Annual Report 2023
Department of Gastrointestinal Medical Oncology
Ken Kato, Atsuo Takashima, Hirokazu Shoji, Hidekazu Hirano, Toshiharu Hirose, Natsuko Okita, Shun Yamamoto Yoshitaka Honma,Yuri Yoshinami, Nozomu Ogura, Kunihito Matuguma, Shotaro Yamaguchi, Momoko Sano
Introduction
The Department of Gastrointestinal Medical Oncology specializes in the medical treatment of malignant tumors originating in the digestive tract, including gastric cancer, esophagogastric junction cancer, colorectal cancer, and rare cancers of the digestive tract such as anal canal cancer, gastrointestinal stromal tumors (GISTs), and small intestine cancer. They collaborate with the Departments of Gastrointestinal Surgery, Colorectal Surgery, and Gastrointestinal Endoscopy to provide comprehensive treatment.
The Team and What We Do
In the outpatient setting, the staff takes the lead, with each member responsible for clinic sessions for 2 to 3 days per week. In the inpatient setting, a team consisting of full-time physicians, cancer specialist trainees, and residents collaboratively provide patient care. Treatment strategies are determined through departmental conferences held in the morning and evening, as well as weekly conferences specifically for gastric cancer and colorectal cancer cases.
In the fiscal year 2023, there were a total of 339 new patients. The breakdown is as follows: 91 with gastric cancer, 182 with colorectal cancer, 4 with appendiceal cancer, 9 with small bowel carcinoma, 23 with GIST, 16 with neuroendocrine neoplasms, and 14 with other types of cancer. There were 928 inpatients (including overlaps), with the breakdown as follows: 282 with gastric cancer, 438 with colorectal cancer, 13 with appendiceal cancer, 32 with small bowel carcinoma, 30 with GIST, 88 with neuroendocrine neoplasms, and 45 with other types of cancer. In the fiscal year 2023, 105 patients were enrolled in clinical trials or studies (Table 1).
Table 1. Clinical trials and the number of registered patients in the fiscal year 2023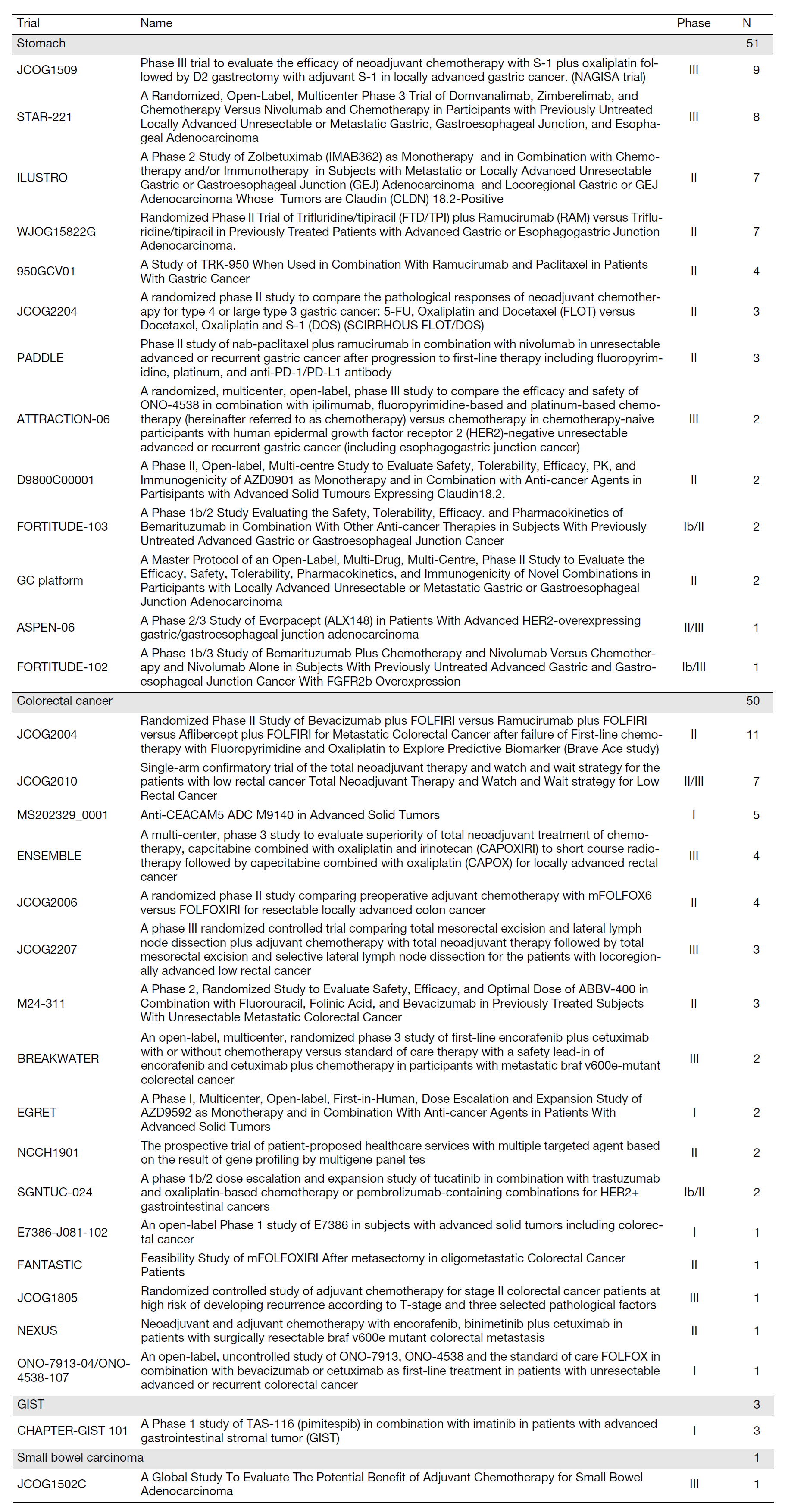

Research Activities
With the aim of establishing better treatment methods, our department actively engages in clinical research. Our staff members play a central role in conceiving new trials for the Japan Clinical Oncology Group (JCOG) and the West Japan Oncology Group (WJOG), actively participating in the development of new standard treatments. As an institution, we proactively enroll patients in these trials, contributing to the establishment of novel treatment standards.
Furthermore, we participate in domestic and international clinical trials ranging from Phase I to Phase III, not only for late-stage treatment development but also for the development of new drugs, starting from the planning stage. Additionally, we conduct physician-led trials for second-line treatment of gastric cancer. We actively engage in translational research (TR) in collaboration with research institutes and other basic research facilities and propose new physician-led trials to pharmaceutical companies.
We published 48 papers, of which 38 were in English-language journals and 4 were English-language reviews. Among these, our department was the lead or responsible author in 9 papers.
Clinical Trials
In the fiscal year 2023, a total of 105 patients were enrolled in clinical trials, as shown in Table 1. We presented 82 papers at conferences, with 54 of them being international conferences. (Table 1).
Education
In patient care, full-time physicians collaborate with cancer specialist trainees/residents to form teams and provide guidance through their interactions with assigned patients. Additionally, cancer specialist trainees/residents are assigned research topics and receive active guidance in developing, managing, presenting at conferences, and writing papers related to clinical research. In the fiscal year 2023, cancer specialist trainees/residents in our department authored a total of 8 English-language papers as lead authors.
Future Prospects
We are dedicated to excellence in patient care, research, and education, with the goal of continuously improving treatment outcomes for gastrointestinal cancers. In patient care, we adhere to standard treatments as a foundation while also striving for innovation and continuous improvement by addressing unmet patient needs, always aiming for "better than before." We are also committed to fostering collaboration with local hospitals.
In research, we not only lead clinical trials and multicenter collaborative studies using our specialized expertise, but also effectively leverage translational research (TR) in partnership with research institutes. Our objective is to develop treatment approaches that have the potential to become the next standard of care. We actively participate in web conferences with industry partners and international researchers to incorporate a broader perspective into our development efforts.
In education, our aim is not only to train residents in cancer pharmacotherapy but also to cultivate young oncologists with a strong research mindset. We are committed to developing the next generation of researchers who are not only highly specialized, but also critical and creative thinkers.
List of papers published in 2023
Journal
1. Kita R, Matsuda S, Nomura M, Machida R, Sasaki K, Kato K, Goto R, Yoshioka T, Yamamoto S, Tsushima T, Fukuda H, Takeuchi H, Kitagawa Y, Japan Esophageal Oncology Group of the Japan Clinical Oncology Group. Protocol digest of a randomized controlled Phase III study comparing surgery alone versus adjuvant nivolumab versus adjuvant S-1 for locally advanced oesophageal squamous cell carcinoma with no pathological complete response after neoadjuvant chemotherapy followed by curative esophagectomy: Japan Clinical Oncology Group study JCOG2206 (SUNRISE Trial). Japanese Journal of Clinical Oncology, 54:212–216, 2024
2. Ito Y, Hamaguchi T, Takashima A, Mizusawa J, Shimada Y, Shiozawa M, Mizoguchi N, Kodaira T, Komori K, Ohue M, Konishi K, Teraishi F, Kinouchi M, Murata K, Fujita F, Watanabe M, Iinuma G, Ishida F, Saida Y, Matsuda T, Katayama H, Fukuda H, Kanemitsu Y. Definitive S-1/mitomycin-C chemoradiotherapy for stage II/III anal canal squamous cell carcinoma: a phase I/II dose-finding and single-arm confirmatory study (JCOG0903). International journal of clinical oncology, 28:1063-1072, 2023
3. Hashimoto T, Otsu S, Hironaka S, Takashima A, Mizusawa J, Kataoka T, Fukuda H, Tsukamoto S, Hamaguchi T, Kanemitsu Y. Phase II biomarker identification study of anti-VEGF agents with FOLFIRI for pretreated metastatic colorectal cancer. Future oncology (London, England), 19:1593-1600, 2023
4. Hashimoto T, Tsukada Y, Ito M, Kanato K, Mizusawa J, Fukuda H, Tsukamoto S, Takashima A, Kanemitsu Y. Utility of circulating tumour DNA for prognosis and prediction of therapeutic effect in locally recurrent rectal cancer: study protocol for a multi-institutional, prospective observational study (JCOG1801A1, CAP-LR study). BMJ open, 13:e073217, 2023
5. Hashimoto T, Maruyama S, Takii Y, Mizusawa J, Kataoka T, Fukuda H, Tsukamoto S, Takashima A, Hamaguchi T, Kanemitsu Y. A randomized phase II study comparing preoperative mFOLFOX6 versus FOLFOXIRI for locally advanced colon cancer: JCOG2006. Future oncology (London, England), 19:1897-1904, 2023
6. Hashimoto T, Tsukamoto S, Murofushi K, Ito Y, Hirano H, Tsukada Y, Sasaki K, Mizusawa J, Fukuda H, Takashima A, Kanemitsu Y. Total neoadjuvant therapy followed by a watch-and-wait strategy for patients with rectal cancer (TOWARd): protocol for single-arm phase II/III confirmatory trial (JCOG2010). BJS open, 7:zrad110, 2023
7. Imaizumi J, Shida D, Boku N, Igaki H, Itami J, Miyakita Y, Narita Y, Takashima A, Kanemitsu Y. Prognostic factors associated with the transition in treatment methods for brain metastases from colorectal cancer. International journal of clinical oncology, 28:1043-1053, 2023
8. Uehara Y, Koyama T, Katsuya Y, Sato J, Sudo K, Kondo S, Yoshida T, Shoji H, Shimoi T, Yonemori K, Yamamoto N. Travel Time and Distance and Participation in Precision Oncology Trials at the National Cancer Center Hospital. JAMA network open, 6:e2333188, 2023
9. Takahari D, Katai H, Takashima A, Izawa N, Ishizuka N, Ohashi M, Mikami S, Wakatsuki T, Nakayama I, Chin K, Ida S, Kumagai K, Nunobe S, Iwasa S, Shoji H, Wada T, Doi A, Yoshikawa T, Sano T, Boku N, Yamaguchi K. Perioperative TAS-118 plus oxaliplatin in patients with locally advanced gastric cancer: APOLLO-11 study. Gastric cancer, 26:614-625, 2023
10. Ito T, Takayanagi D, Sekine S, Hashimoto T, Shimada Y, Matsuda M, Yamada M, Hamamoto R, Kato T, Shida D, Kanemitsu Y, Boku N, Kohno T, Takashima A, Shiraishi K. Comparison of clinicopathological and genomic profiles in anal squamous cell carcinoma between Japanese and Caucasian cohorts. Scientific reports, 13:3587, 2023
11. Hashimoto T, Takayanagi D, Yonemaru J, Naka T, Nagashima K, Machida E, Kohno T, Yatabe Y, Kanemitsu Y, Hamamoto R, Takashima A, Shiraishi K, Sekine S. A comprehensive appraisal of HER2 heterogeneity in HER2-amplified and HER2-low colorectal cancer. British journal of cancer, 129:1176-1183, 2023
12. Aoki M, Kadowaki S, Takahashi N, Suzuki T, Oshima K, Ando T, Yamamoto Y, Kawakami K, Kito Y, Matsumoto T, Shimozaki K, Miyazaki Y, Yamaguchi T, Nagase M, Tamura T, Amanuma Y, Esaki T, Miura Y, Akiyoshi K, Baba E, Makiyama A, Negoro Y, Nakashima K, Sugimoto N, Nagashima K, Shoji H, Boku N. Pattern of disease progression during third-line or later chemotherapy with nivolumab associated with poor prognosis in advanced gastric cancer: a multicenter retrospective study in Japan. Gastric cancer, 26:132-144, 2023
13. Kato K, Doki Y, Ogata T, Motoyama S, Kawakami H, Ueno M, Kojima T, Shirakawa Y, Okada M, Ishihara R, Kubota Y, Amaya-Chanaga C, Chen T, Matsumura Y, Kitagawa Y. First-line nivolumab plus ipilimumab or chemotherapy versus chemotherapy alone in advanced esophageal squamous cell carcinoma: a Japanese subgroup analysis of open-label, phase 3 trial (CheckMate 648/ONO-4538-50). Esophagus, 20:291-301, 2023
14. Yamaguchi T, Takashima A, Nagashima K, Kumagai K, Yamada T, Terashima M, Yabusaki H, Nishikawa K, Tanabe K, Yunome G, Kawachi Y, Yamada T, Fukagawa T, Kinoshita T, Watanabe M, Ishiyama K, Inoue K, Boku N. Evaluating the efficacy of post-operative chemotherapy after curative resection of stage IV gastric cancer with synchronous oligo metastasis: a multicenter retrospective study. Gastric cancer, 26:307-316, 2023
15. Kadono T, Iwasa S, Nagashima K, Oshima K, Yamamoto S, Hirano H, Okita N, Shoji H, Honma Y, Takashima A, Kato K, Ushijima T, Boku N. Progression patterns and site-specific responses in advanced gastric cancer patients treated with nivolumab. Cancer medicine, 12:9322-9331, 2023
16. Kuboki Y, Terazawa T, Masuishi T, Nakamura M, Watanabe J, Ojima H, Makiyama A, Kotaka M, Hara H, Kagawa Y, Sugimoto N, Kawakami H, Takashima A, Kajiwara T, Oki E, Sunakawa Y, Ishihara S, Taniguchi H, Nakajima TE, Morita S, Shirao K, Takenaka N, Ozawa D, Yoshino T. Trifluridine/tipiracil+bevacizumab (BEV) vs. fluoropyrimidine-irinotecan+BEV as second-line therapy for metastatic colorectal cancer: a randomised noninferiority trial. British journal of cancer, 128:1897-1905, 2023
17. Nojima Y, Aoki M, Re S, Hirano H, Abe Y, Narumi R, Muraoka S, Shoji H, Honda K, Tomonaga T, Mizuguchi K, Boku N, Adachi J. Integration of pharmacoproteomic and computational approaches reveals the cellular signal transduction pathways affected by apatinib in gastric cancer cell lines. Computational and structural biotechnology journal, 21:2172-2187, 2023
18. Watanabe J, Muro K, Shitara K, Yamazaki K, Shiozawa M, Ohori H, Takashima A, Yokota M, Makiyama A, Akazawa N, Ojima H, Yuasa Y, Miwa K, Yasui H, Oki E, Sato T, Naitoh T, Komatsu Y, Kato T, Hihara M, Soeda J, Misumi T, Yamamoto K, Akagi K, Ochiai A, Uetake H, Tsuchihara K, Yoshino T. Panitumumab vs Bevacizumab Added to Standard First-line Chemotherapy and Overall Survival Among Patients With RAS Wild-type, Left-Sided Metastatic Colorectal Cancer: A Randomized Clinical Trial. JAMA, 329:1271-1282, 2023
19. Xu J, Kato K, Raymond E, Hubner RA, Shu Y, Pan Y, Park SR, Ping L, Jiang Y, Zhang J, Wu X, Yao Y, Shen L, Kojima T, Gotovkin E, Ishihara R, Wyrwicz L, Van Cutsem E, Jimenez-Fonseca P, Lin CY, Wang L, Shi J, Li L, Yoon HH. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first-line treatment for advanced or metastatic oesophageal squamous cell carcinoma (RATIONALE-306): a global, randomised, placebo-controlled, phase 3 study. The Lancet. Oncology, 24:483-495, 2023
20. Yamada K, Shiraishi K, Takashima A, Takayanagi D, Saiki Y, Takano S, Tanaka M, Fukunaga M, Sugimoto K, Iwasaki Y, Nakamura Y, Kuwahara D, Tsuji Y, Takano M, Sugihara K, Ajioka Y. Characteristics of anal canal squamous cell carcinoma as an HPV-associated cancer in Japan. International journal of clinical oncology, 28:990-998, 2023
21. Izawa N, Masuishi T, Takahashi N, Shoji H, Yamamoto Y, Matsumoto T, Sugiyama K, Kajiwara T, Kawakami K, Aomatsu N, Kondoh C, Kawakami H, Takegawa N, Esaki T, Shimokawa M, Nishio K, Narita Y, Hara H, Sunakawa Y, Boku N, Moriwaki T, Eguchi Nakajima T, Muro K. A Phase II Trial of Trifluridine/Tipiracil in Combination with Cetuximab Rechallenge in Patients with RAS Wild-Type mCRC Refractory to Prior Anti-EGFR Antibodies: WJOG8916G Trial. Targeted oncology, 18:369-381, 2023
22. Osumi H, Takashima A, Ooki A, Yoshinari Y, Wakatsuki T, Hirano H, Nakayama I, Okita N, Sawada R, Ouchi K, Fukuda K, Fukuoka S, Ogura M, Takahari D, Chin K, Shoji H, Kato K, Ishizuka N, Boku N, Yamaguchi K, Shinozaki E. A multi-institutional observational study evaluating the incidence and the clinicopathological characteristics of NeoRAS wild-type metastatic colorectal cancer. Translational oncology, 35:101718, 2023
23. Pape M, Vissers PAJ, Kato K, Haj Mohammad N, Klarenbeek B, van Laarhoven HWM, Matsuda T, Verhoeven RHA. A population-based comparison of patients with metastatic esophagogastric carcinoma between Japan and the Netherlands. Journal of cancer research and clinical oncology, 149:13323-13330, 2023
24. Lee DH, Kim HR, Keam B, Kato K, Kuboki Y, Gao H, Yovine A, Robbins SH, Ahn MJ. Safety and tolerability of first-line durvalumab with tremelimumab and chemotherapy in esophageal squamous cell carcinoma. Cancer medicine, 12:16066-16075, 2023
25. Ando K, Nakamura Y, Kitao H, Shimokawa M, Kotani D, Bando H, Nishina T, Yamada T, Yuki S, Narita Y, Hara H, Ohta T, Esaki T, Hamamoto Y, Kato K, Yamamoto Y, Minashi K, Ohtsubo K, Izawa N, Kawakami H, Kato T, Satoh T, Okano N, Tsuji A, Yamazaki K, Yoshino T, Maehara Y, Oki E. Mutational spectrum of TP53 gene correlates with nivolumab treatment efficacy in advanced gastric cancer (TP53MUT study). British journal of cancer, 129:1032-1039, 2023
26. Yuki S, Yamazaki K, Sunakawa Y, Taniguchi H, Bando H, Shiozawa M, Nishina T, Yasui H, Kagawa Y, Takahashi N, Denda T, Esaki T, Kawakami H, Satake H, Takashima A, Matsuhashi N, Kato T, Asano C, Abe Y, Nomura S, Yoshino T. Role of plasma angiogenesis factors in the efficacy of first-line chemotherapy combined with biologics in RAS wild-type metastatic colorectal cancer: Results from the GI-SCREEN CRC-Ukit study. Cancer medicine, 12:18702-18716, 2023
27. Mitani S, Kito Y, Hino K, Kawakami K, Izawa N, Hanamura F, Yamamoto Y, Shoji H, Komori A, Boku S, Tsuchihashi K, Kato K, Nonagase Y, Matsumoto T, Furuta M, Kawakami H. Real-World Treatment Sequencing in Vulnerable Patients with Metastatic Colorectal Cancer: A Multicenter Retrospective Study. Targeted oncology, 18:707-715, 2023
28. Baba T, Kusumoto M, Kato T, Kurihara Y, Sasaki S, Oikado K, Saito Y, Endo M, Fujiwara Y, Kenmotsu H, Sata M, Takano T, Kato K, Hirata K, Katagiri T, Saito H, Kuwano K. Clinical and imaging features of interstitial lung disease in cancer patients treated with trastuzumab deruxtecan. International journal of clinical oncology, 28:1585-1596, 2023
29. Shiroyama M, Fukuoka S, Masuishi T, Takashima A, Kumekawa Y, Kajiwara T, Yamazaki K, Shimada Y, Esaki T, Makiyama A, Moriwaki T. Renal impairment as a risk factor for trifluridine/tipiracil-induced adverse events in metastatic colorectal cancer patients from the REGOTAS study. Scientific reports, 13:17931, 2023
30. Narita Y, Matsushima T, Sakamoto Y, Matsuoka H, Tanioka H, Kawakami T, Shoji H, Mizukami T, Izawa N, Nishina T, Yamamoto Y, Mitani S, Nakamura M, Misumi T, Muro K. Chemotherapy after nivolumab for advanced gastric cancer (REVIVE): a prospective observational study. ESMO open, 8:102071, 2023
31. Mishima S, Nakamura Y, Tukachinsky H, Taniguchi H, Kadowaki S, Kato K, Oki E, Satoh T, Aoki D, Yamazaki K, Esaki T, Ueno M, Nishina T, Sunakawa Y, Denda T, Bando H, Kuramoto N, Horasawa S, Abutani H, Lee JK, Madison RW, Oxnard GR, Yoshino T. Validity and utility of blood tumor mutational burden (bTMB) is dependent on circulating tumor DNA (ctDNA) shed: SCRUM-Japan MONSTAR-SCREEN. The Journal of Liquid Biopsy, 1:100003, 2023
32. Tsunoda S, Tsubosa Y, Sasaki K, Machida R, Kita R, Fukuda H, Koyanagi K, Takeuchi H, Kamei T, Mine S, Noma K, Kato K, Kitagawa Y, the Japan Esophageal Oncology Group of Japan Clinical Oncology Group. A multicenter randomized controlled trial of esophagectomy with or without prophylactic supraclavicular node dissection: a phase 3 trial (JCOG2013, MODERN3). Japanese Journal of Clinical Oncology, 53:858–862, 2023
