Annual Report 2023
Department of Thoracic Oncology
Yuichiro Ohe, Noboru Yamamoto, Hidehito Horinouchi, Yasushi Goto, Tatsuya Yoshida, Yusuke Okuma, Yuki Shinno, Ken Masuda, Yuji Matsumoto, Yukiko Igawa, Akiko Tateishi, Yoh Yamaguchi
Introduction
Lung cancer is the leading cause of cancer death in Japan and worldwide. The incidence of lung cancer in Japan is still increasing, especially in the elderly. The Department of Thoracic Oncology provides care for patients with primary lung cancer, mediastinal tumors, and pleural tumors. The goals of the department are to provide treatment with the highest quality and to establish new effective treatments against lung cancer and other thoracic malignancies through innovative clinical and translational research. To assist our patients through multidisciplinary care, the staff members of the department work closely with thoracic surgeons, radiation oncologists, pathologists, pharmacists, clinical research coordinators, and psychiatrists who have expertise in these areas. The department includes nine staff physicians. Moreover, residents and trainees from other institutions have joined the Thoracic Oncology Program.
The Team and What We Do
The staff physicians attend outpatient services for thoracic diseases, and the department has approximately 50 beds in the hospital. Inpatient care is carried out by five teams. Each team consists of one staff physician and one or two residents and/or trainee doctors. Case conferences are scheduled every Monday and Tuesday morning. Protocol conference and journal club are scheduled every Monday afternoon and Thursday morning, respectively.
A total of 570 new patients started treatment in 2023, and their backgrounds and initial treatments of these patients are shown in Tables 1 and 2. The initial treatments were chemotherapy in 276 cases, adjuvant chemotherapy after surgery in 66, chemoradiotherapy in 84, curative radiotherapy in 9, and supportive care including palliative radiotherapy in 67. The survival rates of lung cancer patients treated in 2014-2018 in our department are shown in Table 3.
Table 1. Number of patients in 2023

Table 2. Type of procedure in 2023

Table 3. Survival rates of lung cancer patients treated in 2014-2018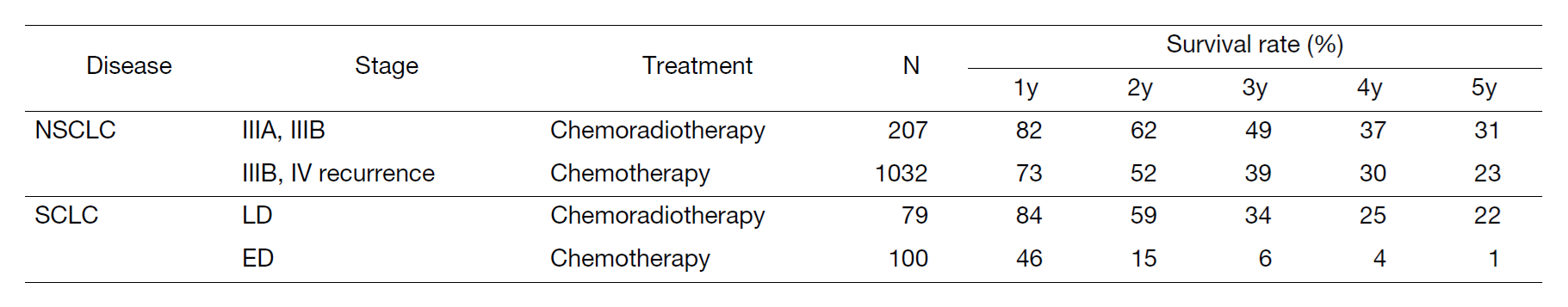

Research Activities
Research activities of the department can be classified into four categories: (1) multi-institutional phase III studies to establish new standard treatments against lung cancer; (2) phase I and II studies to evaluate new anticancer drugs; (3) pharmacokinetic and pharmacodynamic (PK/PD) studies to investigate interpatient variability, optimal administration schedules and drug-drug interactions; and (4) translational research using clinical samples from bench to bed-side or from bed-side to bench for the development of innovative treatment strategies.
Clinical Trials
The department is currently conducting and participating in multi-institutional phase III studies to establish new standard treatments against lung cancer such as the Japan Clinical Oncology Group (JCOG) trials and global trials conducted by pharmaceutical companies. Six JCOG clinical studies, namely JCOG1408 (J-SBRT trial), a phase III study of SBRT for c-stage IA NSCLC, JCOG1701 (SAVE study), a phase III study of immune check point inhibitors to evaluate optimal treatment period, JCOG1807C (DEEP OCEAN), chemoradiotherapy with durvalumab and surgery for superior sulcus NSCLC, JCOG1914, a phase III of chemoradiotherapy for locally advanced NSCLC in the elderly, JCOG2002 (yES-TRT), a phase III study of thoracic radiotherapy for ED-SCLC after chemotherapy with immune checkpoint inhibitor (ICI), JCOG2007 (NIPPON), a phase III study of chemotherapy with ICIs for NSCLC, are ongoing. The department is also participating in a nationwide screening project for lung cancer with rare driver mutation (LC-SCRUM). Moreover, the department has conducted many clinical trials using TKIs, ICIs, antibody-drug conjugates (ADC) and bispecific antibodies.
Education
In 2023, 3 chief residents, 11 residents, and 5 trainee doctors joined the department. A monthly research conference is held to discuss the clinical and translational research conducted by young doctors.
Future Prospects
Recent progression of lung cancer treatment is very rapid. The driver gene alteration targeted therapy, such as EGFR-TKIs for EGFR mutation positive lung cancer, ALK inhibitors for ALK fusion gene-positive lung cancer, ROS1 inhibitors for ROS1 fusion gene-positive lung cancer, BRAF plus MEK inhibitor for BRAF V600E-positive lung cancer, NTRK inhibitors for NTRK fusion-positive lung cancer, RET inhibitor for RET fusion-positive lung cancer, MET inhibitors for MET exon 14 skipping mutation-positive lung cancer and KRAS inhibitor for KRAS G12C lung cancer, HER2 antibody-drug conjugates (ADC) for HER2 mutation-positive lung cancer have already been established as the standard treatment. Other rare driver gene alterations, including NRG and CLIP-LTK fusion will be good targets for the treatment of lung cancer. Immune check point inhibitors, anti-PD-1 Ab and anti-PD-L1 Ab plus chemotherapy have been established as a standard 1st line treatment for NSCLC. A combination of anti-PD-1 Ab and anti-CTLA-4 Ab with or without chemotherapy has also been established as a new standard treatment for advanced NSCLC. Anti-PD-L1 Ab, durvalumab for stage III NSCLC after chemoradiotherapy will also be established as a standard treatment. An immune checkpoint inhibitor also has been an incorporated treatment for early-stage lung cancer with surgery. Moreover, bispecific antibodies will now be treatment options for lung cancer patients.
List of papers published in 2023
Journal
1. Shiraishi K, Takahashi A, Momozawa Y, Daigo Y, Kaneko S, Kawaguchi T, Kunitoh H, Matsumoto S, Horinouchi H, Goto A, Honda T, Shimizu K, Torasawa M, Takayanagi D, Saito M, Saito A, Ohe Y, Watanabe SI, Goto K, Tsuboi M, Tsuchihara K, Takata S, Aoi T, Takano A, Kobayashi M, Miyagi Y, Tanaka K, Suzuki H, Maeda D, Yamaura T, Matsuda M, Shimada Y, Mizuno T, Sakamoto H, Yoshida T, Goto Y, Yoshida T, Yamaji T, Sonobe M, Toyooka S, Yoneda K, Masago K, Tanaka F, Hara M, Fuse N, Nishizuka SS, Motoi N, Sawada N, Nishida Y, Kumada K, Takeuchi K, Tanno K, Yatabe Y, Sunami K, Hishida T, Miyazaki Y, Ito H, Amemiya M, Totsuka H, Nakayama H, Yokose T, Ishigaki K, Nagashima T, Ohtaki Y, Imai K, Takasawa K, Minamiya Y, Kobayashi K, Okubo K, Wakai K, Shimizu A, Yamamoto M, Iwasaki M, Matsuda K, Inazawa J, Shiraishi Y, Nishikawa H, Murakami Y, Kubo M, Matsuda F, Kamatani Y, Hamamoto R, Matsuo K, Kohno T. Identification of telomere maintenance gene variations related to lung adenocarcinoma risk by genome-wide association and whole genome sequencing analyses. Cancer communications (London, England), 44:287-293, 2024
2. Takasawa K, Asada K, Kaneko S, Shiraishi K, Machino H, Takahashi S, Shinkai N, Kouno N, Kobayashi K, Komatsu M, Mizuno T, Okubo Y, Mukai M, Yoshida T, Yoshida Y, Horinouchi H, Watanabe SI, Ohe Y, Yatabe Y, Kohno T, Hamamoto R. Advances in cancer DNA methylation analysis with methPLIER: use of non-negative matrix factorization and knowledge-based constraints to enhance biological interpretability. Experimental & molecular medicine, 56:646-655, 2024
3. Torasawa M, Horinouchi H, Nomura S, Igawa S, Asai M, Ishii H, Wakui H, Ushio R, Asao T, Namba Y, Koyama R, Hayakawa D, Katayama I, Matsuda H, Sasaki S, Takahashi K, Hosomi Y, Naoki K, Ohe Y. Reconsidering the Cutoff Value for Sensitive and Refractory Relapses in Extensive-Stage SCLC in the Era of Immunotherapy. Journal of thoracic oncology, 19:325-336, 2024
4. Shirasawa M, Yoshida T, Ohe Y. Biomarkers of immunotherapy for non-small cell lung cancer. Japanese journal of clinical oncology, 54:13-22, 2024
5. Okuma Y, Kubota K, Shimokawa M, Hashimoto K, Kawashima Y, Sakamoto T, Wakui H, Murakami S, Okishio K, Hayashihara K, Ohe Y. First-Line Osimertinib for Previously Untreated Patients With NSCLC and Uncommon EGFR Mutations: The UNICORN Phase 2 Nonrandomized Clinical Trial. JAMA oncology, 10:43-51, 2024
6. Sunami K, Naito Y, Saigusa Y, Amano T, Ennishi D, Imai M, Kage H, Kanai M, Kenmotsu H, Komine K, Koyama T, Maeda T, Morita S, Sakai D, Hirata M, Ito M, Kozuki T, Sakashita H, Horinouchi H, Okuma Y, Takashima A, Kubo T, Hironaka S, Segawa Y, Yakushijin Y, Bando H, Makiyama A, Suzuki T, Kinoshita I, Kohsaka S, Ohe Y, Ishioka C, Yamamoto K, Tsuchihara K, Yoshino T. A Learning Program for Treatment Recommendations by Molecular Tumor Boards and Artificial Intelligence. JAMA oncology, 10:95-102, 2024
7. Yamamoto N, Koyama T, Sato J, Yoshida T, Sudo K, Iwasa S, Kondo S, Yonemori K, Kawasaki A, Satake K, Shibata S, Shimizu T. Phase I study of the anti-TIGIT antibody tiragolumab in combination with atezolizumab in Japanese patients with advanced or metastatic solid tumors. Cancer chemotherapy and pharmacology, 2024
8. Imai H, Kijima T, Azuma K, Kishi K, Saito H, Yamaguchi T, Tanizaki J, Yoneshima Y, Fujita K, Watanabe S, Kitazono S, Fukuhara T, Hataji O, Toi Y, Mizutani H, Hamakawa Y, Maemondo M, Ohsugi T, Suzuki K, Horinouchi H, Ohe Y. First-line nivolumab plus ipilimumab with or without chemotherapy for Japanese patients with non-small cell lung cancer: LIGHT-NING study. Japanese journal of clinical oncology, 54:452-462, 2024
9. Kage H, Akiyama N, Chang H, Shinozaki-Ushiku A, Ka M, Kawata J, Muto M, Okuma Y, Okita N, Tsuchihara K, Kikuchi J, Shirota H, Hayashi H, Kokuryo T, Yachida S, Hirasawa A, Kubo M, Kenmotsu H, Tanabe M, Ushiku T, Muto K, Seto Y, Oda K. Patient survey on cancer genomic medicine in Japan under the national health insurance system. Cancer science, 115:954-962, 2024
10. Torasawa M, Horinouchi H. A Response to the Letter to the Editor: "A Paradigm Shift in First-Line Treatment Can Have Many Implications for Second-Line Treatment". Journal of thoracic oncology, 19:351-352, 2024
11. Okuma Y, Shintani Y, Sekine I, Shukuya T, Takayama K, Inoue A, Okamoto I, Kiura K, Yamamoto N, Kawaguchi T, Miyaoka E, Yoshino I, Date H. Efficacy of Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors in Metastatic Non-Small Cell Lung Cancer Patients with Poor Performance Status and Epidermal Growth Factor Receptor Mutations: Findings from the Japanese Lung Cancer Registry Database. Clinical lung cancer, 2024
12. Takeyasu Y, Yoshida T, Masuda K, Matsumoto Y, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. Distinct Progression and Efficacy of First-Line Osimertinib Treatment According to Mutation Subtypes in Metastatic NSCLC Harboring EGFR Mutations. JTO clinical and research reports, 5:100636, 2024
13. Imai T, Yoshida T, Ohe Y. Adenovirus pneumonia mimicking osimertinib-induced pneumonitis in a patient with advanced NSCLC with EGFR mutation: A case report. Thoracic cancer, 15:749-751, 2024
14. Katsuya Y, Yoshida T, Takashima A, Yonemori K, Ohba A, Yazaki S, Yagishita S, Nakahama H, Kobayashi O, Yanagida M, Irino Y, Hamada A, Yamamoto N. Immunogenicity after vaccination of COVID-19 vaccines in patients with cancer: a prospective, single center, observational study. International journal of clinical oncology, 29:386-397, 2024
15. Okuma Y, Nomura S, Sakakibara-Konishi J, Tsukita Y, Murakami S, Hosomi Y, Tambo Y, Kogure Y, Yoshioka H, Tamiya M, Ninomiya K, Iwama E. Artemis: A Multicenter, Open-Label, Single-Arm, Phase II Study to Evaluate the Efficacy and Safety of First-Line Carboplatin/Paclitaxel/Lenvatinib/Pembrolizumab Combination for Previously Untreated Advanced or Recurrent Thymic Carcinomas. Clinical lung cancer, 25:389-394, 2024
16. Shimokawa T, Okamoto H, Machida R, Misumi Y, Hosomi Y, Yoneshima Y, Tanaka H, Okishio K, Simizu J, Goto K, Akamatsu H, Kubota K, Nakagawa K, Horinouchi H, Ando M, Kataoka T, Ohe Y. Carboplatin and irinotecan (CI) vs. carboplatin and etoposide (CE) for the treatment of extended-stage small-cell lung cancer in an elderly population: A phase II/III randomized control trial. Lung cancer (Amsterdam, Netherlands), 181:107195, 2023
17. Shirasawa M, Yoshida T, Shiraishi K, Takigami A, Takayanagi D, Imabayashi T, Matsumoto Y, Masuda K, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Yotsukura M, Yoshida Y, Nakagawa K, Tsuchida T, Hamamoto R, Yamamoto N, Motoi N, Kohno T, Watanabe SI, Ohe Y. Identification of inflamed-phenotype of small cell lung cancer leading to the efficacy of anti-PD-L1 antibody and chemotherapy. Lung cancer (Amsterdam, Netherlands), 179:107183, 2023
18. Shi J, Shiraishi K, Choi J, Matsuo K, Chen TY, Dai J, Hung RJ, Chen K, Shu XO, Kim YT, Landi MT, Lin D, Zheng W, Yin Z, Zhou B, Song B, Wang J, Seow WJ, Song L, Chang IS, Hu W, Chien LH, Cai Q, Hong YC, Kim HN, Wu YL, Wong MP, Richardson BD, Funderburk KM, Li S, Zhang T, Breeze C, Wang Z, Blechter B, Bassig BA, Kim JH, Albanes D, Wong JYY, Shin MH, Chung LP, Yang Y, An SJ, Zheng H, Yatabe Y, Zhang XC, Kim YC, Caporaso NE, Chang J, Ho JCM, Kubo M, Daigo Y, Song M, Momozawa Y, Kamatani Y, Kobayashi M, Okubo K, Honda T, Hosgood DH, Kunitoh H, Patel H, Watanabe SI, Miyagi Y, Nakayama H, Matsumoto S, Horinouchi H, Tsuboi M, Hamamoto R, Goto K, Ohe Y, Takahashi A, Goto A, Minamiya Y, Hara M, Nishida Y, Takeuchi K, Wakai K, Matsuda K, Murakami Y, Shimizu K, Suzuki H, Saito M, Ohtaki Y, Tanaka K, Wu T, Wei F, Dai H, Machiela MJ, Su J, Kim YH, Oh IJ, Lee VHF, Chang GC, Tsai YH, Chen KY, Huang MS, Su WC, Chen YM, Seow A, Park JY, Kweon SS, Chen KC, Gao YT, Qian B, Wu C, Lu D, Liu J, Schwartz AG, Houlston R, Spitz MR, Gorlov IP, Wu X, Yang P, Lam S, Tardon A, Chen C, Bojesen SE, Johansson M, Risch A, Bickeböller H, Ji BT, Wichmann HE, Christiani DC, Rennert G, Arnold S, Brennan P, McKay J, Field JK, Shete SS, Le Marchand L, Liu G, Andrew A, Kiemeney LA, Zienolddiny-Narui S, Grankvist K, Johansson M, Cox A, Taylor F, Yuan JM, Lazarus P, Schabath MB, Aldrich MC, Jeon HS, Jiang SS, Sung JS, Chen CH, Hsiao CF, Jung YJ, Guo H, Hu Z, Burdett L, Yeager M, Hutchinson A, Hicks B, Liu J, Zhu B, Berndt SI, Wu W, Wang J, Li Y, Choi JE, Park KH, Sung SW, Liu L, Kang CH, Wang WC, Xu J, Guan P, Tan W, Yu CJ, Yang G, Sihoe ADL, Chen Y, Choi YY, Kim JS, Yoon HI, Park IK, Xu P, He Q, Wang CL, Hung HH, Vermeulen RCH, Cheng I, Wu J, Lim WY, Tsai FY, Chan JKC, Li J, Chen H, Lin HC, Jin L, Liu J, Sawada N, Yamaji T, Wyatt K, Li SA, Ma H, Zhu M, Wang Z, Cheng S, Li X, Ren Y, Chao A, Iwasaki M, Zhu J, Jiang G, Fei K, Wu G, Chen CY, Chen CJ, Yang PC, Yu J, Stevens VL, Fraumeni JF Jr, Chatterjee N, Gorlova OY, Hsiung CA, Amos CI, Shen H, Chanock SJ,. Genome-wide association study of lung adenocarcinoma in East Asia and comparison with a European population. Nature communications, 14:3043, 2023
19. Igarashi T, Mazevet M, Yasuhara T, Yano K, Mochizuki A, Nishino M, Yoshida T, Yoshida Y, Takamatsu N, Yoshimi A, Shiraishi K, Horinouchi H, Kohno T, Hamamoto R, Adachi J, Zou L, Shiotani B. An ATR-PrimPol pathway confers tolerance to oncogenic KRAS-induced and heterochromatin-associated replication stress. Nature communications, 14:4991, 2023
20. Shirasawa M, Yoshida T, Shiraishi K, Goto N, Yagishita S, Imabayashi T, Matsumoto Y, Masuda K, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Yotsukura M, Yoshida Y, Nakagawa K, Naoki K, Tsuchida T, Hamamoto R, Yamamoto N, Motoi N, Kohno T, Watanabe SI, Ohe Y. Tumor microenvironment-mediated immune profiles and efficacy of anti-PD-L1 antibody plus chemotherapy stratified by DLL3 expression in small-cell lung cancer. British journal of cancer, 129:2003-2013, 2023
21. Shimoda Y, Yoshida T, Miyakoshi J, Torasawa M, Tateishi A, Matsumoto Y, Masuda K, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. Incidence of serious adverse events caused by tyrosine kinase inhibitor treatment following immune checkpoint inhibitor therapy in advanced NSCLC patients with oncogenic driver alterations. Cancer immunology, immunotherapy, 72:2613-2621, 2023
22. Torasawa M, Yoshida T, Takeyasu Y, Shimoda Y, Tateishi A, Matsumoto Y, Masuda K, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N, Takahashi K, Ohe Y. Disease progression status during initial immune checkpoint inhibitor (ICI) affects the clinical outcome of ICI retreatment in advanced non-small cell lung cancer patients. Cancer medicine, 12:12388-12401, 2023
23. Horinouchi H. Another Pirate in the Red Ocean? CD73-Targeted Therapy in EGFR-Mutated NSCLC. Journal of thoracic oncology, 18:552-555, 2023
24. Kage H, Oda K, Muto M, Tsuchihara K, Okita N, Okuma Y, Kikuchi J, Shirota H, Hayashi H, Kokuryo T, Sakai D, Hirasawa A, Kubo M, Kenmotsu H, Akiyama N, Shinozaki-Ushiku A, Tanabe M, Ushiku T, Miyagawa K, Seto Y. Human resources for administrative work to carry out a comprehensive genomic profiling test in Japan. Cancer science, 114:3041-3049, 2023
25. Sugawara S, Tanaka K, Imamura F, Yamamoto N, Nishio M, Okishio K, Hirashima T, Tanaka H, Fukuhara T, Nakahara Y, Kurata T, Katakami N, Okada M, Horinouchi H, Udagawa H, Kasahara K, Satouchi M, Saka H, Tokito T, Hosomi Y, Aoe K, Kishi K, Ohashi K, Yokoyama T, Adachi N, Noguchi K, Schwarzenberger P, Kato T. Pembrolizumab plus chemotherapy in Japanese patients with metastatic squamous non-small-cell lung cancer in KEYNOTE-407. Cancer science, 114:3330-3341, 2023
26. Sumiyoshi A, Fujii H, Okuma Y. Targeting microbiome, drug metabolism, and drug delivery in oncology. Advanced drug delivery reviews, 199:114902, 2023
27. Araki T, Kanda S, Horinouchi H, Ohe Y. Current treatment strategies for EGFR-mutated non-small cell lung cancer: from first line to beyond osimertinib resistance. Japanese journal of clinical oncology, 53:547-561, 2023
28. Takata S, Morikawa K, Tanaka H, Itani H, Ishihara M, Horiuchi K, Kato Y, Ikemura S, Nakagawa H, Nakahara Y, Seki Y, Bessho A, Takahashi N, Hayashi K, Endo T, Takeyama K, Maekura T, Takigawa N, Kawase A, Endoh M, Nemoto K, Kishi K, Soejima K, Okuma Y, Yoshimura K, Saigusa D, Kanai Y, Ueda K, Togashi A, Matsutani N, Seki N. Prospective exosome-focused translational research for afatinib (EXTRA) study of patients with nonsmall cell lung cancer harboring EGFR mutation: an observational clinical study. Therapeutic advances in medical oncology, 15:17588359231177021, 2023
29. Nishimatsu K, Matsumoto Y, Kashima J, Imabayashi T, Uchimura K, Furuse H, Masuda K, Shinno Y, Okuma Y, Yoshida T, Goto Y, Horinouchi H, Yamamoto N, Tsuchida T, Ohe Y, Yatabe Y. Concordance between cryobiopsy and forceps biopsy specimens in assessment of immunohistochemistry staining for non-small cell lung carcinoma. Translational lung cancer research, 12:1245-1255, 2023
30. Yoshida T, Kumagai T, Toyozawa R, Katayama R, Nishio M, Seto T, Goto K, Yamamoto N, Ohe Y, Kudou K, Asato T, Zhang P, Nakagawa K. Brigatinib in Japanese patients with ALK-positive non-small-cell lung cancer: Final results of the phase 2 J-ALTA trial. Cancer science, 114:3698-3707, 2023
31. Nakamura T, Yoshida T, Takeyasu Y, Masuda K, Sinno Y, Matsumoto Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. Distinct metastatic spread and progression patterns in patients treated with crizotinib for ROS1- and ALK-rearranged non-small cell lung cancer: a single-center retrospective study. Translational lung cancer research, 12:1436-1444, 2023
32. Okuma Y, Nomura S, Ninomiya K, Gyotoku H, Murakami S, Kogure Y, Harada D, Okishio K, Okamoto H, Goto Y. Continuation of osimertinib in EGFR-mutant non-small-cell lung cancer patients bearing CNS metastasis (EPONA study). Future oncology (London, England), 19:1515-1521, 2023
33. Torasawa M, Horinouchi H, Yagishita S, Utsumi H, Okuda K, Takekoshi D, Ito S, Wakui H, Murata S, Kaku S, Okuma K, Matsumoto Y, Shinno Y, Okuma Y, Yoshida T, Goto Y, Yamamoto N, Araya J, Ohe Y, Fujita Y. Exploratory analysis to predict pneumonitis during durvalumab consolidation therapy for patients with locally advanced non-small cell lung cancer from proteomic profiling of circulating extracellular vesicles. Thoracic cancer, 14:2909-2923, 2023
34. Tanaka T, Goto Y, Horie M, Masuda K, Shinno Y, Matsumoto Y, Okuma Y, Yoshida T, Horinouchi H, Motoi N, Yatabe Y, Watanabe S, Yamamoto N, Ohe Y. Whole Exome Sequencing of Thymoma Patients Exhibiting Exceptional Responses to Pemetrexed Monotherapy. Cancers, 15:4018, 2023
35. Kim HR, Sugawara S, Lee JS, Kang JH, Inui N, Hida T, Lee KH, Yoshida T, Tanaka H, Yang CT, Nishio M, Ohe Y, Tamura T, Yamamoto N, Yu CJ, Akamatsu H, Takahashi S, Nakagawa K. First-line nivolumab, paclitaxel, carboplatin, and bevacizumab for advanced non-squamous non-small cell lung cancer: Updated survival analysis of the ONO-4538-52/TASUKI-52 randomized controlled trial. Cancer medicine, 12:17061-17067, 2023
36. Uehara Y, Koyama T, Katsuya Y, Sato J, Sudo K, Kondo S, Yoshida T, Shoji H, Shimoi T, Yonemori K, Yamamoto N. Travel Time and Distance and Participation in Precision Oncology Trials at the National Cancer Center Hospital. JAMA network open, 6:e2333188, 2023
37. Fukuda A, Yoshida T. Treatment of advanced ALK-rearranged NSCLC following second-generation ALK-TKI failure. Expert review of anticancer therapy, 23:1157-1167, 2023
38. Ikeda H, Nagasaki J, Shimizu D, Katsuya Y, Horinouchi H, Hosomi Y, Tanji E, Iwata T, Itami M, Kawazu M, Ohe Y, Suzuki T, Togashi Y. Immunologic Significance of CD80/CD86 or Major Histocompatibility Complex-II Expression in Thymic Epithelial Tumors. JTO clinical and research reports, 4:100573, 2023
39. Satake T, Kondo S, Tanabe N, Mizuno T, Katsuya Y, Sato J, Koyama T, Yoshida T, Hirata M, Yamamoto N. Pathogenic Germline Variants in BRCA1/2 and p53 Identified by Real-world Comprehensive Cancer Genome Profiling Tests in Asian Patients. Cancer research communications, 3:2302-2311, 2023
