Annual Report 2023
Department of Medical Oncology
Kan Yonemori, Tatsunori Shimoi, Kazuki Sudo, Tadaaki Nishikawa, Yuki Kojima, Aiko Maejima, Shosuke Kita, Munehiro Ito, Ayumi Saito, Mai Hoshino, Eijiro Nakamura, Hitomi Sumiyoshi-Okuma, Asuka Kawachi,Rui Kitadai, Taro Yamanaka, Yasuhiro Fujiwara, Emi Noguchi
Introduction
The Department of Medical Oncology provides the most effective treatments using chemotherapy and works on the establishment of new standards of care for adult malignancies including breast cancer, gynecologic cancer, urological cancer, soft-tissue sarcoma, extra-gonadal germ cell tumor, cancer of unknown primary site and other rare types of solid tumors.
We envision becoming a leading medical oncology department, which makes a difference in cancer care in Japan and worldwide. Our mission is to provide patient-centered, state-of-the-art medical care to cancer patients, to develop new effective cancer treatments through clinical and translational research, and to nurture medical oncologists. An evidence-based, research-oriented and multidisciplinary approach is the core value of our practice.
The Team and What We Do
The Department of Medical Oncology conducts clinical, research and educational activities with a focus on pharmacotherapy for breast, uterine, ovarian, urological cancers, malignant soft tissue tumors (e.g. sarcomas), germ cell tumors, cancers of unknown primary, and other rare malignancies (Table 1).
The five basic missions are as follows:
- Contribute to the development of new cancer therapies domestically and globally by planning, supervising or participating in clinical trials.
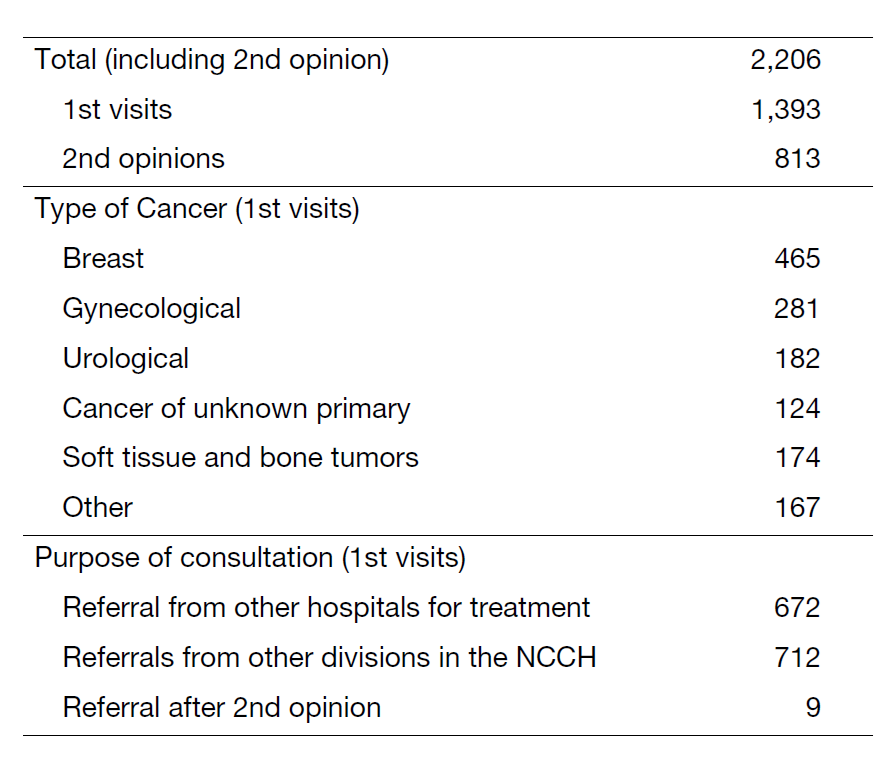
- Promote translational research and strengthen cooperation with other departments, professions and research centers.
- Lead the world in the development of treatments for breast cancers, gynecological cancers, urological cancers and sarcoma, and in the early clinical development of anti-cancer drugs.
- Educate and train specialists in cancer pharmacotherapy or breast cancer treatment (medical specialists).
- Understand the physical, mental and social suffering of cancer patients and aim for holistic medical care.
In addition to providing the latest evidence-based pharmacotherapy, the department focuses on research activities aimed at building new evidence, focusing on the social environment surrounding cancer care, and developing treatments for rare cancers.
Research Activities
Our research interests extend across a wide range of topics related to treatment and clinical program development. Many of our research programs are secured by public and consignment research grants. We conducted many clinical studies as a primary investigator and participated in additional programs as a co-investigator in research programs secured by competitive public research funds. We published 68 international manuscripts, focusing on early-phase anti-cancer drug development, molecular imaging, drug efficacy studies using patient derived xenografts, translational research, novel chemotherapy against breast and gynecological cancer, novel biomarkers to predict the efficacy and adverse events of anti-cancer drugs and other basic research. We value cancer survivorship as a research theme to develop a comprehensive patient-centered care program.
Clinical Trials
In 2023, we actively enrolled patients in phase I studies (including first in human or global) as well as domestic and international phase II and III studies. Of note we enrolled patients in IIT for rare cancers. We also conducted many types of translational studies (TR) to identify novel biomarkers.
Education
We provide rich educational opportunities to both residents and chief residents through clinical experience as well as research activities. Residents are encouraged to make presentations at local and national conferences. We vigorously support basic, clinical, and translational research conducted by postdoctoral researchers.
Future Prospects
We will continue to establish new standard treatments and propose a near-future model for the clinical management of adult solid tumors, including breast cancer, gynecologic cancer, genitourinary cancer and rare cancer. Moreover, we aim to build a comprehensive program, which includes tumor registry, translational research, clinical trials and patient care in rare adult tumors based on our rich clinical experience. We would also like to improve the efficiency of anti-cancer drug development by coordinating basic and translational research in early-phase clinical trials.
Table 2. Active clinical trials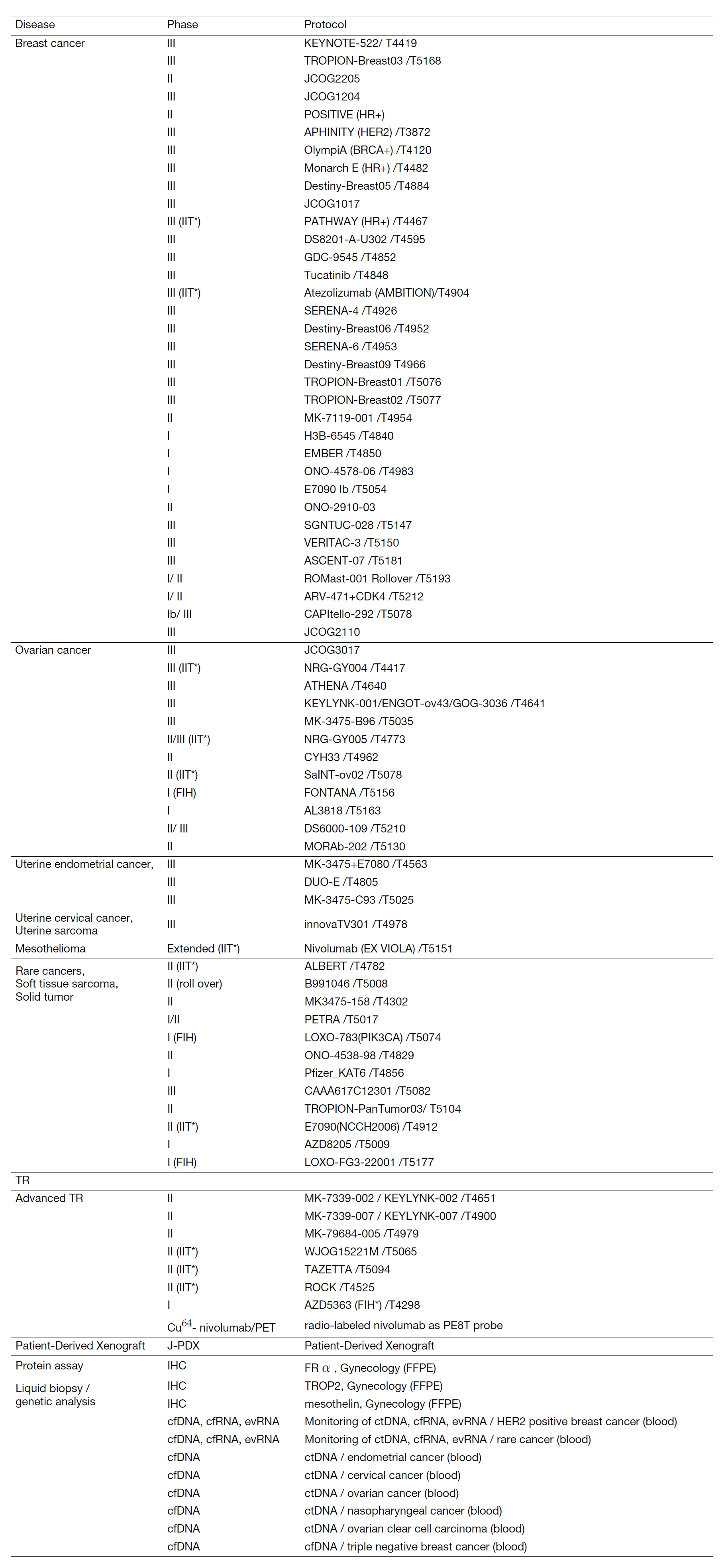

List of papers published in 2023
Journal
1. Ando Y, Shimoi T, Sunami K, Okita N, Nakamura K, Shibata T, Fujiwara Y, Yamamoto N. Progress report of a cross-organ and biomarker-based basket-type clinical trial: BELIEVE Trial. Cancer science, 115:555-563, 2024
2. Shimoi T, Sunami K, Tahara M, Nishiwaki S, Tanaka S, Baba E, Kanai M, Kinoshita I, Shirota H, Hayashi H, Nishida N, Kubo T, Mamesaya N, Ando Y, Okita N, Shibata T, Nakamura K, Yamamoto N. Dabrafenib and trametinib administration in patients with BRAF V600E/R or non-V600 BRAF mutated advanced solid tumours (BELIEVE, NCCH1901): a multicentre, open-label, and single-arm phase II trial. EClinicalMedicine, 69:102447, 2024
3. Watanabe T, Honma Y, Yonemori K, Sunami K, Yoshimoto S, Mori T. High-grade intraductal carcinoma of the parotid gland harboring CTNNA1::ALK rearrangement: Changes in genetic status using genetic testing during treatment with an ALK inhibitor. Head & neck, 46:E26-E31, 2024
4. Yamamoto N, Koyama T, Sato J, Yoshida T, Sudo K, Iwasa S, Kondo S, Yonemori K, Kawasaki A, Satake K, Shibata S, Shimizu T. Phase I study of the anti-TIGIT antibody tiragolumab in combination with atezolizumab in Japanese patients with advanced or metastatic solid tumors. Cancer chemotherapy and pharmacology, 2024
5. Katsuya Y, Yoshida T, Takashima A, Yonemori K, Ohba A, Yazaki S, Yagishita S, Nakahama H, Kobayashi O, Yanagida M, Irino Y, Hamada A, Yamamoto N. Immunogenicity after vaccination of COVID-19 vaccines in patients with cancer: a prospective, single center, observational study. International journal of clinical oncology, 29:386-397, 2024
6. Takamizawa S, Koyama T, Sunami K, Sudo K, Hirata M, Kubo T, Tao K, Cho H, Narita Y, Kato K, Yamazaki N, Ohe Y, Okusaka T, Matsui Y, Ogawa C, Yonemori K, Yamamoto N. Identification of barriers to implementation of precision oncology in patients with rare cancers. Cancer science, 115:2023-2035, 2024
7. Kitadai R, Nishikawa T, Yoshida H, Mizoguchi C, Yamamoto K, Kato T, Yonemori K. Mesothelin expression in gynecologic carcinosarcoma: clinicopathological significance and correlation with HER2 expression. Journal of gynecologic oncology, 35:e11, 2024
8. Odate T, Satomi K, Kubo T, Matsushita Y, Ueno T, Kurose A, Shomori K, Nakai T, Watanabe R, Segawa K, Ohshika S, Miyake N, Kudo S, Shimoi T, Kobayashi E, Komiyama M, Yoshimoto S, Nakatani F, Kawai A, Yatabe Y, Kohsaka S, Ichimura K, Ichikawa H, Yoshida A. Inflammatory Rhabdomyoblastic Tumor: Clinicopathologic and Molecular Analysis of 13 Cases. Modern pathology, 37:100359, 2024
9. Aiba H, Kojima Y, Shimoi T, Sudo K, Yazaki S, Imai T, Yoshida A, Iwata S, Kobayashi E, Kawai A, Arakawa A, Ogawa C, Kimura H, Yonemori K. Incidence of skeletal-related events in patients with Ewing sarcoma: An observational retrospective study in Japan. Cancer medicine, 13:e7060, 2024
10. Ishimaru S, Shimoi T, Sunami K, Nakajima M, Ando Y, Okita N, Nakamura K, Shibata T, Fujiwara Y, Yamamoto N. Platform trial for off-label oncology drugs using comprehensive genomic profiling under the universal public healthcare system: the BELIEVE trial. International journal of clinical oncology, 29:89-95, 2024
11. Hirayama T, Ishiki H, Yanai Y, Horiguchi S, Sugisawa A, Sato J, Kojima R, Sato K, Mizuta T, Kojima R, Udagawa R, Kojima Y, Satomi E. Feasibility of an Electronic Patient-Reported Outcome Tool for Screening Distress and Supportive Care Needs of Adolescents and Young Adults with Cancer. Journal of adolescent and young adult oncology, 13:138-146, 2024
12. Tagami K, Chiu SW, Kosugi K, Ishiki H, Hiratsuka Y, Shimizu M, Mori M, Kubo E, Ikari T, Arakawa S, Eto T, Shimoda M, Hirayama H, Nishijima K, Ouchi K, Shimoi T, Shigeno T, Yamaguchi T, Miyashita M, Morita T, Inoue A, Satomi E. Cancer Pain Management in Patients Receiving Inpatient Specialized Palliative Care Services. Journal of pain and symptom management, 67:27-38.e1, 2024
13. Yonemori K, Fujiwara K, Hasegawa K, Yunokawa M, Ushijima K, Suzuki S, Shikama A, Minobe S, Usami T, Kim JW, Kim BG, Wang PH, Chang TC, Yamamoto K, Han S, McKenzie J, Orlowski RJ, Miura T, Makker V, Man Kim Y. Analysis of East Asia subgroup in Study 309/KEYNOTE-775: lenvatinib plus pembrolizumab versus treatment of physician's choice chemotherapy in patients with previously treated advanced or recurrent endometrial cancer. Journal of gynecologic oncology, 35:e40, 2024
14. Doi T, Takahashi S, Aoki D, Yonemori K, Hara H, Hasegawa K, Takehara K, Harano K, Yunokawa M, Nomura H, Shimoi T, Horie K, Ogasawara A, Okame S. A first-in-human phase I study of TAS-117, an allosteric AKT inhibitor, in patients with advanced solid tumors. Cancer chemotherapy and pharmacology, 93:605-616, 2024
15. Kondo S, Katsuya Y, Yonemori K, Komuro K, Sugeno M, Kawata T, Ghiorghiu D, Meulendijks D, Yamamoto N. Safety, tolerability, pharmacokinetics, and antitumor activity of adavosertib in Japanese patients with advanced solid tumors: A phase I, open-label study . Cancer treatment and research communications, 39:100809 , 2024
16. Westin SN, Moore K, Chon HS, Lee JY, Thomes Pepin J, Sundborg M, Shai A, de la Garza J, Nishio S, Gold MA, Wang K, McIntyre K, Tillmanns TD, Blank SV, Liu JH, McCollum M, Contreras Mejia F, Nishikawa T, Pennington K, Novak Z, De Melo AC, Sehouli J, Klasa-Mazurkiewicz D, Papadimitriou C, Gil-Martin M, Brasiuniene B, Donnelly C, Del Rosario PM, Liu X, Van Nieuwenhuysen E. Durvalumab Plus Carboplatin/Paclitaxel Followed by Maintenance Durvalumab With or Without Olaparib as First-Line Treatment for Advanced Endometrial Cancer: The Phase III DUO-E Trial . Journal of clinical oncology, 42:283-299 , 2024
17. Mizusawa J, Sato H, Rubinstein LV, Fujiwara T, Yonemori K, Hirakawa A. Racial differences in longitudinal toxicities of anticancer agents in early phase cancer clinical trials. Cancer medicine, 12:18098-18109, 2023
18. Okuma HS, Watanabe K, Tsuchihashi K, Machida R, Sadachi R, Hirakawa A, Ariyama H, Kanai M, Kamikura M, Anjo K, Hiramitsu A, Sekine S, Okita N, Mano H, Nishikawa H, Nakamura K, Yonemori K. Phase II Trial of Nivolumab in Metastatic Rare Cancer with dMMR or MSI-H and Relation with Immune Phenotypic Analysis (the ROCK Trial). Clinical cancer research, 29:5079-5086, 2023
19. Eguchi K, Omura G, Shimoi T, Kageyama D, Igaki H, Abe Y, Watanabe T, Aihara Y, Sakai A, Matsumoto Y, Sakai T, Yonemori K, Mori T, Yoshida A, Yoshimoto S. BCOR-CCNB3 sarcoma arising in the pharynx. Auris, nasus, larynx, 50:618-622, 2023
20. Miyakoshi J, Yazaki S, Shimoi T, Onishi M, Saito A, Kita S, Yamamoto K, Kojima Y, Sumiyoshi-Okuma H, Nishikawa T, Sudo K, Noguchi E, Murata T, Shiino S, Takayama S, Suto A, Fujiwara Y, Yoshida M, Yonemori K. Discordance in PD-L1 expression using 22C3 and SP142 assays between primary and metastatic triple-negative breast cancer. Virchows Archiv, 483:855-863, 2023
21. Fujii H, Yamada Y, Yamamura K, Ishida Y, Tsujimura M, Matsumoto K, Tanaka S, Date H, Nishikawa T, Yoshida Y, Kashima J, Yatabe Y, Ogawa S, Marx A, Ulbright TM, Haga H. A case of vasculogenic mesenchymal tumor in the mediastinum: whole-exome sequencing reveals origin from pre-existing germ cell tumor. Virchows Archiv, 482:923-927, 2023
22. Uehara Y, Koyama T, Katsuya Y, Sato J, Sudo K, Kondo S, Yoshida T, Shoji H, Shimoi T, Yonemori K, Yamamoto N. Travel Time and Distance and Participation in Precision Oncology Trials at the National Cancer Center Hospital. JAMA network open, 6:e2333188, 2023
23. Takamori H, Yamasaki T, Kitadai R, Minamishima YA, Nakamura E. Development of drugs targeting hypoxia-inducible factor against tumor cells with VHL mutation: Story of 127 years. Cancer science, 114:1208-1217, 2023
24. Natsume T, Yoshida H, Nishikawa T, Kikkawa N, Naka T, Kobayashi-Kato M, Tanase Y, Uno M, Ishikawa M, Kato T. Uterine Leiomyosarcoma Masquerading as a Malignant Perivascular Epithelioid Cell Tumor: A Diagnostic Challenge. International journal of surgical pathology, 31:778-784, 2023
25. Nishikawa T, Hasegawa K, Matsumoto K, Mori M, Hirashima Y, Takehara K, Ariyoshi K, Kato T, Yagishita S, Hamada A, Kawasaki M, Kawashima S, Tomatsuri S, Nagasaka Y, Yoshida H, Machida R, Hirakawa A, Nakamura K, Yonemori K. Trastuzumab Deruxtecan for Human Epidermal Growth Factor Receptor 2-Expressing Advanced or Recurrent Uterine Carcinosarcoma (NCCH1615): The STATICE Trial. Journal of clinical oncology, 41:2789-2799, 2023
26. Yoshida H, Mizoguchi C, Saito A, Kitadai R, Yamamoto K, Nishikawa T, Kato T, Yonemori K. Discordances in expression of human epidermal growth factor receptor 2 between primary and metastatic uterine carcinosarcoma: A proposal for HER2-targeted therapy specimen selection. Annals of diagnostic pathology, 65:152150, 2023
27. Saito R, Kuroda T, Yoshida H, Sudo K, Saito M, Tanabe H, Takano H, Yamada K, Kiyokawa T, Yonemori K, Kato T, Okamoto A, Kohno T. Genetic characteristics of platinum-sensitive ovarian clear cell carcinoma. Japanese journal of clinical oncology, 53:781-790, 2023
28. Saito A, Nishikawa T, Yoshida H, Mizoguchi C, Kitadai R, Yamamoto K, Yazaki S, Kojima Y, Ishikawa M, Kato T, Yonemori K. Folate receptor alpha is widely expressed and a potential therapeutic target in uterine and ovarian carcinosarcoma. Gynecologic oncology, 176:115-121, 2023
29. Hiranuma K, Asami Y, Kato MK, Murakami N, Shimada Y, Matsuda M, Yazaki S, Fujii E, Sudo K, Kuno I, Komatsu M, Hamamoto R, Makinoshima H, Matsumoto K, Ishikawa M, Kohno T, Terao Y, Itakura A, Yoshida H, Shiraishi K, Kato T. Rare FGFR fusion genes in cervical cancer and transcriptome-based subgrouping of patients with a poor prognosis. Cancer medicine, 12:17835-17848, 2023
30. Yoshida H, Takigawa W, Kobayashi-Kato M, Nishikawa T, Shiraishi K, Ishikawa M. Mismatch Repair Protein Expression in Endometrial Cancer: Assessing Concordance and Unveiling Pitfalls in Two Different Immunohistochemistry Assays. Journal of personalized medicine, 13:1260, 2023
31. Kojima Y, Yoshida H, Okuya T, Okuma HS, Nishikawa T, Tanioka M, Sudo K, Noguchi E, Shimoi T, Tamura K, Tanase Y, Uno M, Ishikawa M, Arakaki M, Ichikawa H, Yagishita S, Hamada A, Fujiwara Y, Yonemori K, Kato T. Therapeutic target biomarkers of patient-derived xenograft models of gastric-type cervical adenocarcinoma. Gynecologic oncology reports, 50:101302, 2023
32. Saito Y, Shimoi T, Iwata S, Maejima A, Abe K, Udagawa R, Yonemori K, Furukawa T, Wakao F. Impact of relative dose intensity of trabectedin with pegfilgrastim support: a single-centre retrospective study. Journal of chemotherapy (Florence, Italy), 35:737-744, 2023
33. Kojima N, Mori T, Motoi T, Kobayashi E, Yoshida M, Yatabe Y, Ichikawa H, Kawai A, Yonemori K, Antonescu CR, Yoshida A. Frequent CD30 Expression in an Emerging Group of Mesenchymal Tumors With NTRK, BRAF, RAF1, or RET Fusions. Modern pathology, 36:100083, 2023
34. Ishihara S, Ogura K, Maejima A, Shimoi T, Sudo K, Kojima Y, Fukushima S, Osaki S, Kobayashi E, Iwata S, Matsui Y, Yonemori K, Kawai A. Predictive value of peripheral blood markers in soft tissue sarcoma patients treated with eribulin. Japanese journal of clinical oncology, 53:494-500, 2023
35. Aiba H, Kojima Y, Shimoi T, Sudo K, Yazaki S, Imai T, Yoshida A, Iwata S, Kobayashi E, Kawai A, Arakawa A, Ogawa C, Kimura H, Yonemori K. Clinical characteristics of primary cutaneous and subcutaneous Ewing sarcoma. Japanese journal of clinical oncology, 53:604-610, 2023
36. Watanabe S, Shimoi T, Nishikawa T, Kawachi A, Okuma HS, Tokura M, Yazaki S, Mizoguchi C, Arakaki M, Saito A, Kita S, Yamamoto K, Kojima Y, Sudo K, Noguchi E, Yoshida A, Kawai A, Fujiwara Y, Yonemori K. Lymphocyte-to-monocyte ratio as a prognostic and potential tumor microenvironment indicator in advanced soft tissue sarcoma treated with first-line doxorubicin therapy. Scientific reports, 13:10734, 2023
37. Tao K, Yamazaki F, Kubo T, Sunami K, Kumamoto T, Arakawa A, Sugiyama M, Watanabe Y, Nakajima M, Shirakawa N, Tanimura K, Koyama T, Hirata M, Sudo K, Tanabe N, Watanabe T, Yoshida T, Kitami M, Yoshida A, Yatabe Y, Nakano Y, Ohira M, Kamijo T, Nakazawa A, Kato M, Ichimura K, Kohno T, Yamamoto N, Hishiki T, Ichikawa H, Ogawa C. Pediatric Precision Medicine at the National Cancer Center Japan: Prospective Genomic Study of Pediatric Patients with Cancer as Part of the TOP-GEAR Project. JCO precision oncology, 7:e2200266, 2023
38. Hiratsuka Y, Tagami K, Inoue A, Sato M, Matsuda Y, Kosugi K, Kubo E, Natsume M, Ishiki H, Arakawa S, Shimizu M, Yokomichi N, Chiu SW, Shimoda M, Hirayama H, Nishijima K, Ouchi K, Shimoi T, Shigeno T, Yamaguchi T, Miyashita M, Morita T, Satomi E. Prevalence of opioid-induced adverse events across opioids commonly used for analgesic treatment in Japan: a multicenter prospective longitudinal study. Supportive care in cancer, 31:632, 2023
39. Shitara K, Hirao M, Iwasa S, Oshima T, Komatsu Y, Kawazoe A, Sato Y, Hamakawa T, Yonemori K, Machida N, Yuki S, Suzuki T, Okumura S, Takase T, Semba T, Zimmermann B, Teng A, Yamaguchi K. Phase I Study of the Liposomal Formulation of Eribulin (E7389-LF): Results from the Advanced Gastric Cancer Expansion Cohort. Clinical cancer research, 29:1460-1467, 2023
40. Okuma HS, Yoshida H, Kobayashi Y, Arakaki M, Mizoguchi C, Inagaki L, Voon PJ, Malik Bin Ismail A, Fen Soo Hoo H, Yusak S, Severino B Imasa M, Nguyen Huy T, Thai Anh T, Kohsaka S, Mano H, Yonemori K, Nakamura K, Yatabe Y. Molecular pathology quality control in Southeast Asia: Results of a multiregional quality assurance study from MASTER KEY Asia. Cancer science, 114:2664-2673, 2023
41. Yagishita S, Nishikawa T, Yoshida H, Shintani D, Sato S, Miwa M, Suzuki M, Yasuda M, Ogitani Y, Jikoh T, Yonemori K, Hasegawa K, Hamada A. Co-Clinical Study of [fam-] Trastuzumab Deruxtecan (DS8201a) in Patient-Derived Xenograft Models of Uterine Carcinosarcoma and Its Association with Clinical Efficacy. Clinical cancer research, 29:2239-2249, 2023
42. Tamura K, Mukohara T, Yonemori K, Kawabata Y, Nicolas X, Tanaka T, Iwata H. Phase 1 study of oral selective estrogen receptor degrader (SERD) amcenestrant (SAR439859), in Japanese women with ER-positive and HER2-negative advanced breast cancer (AMEERA-2). Breast cancer (Tokyo, Japan), 30:506-517, 2023
43. Makker V, Colombo N, Casado Herráez A, Monk BJ, Mackay H, Santin AD, Miller DS, Moore RG, Baron-Hay S, Ray-Coquard I, Ushijima K, Yonemori K, Kim YM, Guerra Alia EM, Sanli UA, Bird S, Orlowski R, McKenzie J, Okpara C, Barresi G, Lorusso D. Lenvatinib Plus Pembrolizumab in Previously Treated Advanced Endometrial Cancer: Updated Efficacy and Safety From the Randomized Phase III Study 309/KEYNOTE-775. Journal of clinical oncology, 41:2904-2910, 2023
44. Subbiah V, Kreitman RJ, Wainberg ZA, Gazzah A, Lassen U, Stein A, Wen PY, Dietrich S, de Jonge MJA, Blay JY, Italiano A, Yonemori K, Cho DC, de Vos FYFL, Moreau P, Fernandez EE, Schellens JHM, Zielinski CC, Redhu S, Boran A, Passos VQ, Ilankumaran P, Bang YJ. Dabrafenib plus trametinib in BRAFV600E-mutated rare cancers: the phase 2 ROAR trial. Nature medicine, 29:1103-1112, 2023
45. Terada M, Nakamura K, Matsuda T, Okuma HS, Sudo K, Yusof A, Imasa M, Sirachainan E, Anh PT, Fujiwara Y, Yamamoto N, Voon PJ, Chokephaibulkit K, Shibata T, Inoue M, Mano H, Shimoi T, Sriuranpong V, Yonemori K, Shimada K. A new era of the Asian clinical research network: a report from the ATLAS international symposium. Japanese journal of clinical oncology, 53:619-628, 2023
46. Koyama T, Shimizu T, Kojima Y, Sudo K, Okuma HS, Shimoi T, Ichikawa H, Kohsaka S, Sadachi R, Hirakawa A, Yoshida A, Ando RM, Ueno T, Yanagaki M, Matsui N, Nakamura K, Yamamoto N, Yonemori K. Clinical Activity and Exploratory Resistance Mechanism of Milademetan, an MDM2 Inhibitor, in Intimal Sarcoma with MDM2 Amplification: An Open-Label Phase Ib/II Study. Cancer discovery, 13:1814-1825, 2023
47. Kojima Y, Noguchi E, Yoshino T, Yagishita S, Yazaki S, Okuma HS, Nishikawa T, Tanioka M, Sudo K, Shimoi T, Kazama A, Terasaki H, Asano S, Fujiwara Y, Hamada A, Tamura K, Yonemori K. Development of a Detection System for ESR1 Mutations in Circulating Tumour DNA Using PNA-LNA-Mediated PCR Clamping. Diagnostics (Basel, Switzerland), 13:2040, 2023
48. Ida H, Shimizu T, Nishino M, Nakamura Y, Yazaki S, Katsuya Y, Sato J, Koyama T, Iwasa S, Sudo K, Kondo S, Yonemori K, Shitara K, Shiono S, Matsuoka D, Yasuda K, Otake Y, Suzuki T, Takase T, Takashima S, Yamaguchi K, Semba T, Yamamoto N. Phase Ib/II Study of a Liposomal Formulation of Eribulin (E7389-LF) plus Nivolumab in Patients with Advanced Solid Tumors: Results from Phase Ib. Cancer research communications, 3:1189-1199, 2023
49. Tashiro R, Kawazoe H, Mamishin K, Seto K, Udagawa R, Saito Y, Hashimoto H, Shimoi T, Yonemori K, Yonemura M, Terakado H, Nishimura T, Kawasaki T, Furukawa T, Nakamura T. Clinical and biomarker factors affecting survival in patients with platinum-sensitive relapsed ovarian cancer receiving olaparib monotherapy: a multicenter retrospective study. Scientific reports, 13:11962, 2023
50. Krop IE, Masuda N, Mukohara T, Takahashi S, Nakayama T, Inoue K, Iwata H, Yamamoto Y, Alvarez RH, Toyama T, Takahashi M, Osaki A, Saji S, Sagara Y, O'Shaughnessy J, Ohwada S, Koyama K, Inoue T, Li L, Patel P, Mostillo J, Tanaka Y, Sternberg DW, Sellami D, Yonemori K. Patritumab Deruxtecan (HER3-DXd), a Human Epidermal Growth Factor Receptor 3-Directed Antibody-Drug Conjugate, in Patients With Previously Treated Human Epidermal Growth Factor Receptor 3-Expressing Metastatic Breast Cancer: A Multicenter, Phase I/II Trial. Journal of clinical oncology, 41:5550-5560, 2023
51. Callens C, Rodrigues M, Briaux A, Frouin E, Eeckhoutte A, Pujade-Lauraine E, Renault V, Stoppa-Lyonnet D, Bieche I, Bataillon G, Karayan-Tapon L, Rochelle T, Heitz F, Cecere SC, Pérez MJR, Grimm C, Nøttrup TJ, Colombo N, Vergote I, Yonemori K, Ray-Coquard I, Stern MH, Popova T. Shallow whole genome sequencing approach to detect Homologous Recombination Deficiency in the PAOLA-1/ENGOT-OV25 phase-III trial. Oncogene, 42:3556-3563, 2023
52. Ando Y, Shimoi T, Suzuki T, Ueno H, Okita N, Nakamura K. Genomic medicine in clinical practice: national genomic medicine program in Japan. Cancer biology & medicine, 21:4-9, 2023
53. Terada M, Ito A, Kikawa Y, Koizumi K, Naito Y, Shimoi T, Ishihara M, Yamanaka T, Ozaki Y, Hara F, Nakamura R, Hattori M, Miyashita M, Kondo N, Yoshinami T, Takada M, Matsumoto K, Narui K, Sasada S, Iwamoto T, Hosoda M, Takano Y, Oba T, Sakai H, Murakami A, Higuchi T, Tsuchida J, Tanabe Y, Shigechi T, Tokuda E, Harao M, Kashiwagi S, Mase J, Watanabe J, Nagai SE, Yamauchi C, Yamamoto Y, Iwata H, Saji S, Toyama T. The Japanese Breast Cancer Society Clinical Practice Guidelines for systemic treatment of breast cancer, 2022 edition. Breast cancer (Tokyo, Japan), 30:872-884, 2023
54. Nakamura Y, Mizuno N, Sunakawa Y, Canon JL, Galsky MD, Hamilton E, Hayashi H, Jerusalem G, Kim ST, Lee KW, Kankeu Fonkoua LA, Monk BJ, Nguyen D, Oh DY, Okines A, O'Malley DM, Pohlmann P, Reck M, Shin SJ, Sudo K, Takahashi S, Van Marcke C, Yu EY, Groisberg R, Ramos J, Tan S, Stinchcombe TE, Bekaii-Saab T. Tucatinib and Trastuzumab for Previously Treated Human Epidermal Growth Factor Receptor 2-Positive Metastatic Biliary Tract Cancer (SGNTUC-019): A Phase II Basket Study. Journal of clinical oncology, 41:5569-5578, 2023
55. Ishizuka Y, Horimoto Y, Morita M, Kawamura Y, Sekine K, Obayashi S, Kojima Y, Tokuda E, Higuchi T, Shimomura A. The Impact of Being Underweight on the Prognosis of Older Patients With Early Breast Cancer . Cancer diagnosis & prognosis, 3:678-686 , 2023
56. Heishima K, Aketa N, Heishima M, Kawachi A. Hemangiosarcoma in dogs as a potential non-rodent animal model for drug discovery research of angiosarcoma in humans . Frontiers in oncology, 13:1250766 , 2023
