Annual Report 2023
Department of Gastric Surgery
Takaki Yoshikawa, Yukinori Yamagata, Tsutomu Hayashi, Takeyuki Wada, Kenichi Ishizu
Introduction
Our division provides surgical treatment for patients with gastric cancer, adenocarcinoma of esophago-gastric junction, and gastrointestinal stromal tumors (GIST). The aim of our division is to contribute to human health through treatment and research in this field. Our policy to achieve this goal is to (1) provide high quality treatment for patients, (2) efficiently perform clinical tasks, and (3) contribute to developing treatments through clinical trials and clinical research and to provide high quality education.
The Team and What We Do
(1) We conduct daily morning conference to determine the treatment strategy for new patients and those who require any new treatment to provide high quality treatment. All the staff check the endoscopy and CT and determine the tumor staging based on the uniform criteria, and discuss the treatment strategy for each patient based on the problem when applying the standard treatment and the patient's indication to be a candidate for any clinical trial. Surgical morbidity and hospital stay were dramatically improved after introducing the Enhanced Recovery After Surgery program (ERAS) for perioperative care.
(2) We prepared the medical documents for the patients for the uniform explanation and the printed protocol at the conference room and the outpatient clinic to check the applicability to the clinical trial for an efficient clinical task. We also relieve the physicians and residents from the task of preparing the database.
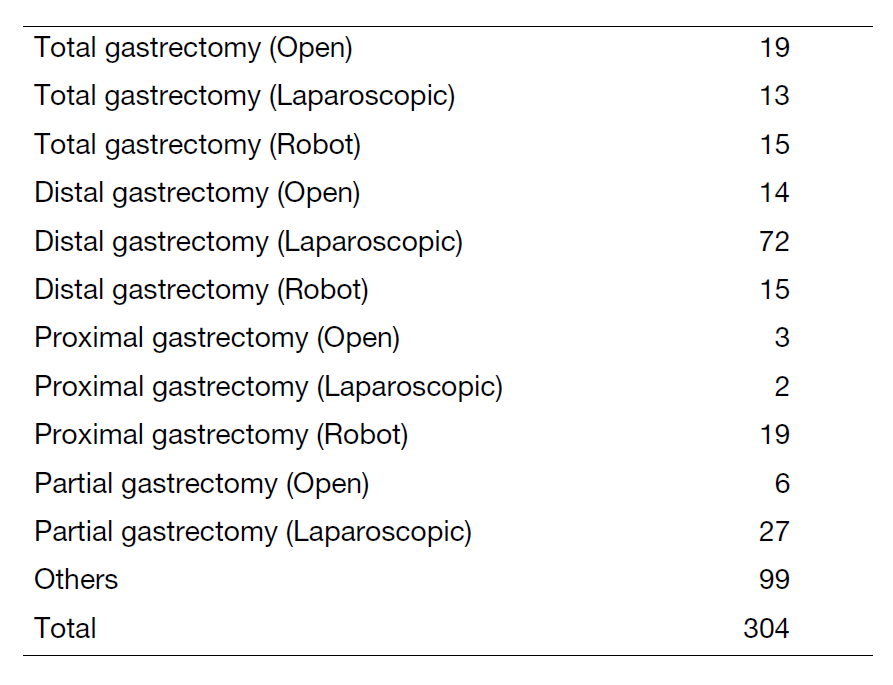
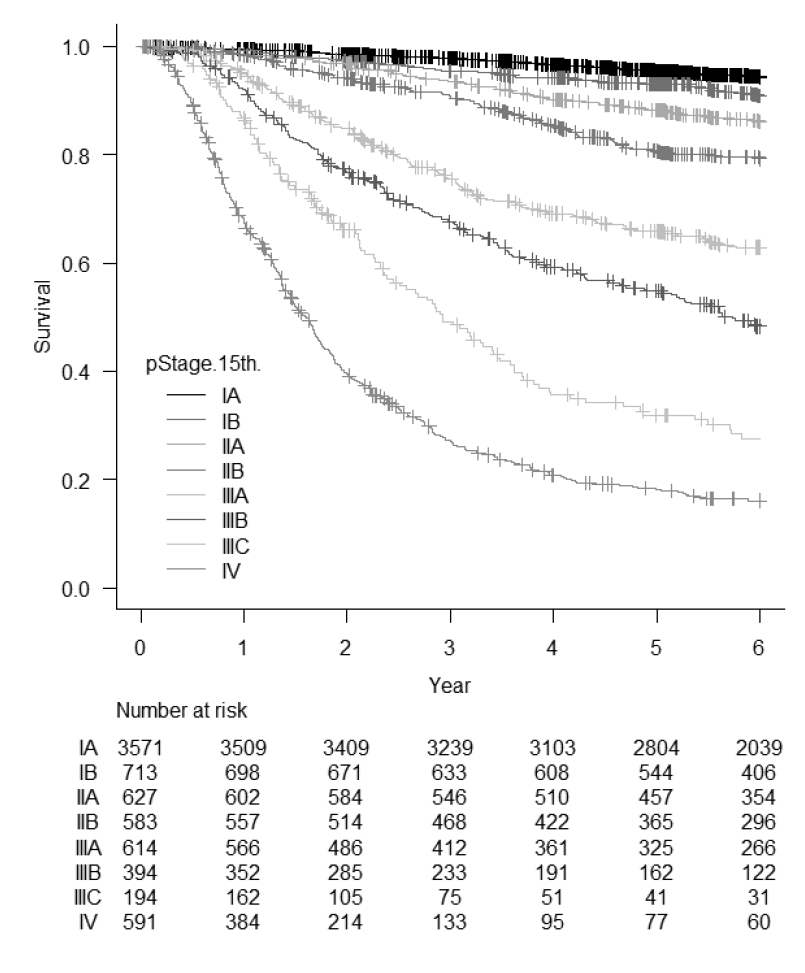
(3) Our division operated a total of 304 cases of gastric cancer, GIST, and others this year. Detailed surgical procedure is shown in Table 1. The proportion of laparoscopic surgery is increased year by year; from 49% (2017) to 65% (2018), 62% (2019), 62% (2020), 74% (2021), 87.7% (2022), and 86.1% (2023) of distal/pylorus-preserving gastrectomy and from 12% (2017) to 21% (2018), 44% (2019), 37% (2020), 46% (2021), 67.8% (2022), and 69.0% (2023) of total/proximal gastrectomy. Especially, we operated 15 cases of robot assisted-distal gastrectomy, 19 cases of proximal gastrectomy, and 15 cases of total gastrectomy without morbidity. On the other hand, extended surgery is not rare. We gained experience of numerous cases of D2 plus para-aortic nodal dissection after neoadjuvant chemotherapy for bulky nodal disease or para-aortic nodal metastasis and several left upper abdominal exenteration cases for bulky GIST invading adjacent organs and lower mediastinal dissection through trans-hiatal approach and by right video-assisted trans-thoracic approach for gastric cancer invading the lower esophagus. We can provide any type of surgery for the local control as applicable. Lastly, we presented the overall survival curves of 7,287 patients who received surgery between 2000 and 2018 (Figure 1). The overall 5-year survival rate was 95.5% in stage IA, 93.0% in stage IB, 88.2% in stage IIA, 80.7% in stage IIB, 66.0% in stage IIIA, 54.8% in stage IIIB, 31.9% in stage IIIC, and 18.1% in stage IV following the 15th edition of the Japanese Classification of Gastric Cancer.
Research Activities
In the JCOG trials, we continued accrual of new patients to JCOG1509 phase III (surgery followed by adjuvant chemotherapy vs neoadjuvant chemotherapy of S-1/oxaliplatin followed by surgery and adjuvant chemotherapy for patients with SS/SE gastric cancer invading lymph nodes), JCOG1711 phase III (omentectomy vs omental preservation for SS/SE gastric cancer), JCOG1809 phase II (laparoscopic splenic hilar nodal dissection for upper gastric cancer invading the greater curvature), JCOG1902 single-arm phase III (ESD for submucosal gastric cancer in the elderly patients), and JCOG1907 phase III (laparoscopic vs robotic gastrectomy for clinical stage I/IIA gastric cancer), JCOG2203 phase II/III (Neoadjuvant chemotherapy of DCS or FLOT for resectable advanced esophago-gastric junction cancer), JCOG2204 phase II (Neoadjuvant chemotherapy of DCS or FLOT for type 4 or giant type 3 gastric cancer), while followed-up the patients who had enrolled in the clinical trial of JCOG1507 phase III (surgery alone vs S-1 for elderly patients with pathological stage II/III gastric cancer), JCOG1301C randomized phase II (neoadjuvant chemotherapy of S-1/CDDP with or without trastuzumab for HER2 positive gastric cancer invading lymph nodes), and JCOG1401 single-arm phase III (laparoscopic total/proximal gastrectomy for stage I), and JCOG1704 phase II (neoadjuvant chemotherapy of DOS followed by D2 plus para-aortic nodal dissection and adjuvant chemotherapy).
In JCOG1104 phase III (8 courses of S-1 vs 4 courses of S-1 for stage II), we published final 5-year follow-up results in Gastric Cancer as a primary investigator. In addition, we are now preparing a new JCOG2301 phase III as a representative investigator to confirm efficacy of conversion surgery after 1st line chemotherapy for stage IV gastric cancer.
On the collaborative research with the UK, the Netherlands, Singapore, and Germany, we published one paper in Gastric Cancer.
On a global phase III study to confirm the efficacy of durvalumab, a new PDL-1 antibody, in addition to FLOT, for stage II/III gastric cancer, we published the primary results in Lancet Oncology as a collaborative investigator.
Education
Through open conferences to determine tumor staging and treatment strategy, the trainees could study the standard treatment, the problem for applying the standard therapy, and the investigational treatment. They also could experience the first/second assistance of surgery performed by experienced staff, the scopist of laparoscopic surgery, and even the operator under the instruction of experienced staff. To obtain the certification of the endoscopic surgeon, the trainees could experience the animal training and receive video-review by experienced staff. Following the success of past residents, the chief resident of this year passed the endoscopic surgeon certification, despite the exam having a pass rate of around 20%. Since 2018, the pass rate in our department has been over 80%.
Young surgeons also continued clinical research and published a total of 5 English papers; Ann Gastroenterol Surg x2, BMC Surgery, Gastric Cancer, and World J Surg.
Future Prospects
We will continue the high quality treatment, efficient clinical tasks, clinical trials, clinical research, and education in the field of gastric surgery and contribute to the human being.
List of papers published in 2023
Journal
1. Kunisaki C, Katai H, Sakuramoto S, Mizusawa J, Katayama H, Kadoya S, Yamada T, Kinoshita T, Yoshikawa T, Terashima M. A nonrandomized controlled trial: long-term outcomes of LATG/LAPG for cStage I gastric cancer: Japan Clinical Oncology Group Study JCOG1401. Gastric cancer, 27:164-175, 2024
2. Kita R, Yanagimoto Y, Imazeki H, Booka E, Tsushima T, Mizusawa J, Sasaki K, Fukuda H, Kurokawa Y, Takeuchi H, Kato K, Kitagawa Y, Boku N, Yoshikawa T, Terashima M. Protocol digest of a randomized controlled adaptive Phase II/III trial of neoadjuvant chemotherapy for Japanese patients with oesophagogastric junction adenocarcinoma: Japan Clinical Oncology Group Study JCOG2203 (NEO-JPEG). Japanese journal of clinical oncology, 54:206-211, 2024
3. Yoshikawa T, Terashima M, Mizusawa J, Nunobe S, Nishida Y, Yamada T, Kaji M, Nomura T, Hato S, Choda Y, Yabusaki H, Yoshida K, Misawa K, Masuzawa T, Tsuda M, Kawachi Y, Katayama H, Fukuda H, Kurokawa Y, Boku N, Sano T, Sasako M. 5-year follow-up results of a JCOG1104 (OPAS-1) phase III non-inferiority trial to compare 4 courses and 8 courses of S-1 adjuvant chemotherapy for pathological stage II gastric cancer. Gastric cancer, 27:155-163, 2024
4. Kurokawa Y, Doki Y, Kitabayashi R, Yoshikawa T, Nomura T, Tsuji K, Goto M, Cho H, Hihara J, Hiki N, Nunobe S, Mizusawa J, Boku N, Terashima M. Short-term outcomes of preoperative chemotherapy with docetaxel, oxaliplatin, and S-1 for gastric cancer with extensive lymph node metastasis (JCOG1704). Gastric cancer, 27:366-374, 2024
5. Tokunaga M, Machida N, Mizusawa J, Ito S, Yabusaki H, Hirao M, Watanabe M, Imamura H, Kinoshita T, Yasuda T, Hihara J, Fukuda H, Yoshikawa T, Boku N, Terashima M. Early endpoints of a randomized phase II trial of preoperative chemotherapy with S-1/CDDP with or without trastuzumab followed by surgery for HER2-positive resectable gastric or esophagogastric junction adenocarcinoma with extensive lymph node metastasis: Japan Clinical Oncology Group study JCOG1301C (Trigger Study). Gastric cancer, 27:580-589, 2024
6. Toriumi T, Terashima M, Mizusawa J, Uemura K, Kurokawa Y, Takiguchi S, Doki Y, Hihara J, Imamura H, Takagane A, Ito S, Yoshikawa T, Sano T, Sasako M. Association between the antiadhesion membrane and small bowel obstruction after open gastrectomy: A supplemental analysis of the randomized controlled JCOG1001 trial. Annals of gastroenterological surgery, 8:30-39, 2024
7. Taki Y, Ito S, Mizusawa J, Yura M, Sato Y, Nomura T, Tsuda M, Omori T, Kunisaki C, Choda Y, Cho H, Hiki N, Boku N, Yoshikawa T, Katai H, Terashima M. Risk factors for abdominal surgical infectious complications after distal gastrectomy for gastric cancer: A post-hoc analysis of a randomized controlled trial (JCOG0912). European journal of surgical oncology, 50:107982, 2024
8. Takeuchi C, Yamashita S, Liu YY, Takeshima H, Sasaki A, Fukuda M, Hashimoto T, Naka T, Ishizu K, Sekine S, Yoshikawa T, Hamada A, Yamamichi N, Fujishiro M, Ushijima T. Precancerous nature of intestinal metaplasia with increased chance of conversion and accelerated DNA methylation. Gut, 73:255-267, 2024
9. Shitara K, Rha SY, Wyrwicz LS, Oshima T, Karaseva N, Osipov M, Yasui H, Yabusaki H, Afanasyev S, Park YK, Al-Batran SE, Yoshikawa T, Yanez P, Dib Bartolomeo M, Lonardi S, Tabernero J, Van Cutsem E, Janjigian YY, Oh DY, Xu J, Fang X, Shih CS, Bhagia P, Bang YJ. Neoadjuvant and adjuvant pembrolizumab plus chemotherapy in locally advanced gastric or gastro-oesophageal cancer (KEYNOTE-585): an interim analysis of the multicentre, double-blind, randomised phase 3 study. The Lancet. Oncology, 25:212-224, 2024
10. Yamagata Y, Komiyama M, Iwata S. Clinical characteristics and management of primary retroperitoneal sarcoma: A literature review. Annals of gastroenterological surgery, 8:21-29, 2024
11. Terashima M, Fujitani K, Yang HK, Mizusawa J, Tsujinaka T, Nakamura K, Katayama H, Lee HJ, Lee JH, An JY, Takagane A, Park YK, Choi SH, Song KY, Ito S, Park DJ, Jin SH, Boku N, Yoshikawa T, Sasako M. Role of reduction gastrectomy in patients with gastric cancer with a single non-curable factor: Supplementary analysis of REGATTA trial. Annals of gastroenterological surgery, 7:741-749, 2023
12. Makuuchi R, Terashima M, Terada M, Mizusawa J, Kita R, Tokunaga M, Omori T, Ojima T, Ehara K, Watanabe M, Yanagimoto Y, Nunobe S, Kinoshita T, Ito S, Nishida Y, Hihara J, Boku N, Kurokawa Y, Yoshikawa T. Randomized controlled phase III trial to investigate superiority of robot-assisted gastrectomy over laparoscopic gastrectomy for clinical stage T1-4aN0-3 gastric cancer patients (JCOG1907, MONA LISA study): a study protocol. BMC cancer, 23:987, 2023
13. Hashimoto T, Nakayama I, Ohashi M, Mizusawa J, Kawachi H, Kita R, Fukuda H, Kurokawa Y, Boku N, Yoshikawa T, Terashima M. Randomized phase II study comparing neoadjuvant 5-fluorouracil/oxaliplatin/docetaxel versus docetaxel/oxaliplatin/S-1 for patients with type 4 or large type 3 gastric cancer. Future oncology (London, England), 19:2147-2155, 2023
14. Hikage M, Hato S, Uemura K, Yura M, Sato Y, Matsushita H, Cho H, Hiki N, Kunisaki C, Inoue K, Choda Y, Boku N, Yoshikawa T, Katai H, Terashima M. Late complication after gastrectomy for clinical stage I cancer: supplementary analysis of JCOG0912. Surgical endoscopy, 37:2958-2968, 2023
15. Satake H, Lee KW, Chung HC, Lee J, Yamaguchi K, Chen JS, Yoshikawa T, Amagai K, Yeh KH, Goto M, Chao Y, Lam KO, Han SR, Shiratori S, Shah S, Shitara K. Pembrolizumab or pembrolizumab plus chemotherapy versus standard of care chemotherapy in patients with advanced gastric or gastroesophageal junction adenocarcinoma: Asian subgroup analysis of KEYNOTE-062. Japanese journal of clinical oncology, 53:221-229, 2023
16. Yamagata Y, Yoshikawa T, Ishizu K, Tsutsui M, Wada T, Hayashi T. Impact of D2 Gastrectomy for Locally Advanced Gastric Cancer in the Era of Effective Adjuvant Chemotherapy. World journal of surgery, 47:1512-1518, 2023
17. Akiyama Y, Katai H, Kitabayashi R, Nunobe S, Koeda K, Yura M, Sato Y, Yoshikawa T, Terashima M. Frequency of lymph node metastasis according to tumor location in clinical T1 early gastric cancer: supplementary analysis of the Japan Clinical Oncology Group study (JCOG0912). Journal of gastroenterology, 58:519-526, 2023
18. Nishino M, Yoshikawa T, Yura M, Sakon R, Ishizu K, Wada T, Hayashi T, Yamagata Y. Possible candidates for splenic hilar nodal dissection among patients with upper advanced gastric cancer without invasion of the greater curvature. Gastric cancer, 26:460-466, 2023
19. Yoshikawa T, Sano T, Terashima M, Yamaguchi K, Bando E, Kawabata R, Yabusaki H, Shinohara H, Oba MS, Kimura T, Takita A, Sasako M. Incidence and risk factors for venous thromboembolism in the Cancer-VTE Registry stomach cancer subcohort. Gastric cancer, 26:493-503, 2023
20. Takahari D, Katai H, Takashima A, Izawa N, Ishizuka N, Ohashi M, Mikami S, Wakatsuki T, Nakayama I, Chin K, Ida S, Kumagai K, Nunobe S, Iwasa S, Shoji H, Wada T, Doi A, Yoshikawa T, Sano T, Boku N, Yamaguchi K. Perioperative TAS-118 plus oxaliplatin in patients with locally advanced gastric cancer: APOLLO-11 study. Gastric cancer, 26:614-625, 2023
21. Muro K, Shitara K, Yamaguchi K, Yoshikawa T, Satake H, Hara H, Sugimoto N, Machida N, Goto M, Kawakami H, Amagai K, Omuro Y, Esaki T, Hironaka S, Nishina T, Komatsu Y, Matsubara H, Shiratori S, Han S, Satoh T, Ohtsu A. Efficacy of Pembrolizumab Monotherapy in Japanese Patients with Advanced Gastric or Gastroesophageal Junction Cancer. Journal of gastrointestinal cancer, 54:951-961, 2023
22. Aoyama T, Oba K, Honda M, Muto M, Mayanagi S, Maeda H, Kanda M, Kashiwabara K, Sakamoto J, Yoshikawa T. The clinical impacts of postoperative complications after colon cancer surgery for the clinical course of adjuvant treatment and survival. International journal of clinical oncology, 28:777-784, 2023
23. Suzuki H, Nonaka S, Maetani I, Matsuda T, Abe S, Yoshinaga S, Oda I, Yamagata Y, Yoshikawa T, Saito Y. Clinical and endoscopic features of metachronous gastric cancer with possible lymph node metastasis after endoscopic submucosal dissection and Helicobacter pylori eradication. Gastric cancer, 26:743-754, 2023
24. Veldhuizen GP, Röcken C, Behrens HM, Cifci D, Muti HS, Yoshikawa T, Arai T, Oshima T, Tan P, Ebert MP, Pearson AT, Calderaro J, Grabsch HI, Kather JN. Deep learning-based subtyping of gastric cancer histology predicts clinical outcome: a multi-institutional retrospective study. Gastric cancer, 26:708-720, 2023
25. Mayanagi S, Oba K, Aoyama T, Tanaka K, Kanda M, Honda M, Maeda H, Kashiwabara K, Muto M, Sakamoto J, Yamagishi H, Yoshikawa T. Feasibility and Safety of Adjuvant Chemotherapy for Resected Colorectal Cancer in Patients With Renal Insufficiency: A Pooled Analysis of Individual Patient Data from Five Japanese Large-scale Clinical Trials. Anticancer research, 43:3089-3095, 2023
26. Watanabe H, Hasegawa S, Kumazu Y, Hashimoto I, Komori K, Suematsu H, Kano K, Fujikawa H, Aoyama T, Yamada T, Yukawa N, Yoshikawa T, Rino Y, Saito A, Ogata T, Oshima T. Can Clinical Stage in the 8(th) Edition of the Union for International Cancer Control TNM Classification Stratify the Prognosis of Patients Undergoing Curative Surgery for Siewert Type II/III Adenocarcinoma of the Esophagogastric Junction? In vivo (Athens, Greece), 37:1790-1796, 2023
27. Kamiya A, Hayashi T, Sakon R, Ishizu K, Wada T, Otsuki S, Yamagata Y, Katai H, Yoshikawa T. Acceleration of sarcopenia in elderly patients who develop asymptomatic pneumonia shadow within one year after surgery for early gastric cancer. BMC surgery, 23:232, 2023
28. Nishikawa K, Kawakami H, Shimokawa T, Fujitani K, Tamura S, Endo S, Kobayashi M, Kawada J, Kurokawa Y, Tsuburaya A, Yoshikawa T, Sakamoto J, Satoh T. Meta-analysis of three randomized trials of capecitabine plus cisplatin (XP) versus S-1 plus cisplatin (SP) as first-line treatment for advanced gastric cancer. International journal of clinical oncology, 28:1501-1510, 2023
29. Hayashi T, Takasawa K, Yoshikawa T, Hashimoto T, Sekine S, Wada T, Yamagata Y, Suzuki H, Abe S, Yoshinaga S, Saito Y, Kouno N, Hamamoto R. A discrimination model by machine learning to avoid gastrectomy for early gastric cancer. Annals of gastroenterological surgery, 7:913-921, 2023
