, Ebi H. Japanese Society of Medical Oncology clinical guidelines: Molecular testing for colorectal cancer treatment, 5th edition. Cancer science, 115:1014-1021, 2024
14. Kano Y, Yamamoto Y, Ikematsu H, Sasabe M, Minakata N, Watanabe T, Yamashita H, Mitsui T, Inaba A, Sunakawa H, Nakajo K, Murano T, Kadota T, Shinmura K, Yano T. Investigation of vertical margin involvement in endoscopic resection for T1 colorectal cancer. Digestive endoscopy, 36:455-462, 2024
15. Furue Y, Yoda Y, Hori K, Nakajo K, Kadota T, Murano T, Shinmura K, Ikematsu H, Yano T. Outcomes of repeated endoscopic submucosal dissection for superficial Esophageal squamous cell carcinoma on endoscopic resection scar. Diseases of the esophagus, doae018, 2024
16. Inoue K, Fukushi K, Yamaguchi S, Taira T, Shibuki T, Satake T, Watanabe K, Sasaki M, Imaoka H, Mitsunaga S, Ikeda M. Treatment outcomes of gemcitabine plus nab-paclitaxel in pancreatic cancer patients with malignant ascites. Pancreatology, 24:616-623, 2024
17. Shibuki T, Fukushi K, Inoue K, Taira T, Satake T, Watanabe K, Sasaki M, Imaoka H, Mitsunaga S, Ikeda M. Covered self-expandable metallic stent placement for tumor bleeding from duodenal invasion in patients with unresectable pancreatic cancer. DEN open, 4:e361, 2024
18. Noda S, Yamaguchi J, Kosugi K, Ishida A, Harada M, Ishizuka K, Inoue Y, Kubo E, Miura T. Peppermint Inhalation Therapy for Persistent Hiccups in Terminally Ill Cancer Patient: A Case Study. Journal of pain and symptom management, 67:e503-e505, 2024
19. Kuboki Y, Fakih M, Strickler J, Yaeger R, Masuishi T, Kim EJ, Bestvina CM, Kopetz S, Falchook GS, Langer C, Krauss J, Puri S, Cardona P, Chan E, Varrieur T, Mukundan L, Anderson A, Tran Q, Hong DS. Sotorasib with panitumumab in chemotherapy-refractory KRAS(G12C)-mutated colorectal cancer: a phase 1b trial. Nature medicine, 30:265-270, 2024
20. Minakata N, Murano T, Wakabayashi M, Sasabe M, Watanabe T, Mitsui T, Yamashita H, Inaba A, Sunakawa H, Nakajo K, Kadota T, Shinmura K, Ikematsu H, Yano T. Hot snare polypectomy vs endoscopic mucosal resection using bipolar snare for intermediate size colorectal lesions: Propensity score matching. World journal of gastroenterology, 29:3668-3677, 2023
21. Sasada S, Kondo N, Hashimoto H, Takahashi Y, Terata K, Kida K, Sagara Y, Ueno T, Anan K, Suto A, Kanbayashi C, Takahashi M, Nakamura R, Ishiba T, Tsuneizumi M, Nishimura S, Naito Y, Hara F, Shien T, Iwata H. Prognostic impact of adjuvant endocrine therapy for estrogen receptor-positive and HER2-negative T1a/bN0M0 breast cancer. Breast cancer research and treatment, 202:473-483, 2023
22. Kuboki Y, Terazawa T, Masuishi T, Nakamura M, Watanabe J, Ojima H, Makiyama A, Kotaka M, Hara H, Kagawa Y, Sugimoto N, Kawakami H, Takashima A, Kajiwara T, Oki E, Sunakawa Y, Ishihara S, Taniguchi H, Nakajima TE, Morita S, Shirao K, Takenaka N, Ozawa D, Yoshino T. Trifluridine/tipiracil+bevacizumab (BEV) vs. fluoropyrimidine-irinotecan+BEV as second-line therapy for metastatic colorectal cancer: a randomised noninferiority trial. British journal of cancer, 128:1897-1905, 2023
23. Lee DH, Kim HR, Keam B, Kato K, Kuboki Y, Gao H, Yovine A, Robbins SH, Ahn MJ. Safety and tolerability of first-line durvalumab with tremelimumab and chemotherapy in esophageal squamous cell carcinoma. Cancer medicine, 12:16066-16075, 2023
24. Sakamoto T, Ikematsu H, Tamai N, Mizuguchi Y, Takamaru H, Murano T, Shinmura K, Sasabe M, Furuhashi H, Sumiyama K, Saito Y. Detection of colorectal adenomas with texture and color enhancement imaging: Multicenter observational study. Digestive endoscopy, 35:529-537, 2023
25. Naito Y, Mishima S, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of tropomyosin receptor kinase inhibitors in adult and pediatric patients with neurotrophic receptor tyrosine kinase fusion-positive advanced solid tumors. International journal of clinical oncology, 28:827-840, 2023
26. Mishima S, Naito Y, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of immunotherapy in patients with high tumor mutational burden tumors. International journal of clinical oncology, 28:941-955, 2023
27. Mishima S, Naito Y, Akagi K, Hayashi N, Hirasawa A, Hishiki T, Igarashi A, Ikeda M, Kadowaki S, Kajiyama H, Kato M, Kenmotsu H, Kodera Y, Komine K, Koyama T, Maeda O, Miyachi M, Nishihara H, Nishiyama H, Ohga S, Okamoto W, Oki E, Ono S, Sanada M, Sekine I, Takano T, Tao K, Terashima K, Tsuchihara K, Yatabe Y, Yoshino T, Baba E. Japanese Society of Medical Oncology/Japan Society of Clinical Oncology/Japanese Society of Pediatric Hematology/Oncology-led clinical recommendations on the diagnosis and use of immunotherapy in patients with DNA mismatch repair deficient (dMMR) tumors, third edition. International journal of clinical oncology, 28:1237-1258, 2023
28. Naito Y, Nishida T, Doi T. Current status of and future prospects for the treatment of unresectable or metastatic gastrointestinal stromal tumours. Gastric cancer, 26:339-351, 2023
29. Baba K, Kawamoto M, Mamishin K, Uematsu M, Kiyohara H, Hirota A, Takahashi N, Fukuda M, Kusuhara S, Nakajima H, Funasaka C, Nakao T, Kondoh C, Harano K, Matsubara N, Naito Y, Hosono A, Kawasaki T, Mukohara T. The impact of the COVID-19 pandemic on perioperative chemotherapy for breast cancer. Cancer medicine, 12:12095-12105, 2023
30. Doi T, Matsubara N, Naito Y, Kuboki Y, Harano K, Ono M, Urasaki T, Ohmoto A, Kawanai T, Hisai T, Ikezawa H, Shiba S, Ito K, Semba T, Asano O, Takahashi S. First-in-human study of E7130 (a tumor microenvironment-ameliorating microtubule inhibitor) in patients with advanced solid tumors: Primary results of the dose-escalation part. Cancer, 129:2348-2359, 2023
31. George S, Heinrich MC, Somaiah N, Oppelt P, McLeod R, Nishioka S, Kundu MG, Qian X, Kumar P, Laadem A, Lau Y, Tran BP, Fallon M, Dosunmu O, Shi J, Naito Y. A Phase I, Multicenter, Open-Label, First-in-Human Study of DS-6157a in Patients with Advanced Gastrointestinal Stromal Tumor. Clinical cancer research, 29:3659-3667, 2023
32. Uematsu M, Nakajima H, Hosono A, Kiyohara H, Hirota A, Takahashi N, Fukuda M, Kusuhara S, Nakao T, Funasaka C, Kondoh C, Harano K, Matsubara N, Naito Y, Akimoto T, Mukohara T. Safety of immune checkpoint inhibitors after proton beam therapy in head and neck mucosal melanoma: a case series. Melanoma research, 33:547-552, 2023
33. Terada M, Ito A, Kikawa Y, Koizumi K, Naito Y, Shimoi T, Ishihara M, Yamanaka T, Ozaki Y, Hara F, Nakamura R, Hattori M, Miyashita M, Kondo N, Yoshinami T, Takada M, Matsumoto K, Narui K, Sasada S, Iwamoto T, Hosoda M, Takano Y, Oba T, Sakai H, Murakami A, Higuchi T, Tsuchida J, Tanabe Y, Shigechi T, Tokuda E, Harao M, Kashiwagi S, Mase J, Watanabe J, Nagai SE, Yamauchi C, Yamamoto Y, Iwata H, Saji S, Toyama T. The Japanese Breast Cancer Society Clinical Practice Guidelines for systemic treatment of breast cancer, 2022 edition. Breast cancer (Tokyo, Japan), 30:872-884, 2023
34. Misawa S, Denda T, Kodama S, Suzuki T, Naito Y, Kogawa T, Takada M, Suichi T, Shiosakai K, Kuwabara S. Efficacy and safety of mirogabalin for chemotherapy-induced peripheral neuropathy: a prospective single-arm trial (MiroCIP study). BMC cancer, 23:1098, 2023
35. Sugimoto A, Matsumoto S, Udagawa H, Itotani R, Usui Y, Umemura S, Nishino K, Nakachi I, Kuyama S, Daga H, Hara S, Miyamoto S, Kato T, Sakakibara-Konishi J, Tabata E, Nakagawa T, Kawaguchi T, Sakai T, Shibata Y, Izumi H, Nosaki K, Zenke Y, Yoh K, Goto K. A Large-Scale Prospective Concordance Study of Plasma- and Tissue-Based Next-Generation Targeted Sequencing for Advanced Non-Small Cell Lung Cancer (LC-SCRUM-Liquid). Clinical cancer research , 29:1506-1514, 2023
36. Tanaka Y, Nakai T, Suzuki A, Kagawa Y, Noritake O, Taki T, Hashimoto H, Sakai T, Shibata Y, Izumi H, Nosaki K, Udagawa H, Zenke Y, Matsumoto S, Yoh K, Miyazaki S, Sakamoto N, Sakashita S, Kojima M, Watanbe R, Tsuboi M, Goto K, Ishii G. Clinicopathological significance of peritumoral alveolar macrophages in patients with resected early-stage lung squamous cell carcinoma. Cancer immunology, immunotherapy, 72:2205-2215, 2023
37. Kagawa Y, Nakai T, Taki T, Hashimoto H, Tanaka Y, Sakai T, Shibata Y, Izumi H, Nosaki K, Udagawa H, Zenke Y, Matsumoto S, Yoh K, Miyazaki S, Watanabe R, Kojima M, Sakashita S, Sakamoto N, Tsuboi M, Goto K, Ishii G. Prognostic impact and gene expression analysis of peri-tumoral alveolar macrophage in resected lung adenocarcinoma. Cancer science, 114:3423-3432, 2023
38. Sakai T, Matsumoto S, Ueda Y, Shibata Y, Ikeda T, Nakamura A, Kodani M, Ohashi K, Furuya N, Izumi H, Nosaki K, Umemura S, Zenke Y, Udagawa H, Sugiyama E, Yoh K, Goto K. Clinicogenomic Features and Targetable Mutations in NSCLCs Harboring BRAF Non-V600E Mutations: A Multi-Institutional Genomic Screening Study (LC-SCRUM-Asia). Journal of thoracic oncology, 18:1538-1549, 2023
39. Habu T, Kumanishi R, Ogata T, Fujisawa T, Mishima S, Kotani D, Kadowaki S, Nakamura M, Hojo H, Fujiwara H, Kumagai S, Koyama S, Fujita T, Kinoshita T, Nishikawa H, Yano T, Tajika M, Muro K, Mitsunaga S, Kojima T, Bando H. Complete response to definitive chemoradiotherapy in unresectable locally advanced esophageal squamous cell carcinoma. Esophagus, 20:533-540, 2023
40. Yoshino T, Cervantes A, Bando H, Martinelli E, Oki E, Xu RH, Mulansari NA, Govind Babu K, Lee MA, Tan CK, Cornelio G, Chong DQ, Chen LT, Tanasanvimon S, Prasongsook N, Yeh KH, Chua C, Sacdalan MD, Sow Jenson WJ, Kim ST, Chacko RT, Syaiful RA, Zhang SZ, Curigliano G, Mishima S, Nakamura Y, Ebi H, Sunakawa Y, Takahashi M, Baba E, Peters S, Ishioka C, Pentheroudakis G. Pan-Asian adapted ESMO Clinical Practice Guidelines for the diagnosis, treatment and follow-up of patients with metastatic colorectal cancer. ESMO open, 8:101558, 2023
41. Okunaka M, Kawazoe A, Nakamura H, Kotani D, Mishima S, Kuboki Y, Nakamura Y, Shitara K. Efficacy and safety of trifluridine/tipiracil plus ramucirumab in comparison with trifluridine/tipiracil monotherapy for patients with advanced gastric cancer-single institutional experience. Gastric cancer, 26:1030-1039, 2023
42. Mitsui T, Sunakawa H, Takayama S, Murano T, Shinmura K, Ikematsu H, Yano T. Development of a novel colorectal endoscopic submucosal dissection training model. Endoscopy, 55:E808-E810, 2023
43. Takamatsu T, Endo Y, Fukushima R, Yasue T, Shinmura K, Ikematsu H, Takemura H. Robotic endoscope with double-balloon and double-bend tube for colonoscopy. Scientific reports, 13:10494, 2023
44. Satake T, Shibuki T, Watanabe K, Sasaki M, Imaoka H, Mitsunaga S, Kojima M, Ikeda M. Case Report: Atezolizumab plus bevacizumab for combined hepatocellular-cholangiocarcinoma. Frontiers in oncology, 13:1234113, 2023
45. Mitsunaga S, Ikeda M, Imaoka H, Sasaki M, Watanabe K, Sato A, Aoki K, Ochiai A, Makikawa M, Nishidate M, Yamaguchi K, Terao K, Sawada N, Fujitomo T, Fujii E, Kato A, Tsunoda H. Fibroblast inhibition by tocilizumab enabled gemcitabine/nab-paclitaxel rechallenge for pancreatic cancer. Cancer science, 114:4006-4019, 2023
46. Shibuki T, Sasaki M, Yamaguchi S, Inoue K, Taira T, Satake T, Watanabe K, Imaoka H, Mitsunaga S, Fujisawa T, Tomizawa K, Oyoshi H, Nakamura M, Hojo H, Ikeda M. Palliative radiotherapy for tumor bleeding in patients with unresectable pancreatic cancer: a single-center retrospective study. Radiation oncology (London, England), 18:178, 2023
47. Kubo E, Harada M, Ishizuka K, Umetsu K, Inoue Y, Kosugi K, Miura T. Letter to the Editor: Three Cases of Pain Treatment in Cancer Patients Using an Alternating Magnetic Field Therapy Device. Journal of palliative medicine, 26:1444-1445, 2023
48. Fujiwara Y, Kuboki Y, Furukawa M, Mizuno N, Hara H, Ioka T, Ueno M, Takahashi Y, Takahashi S, Takeuchi S, Lihou C, Ji T, Tian C, Shimizu T. FIGHT-102: A phase 1 study of pemigatinib in Japanese patients with advanced malignancies. Cancer medicine, 12:10597-10611, 2023
49. Fakih MG, Salvatore L, Esaki T, Modest DP, Lopez-Bravo DP, Taieb J, Karamouzis MV, Ruiz-Garcia E, Kim TW, Kuboki Y, Meriggi F, Cunningham D, Yeh KH, Chan E, Chao J, Saportas Y, Tran Q, Cremolini C, Pietrantonio F. Sotorasib plus Panitumumab in Refractory Colorectal Cancer with Mutated KRAS G12C. The New England journal of medicine, 389:2125-2139, 2023
50. Koganemaru S, Kawai T, Fuchigami H, Maeda N, Koyama K, Kuboki Y, Mukohara T, Doi T, Yasunaga M. Quantitative analysis of drug distribution in heterogeneous tissues using dual-stacking capillary electrophoresis-mass spectrometry. British journal of pharmacology, 180:762-774, 2023
51. Mishima S, Nakamura Y, Tukachinsky H, Taniguchi H, Kadowaki S, Kato K, Oki E, Satoh T, Aoki D, Yamazaki K, Esaki T, Ueno M, Nishina T, Sunakawa Y, Denda T, Bando H, Kuramoto N, Horasawa S, Abutani H, Lee JK, Madison RW, Oxnard GR, Yoshino T. Validity and utility of blood tumor mutational burden (bTMB) is dependent on circulating tumor DNA (ctDNA) shed: SCRUM-Japan MONSTAR-SCREEN. The Journal of Liquid Biopsy, 11:2023
