Annual Report 2023
Department of Hepatobiliary and Pancreatic Oncology
Takuji Okusaka, Hideki Ueno, Chigusa Morizane, Susumu Hijioka, Shunsuke Kondo, Akihiro Ohba, Yoshikuni Nagashio, Yuta Maruki, Tetsuro Takasaki, Shin Yagi
Introduction
The Department of Hepatobiliary and Pancreatic Oncology treats tumors originating from the liver, biliary system or pancreas, including hepatocellular carcinoma (HCC), biliary tract cancer and pancreatic cancer. We work closely with surgeons and radiologists who have special expertise in these areas as part of the multi-disciplinary care given at the National Cancer Center Hospital (NCCH). We also conduct clinical and translational research on hepatobiliary and pancreatic tumors and aim to develop new and more effective diagnostic methods and treatments.
The Team and What We Do
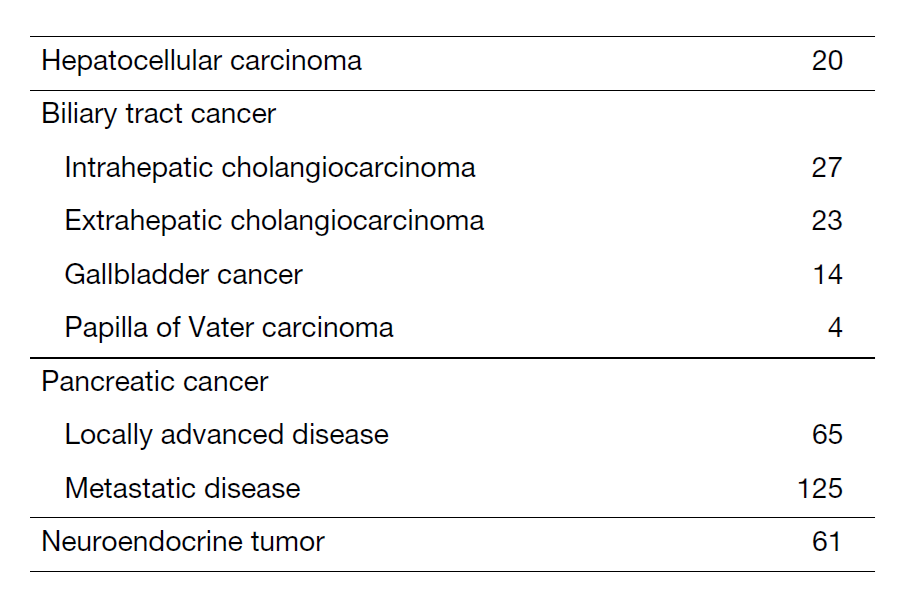
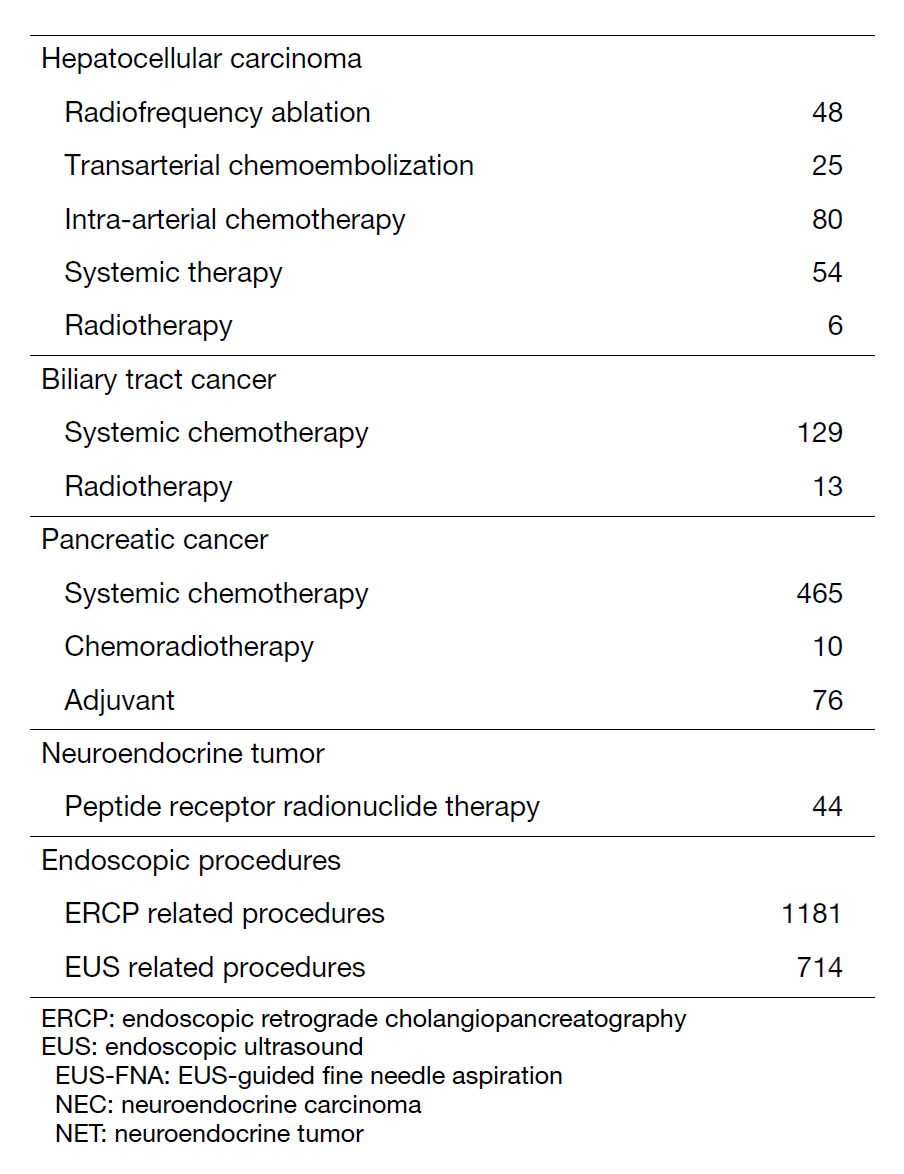
The department consists of eight staff oncologists and several residents. We have used percutaneous ablation therapy for most patients with three or fewer HCC nodules, all of which are smaller than 3 cm in diameter. We also perform transcatheter arterial chemoembolization (TACE), mainly with patients with multiple HCC nodules. Systemic or intra-arterial chemotherapeutic regimens are indicated in patients with advanced HCC for whom locoregional intervention and surgery are unsuitable or have been unsuccessful. In patients with unresectable pancreatic cancer or biliary tract cancer, chemotherapy is performed in clinical practice or as a clinical trial to develop new treatment. We have actively introduced endoscopic procedures for imaging diagnosis (endoscopic ultrasonography (EUS) and endoscopic retrograde cholangiopancreatography (ERCP)), tumor biopsy (EUS-guided tissue acquisition (EUS-TA)), and biliary drainage (including EUS-guided hepaticogastrostomy (EUS-HGS) and EUS-guided choledochoduodenostomy (EUS-CDS)) (Tables 1 and 2).
Research Activities
We published 20 papers as a first author in peer-reviewed journals in 2023.
Clinical Trials
Twenty-four clinical studies are ongoing, including six phase I studies, six phase II studies, and twelve phase III studies, such as adjuvant chemotherapy after resection versus resection alone for patients with resectable tumors, chemotherapy with a new regimen versus standard therapy for patients with advanced tumors, and several observation or registration trials. Our studies are supported by the National Cancer Center Research and Development Fund (2023-J-03, 2022-A-16, 2022-A-05), Project for Development of Innovative Research on Cancer Therapeutics (23ck0106835h0001, 23ck0106682h0003, 23lk0221182h0001, 23lk0201126h0004, 23ck0106766h0002, 23lk0201159h0002, 23ck0106808h0001, 23ama221419h0001, 23ck0106864h0001) from the Japan Agency for Medical Research and Development.
Education
Our staff members are working closely with residents to support their skill development and knowledge expansion in both clinical and research fields. We conduct conferences daily for clinical practice and weekly for research development. The residents in our department published 22 papers as a first author in peer-reviewed journals in 2023, and are performing 30 planning-stage or ongoing studies as a leading researcher, with assistance from staff members.
Future Prospects
Our department will continue to provide the best and latest diagnosis, treatment and supportive care, and develop more effective methods and techniques for all patients with hepatobiliary and pancreatic cancer in this country and globally. Among these, conducting clinical trials with novel promising agents for this disease is considered one of the most important tasks, and establishing cutting-edge endoscopic procedures in this field is the most significant mission for us.
List of papers published in 2023
Journal
1. Okano N, Morizane C, Okusaka T, Sadachi R, Kataoka T, Kobayashi S, Ikeda M, Ozaka M, Mizutani T, Sugimori K, Todaka A, Shimizu S, Mizuno N, Yamamoto T, Sano K, Tobimatsu K, Katanuma A, Gotoh K, Yamaguchi H, Ishii H, Ohba A, Furuse J, Ueno M. Early Tumor Shrinkage and Depth of Response as Predictors of Survival for Advanced Biliary Tract Cancer: An Exploratory Analysis of JCOG1113. The oncologist, 29:e97-e107, 2024
2. Kobayashi S, Nakachi K, Ikeda M, Konishi M, Ogawa G, Sugiura T, Yanagimoto H, Morinaga S, Wada H, Shimada K, Takahashi Y, Nakagohri T, Kamata K, Shimizu Y, Ajiki T, Hirano S, Gotohda N, Ueno M, Okusaka T, Furuse J. Feasibility of S-1 adjuvant chemotherapy after major hepatectomy for biliary tract cancers: An exploratory subset analysis of JCOG1202. European journal of surgical oncology, 50:107324, 2024
3. Komiyama S, Okusaka T, Maruki Y, Ohba A, Nagashio Y, Kondo S, Hijioka S, Morizane C, Ueno H, Sukeda A, Mizui T, Takamoto T, Nara S, Ban D, Esaki M, Hiraoka N, Shimada K. Clinicopathological Findings and Treatment Outcomes of Patients with Primary Hepatobiliary Neuroendocrine Neoplasms: A Retrospective Single-institution Analysis. Internal medicine (Tokyo, Japan), 63:891-901, 2024
4. Sakamoto S, Hiraide H, Minoda M, Iwakura N, Suzuki M, Ando J, Takahashi C, Takahashi I, Murai K, Kagami Y, Mizuno T, Koike T, Nara S, Morizane C, Hijioka S, Kashiro A, Honda K, Watanabe R, Urano Y, Komatsu T. Identification of activity-based biomarkers for early-stage pancreatic tumors in blood using single-molecule enzyme activity screening. Cell reports methods, 4:100688, 2024
5. Kashiro A, Kobayashi M, Oh T, Miyamoto M, Atsumi J, Nagashima K, Takeuchi K, Nara S, Hijioka S, Morizane C, Kikuchi S, Kato S, Kato K, Ochiai H, Obata D, Shizume Y, Konishi H, Nomura Y, Matsuyama K, Xie C, Wong C, Huang Y, Jung G, Srivastava S, Kutsumi H, Honda K. Clinical development of a blood biomarker using apolipoprotein-A2 isoforms for early detection of pancreatic cancer. Journal of gastroenterology, 59:263-278, 2024
6. Hirano K, Maruki Y, Yamashige D, Kobayashi O, Shiotsuka M, Morizane C, Imamura T, Hiraoka N, Okusaka T. Two Cases of Disseminated Alveolar Echinococcosis: The Diagnosis, Management, and Differential Considerations for Liver Lesions. Internal medicine (Tokyo, Japan), 63:1247-1252, 2024
7. Yoo C, Lamarca A, Choi HJ, Vogel A, Pishvaian MJ, Goyal L, Ueno M, Märten A, Teufel M, Geng L, Morizane C. Brightline-2: a phase IIa/IIb trial of brigimadlin (BI 907828) in advanced biliary tract cancer, pancreatic ductal adenocarcinoma or other solid tumors. Future oncology (London, England), 2024
8. Maehara K, Hijioka S, Takeshita K, Kanno A, Okusaka T. Viscosity and Characteristics of Malignant Ascites. Anticancer research, 44:533-542, 2024
9. Rikitake R, Mizushima Y, Yoshimoto S, Higashi T, Satake T, Morizane C, Kawai A. Current status of head and neck sarcomas in Japan in 2016-2019: an analysis using the national cancer registry. International journal of clinical oncology, 29:564-570, 2024
10. Harai S, Hijioka S, Yamada R, Ogura T, Fukasawa M, Okuda A, Horike H, Inoue D, Sekine M, Ishida Y, Koga T, Kitamura H, Tanaka Y, Yoshinari M, Kobayashi K, Chatto M, Yamashige D, Kawasaki Y, Nagashio Y, Okusaka T. Safety of biliary drainage with 6-mm metallic stent for preoperative obstructive jaundice in pancreatic cancer: PURPLE SIX STUDY. Journal of gastroenterology and hepatology, 2024
11. Nakamura M, Sakon M, Sasako M, Okusaka T, Mukai H, Fujiwara K, Kunitoh H, Oba MS, Wada H, Hosokawa J, Takita A, Ikeda M. Association of D-dimer level with thrombotic events, bleeding, and mortality in Japanese patients with solid tumors: a Cancer-VTE Registry subanalysis. International journal of clinical oncology, 29:407-416, 2024
12. Yamashige D, Hijioka S, Nagashio Y, Maruki Y, Fukuda S, Yagi S, Okamoto K, Hara H, Hagiwara Y, Agarie D, Takasaki T, Chatto M, Ohba A, Kondo S, Morizane C, Ueno H, Sone M, Saito Y, Okusaka T. Incidence and factors associated with stent dysfunction and pancreatitis after gastroduodenal stenting for malignant gastric outlet obstruction. Endoscopy international open, 12:E367-E376, 2024
13. Takamizawa S, Koyama T, Sunami K, Sudo K, Hirata M, Kubo T, Tao K, Cho H, Narita Y, Kato K, Yamazaki N, Ohe Y, Okusaka T, Matsui Y, Ogawa C, Yonemori K, Yamamoto N. Identification of barriers to implementation of precision oncology in patients with rare cancers. Cancer science, 115:2023-2035, 2024
14. Ikeda M, Yamashita T, Ogasawara S, Kudo M, Inaba Y, Morimoto M, Tsuchiya K, Shimizu S, Kojima Y, Hiraoka A, Nouso K, Aikata H, Numata K, Sato T, Okusaka T, Furuse J. Multicenter Phase II Trial of Lenvatinib plus Hepatic Intra-Arterial Infusion Chemotherapy with Cisplatin for Advanced Hepatocellular Carcinoma: LEOPARD. Liver cancer, 13:193-202, 2024
15. Mizuno N, Ioka T, Ogawa G, Nakamura S, Hiraoka N, Ito Y, Katayama H, Takada R, Kobayashi S, Ikeda M, Miwa H, Okano N, Kuramochi H, Sekimoto M, Okusaka T, Ozaka M, Todaka A, Gotoh K, Tobimatsu K, Yamaguchi H, Nakagohri T, Kajiura S, Sudo K, Okamura K, Shimizu S, Shirakawa H, Kato N, Sano K, Iwai T, Fujimori N, Ueno M, Ishii H, Furuse J. Effect of systemic inflammatory response on induction chemotherapy followed by chemoradiotherapy for locally advanced pancreatic cancer: an exploratory subgroup analysis on systemic inflammatory response in JCOG1106. Japanese journal of clinical oncology, 53:704-713, 2023
16. Imaoka H, Ikeda M, Nomura S, Morizane C, Okusaka T, Ozaka M, Shimizu S, Yamazaki K, Okano N, Sugimori K, Shirakawa H, Mizuno N, Satoi S, Yamaguchi H, Sugimoto R, Gotoh K, Sano K, Asagi A, Nakamura K, Ueno M. Development of a nomogram to predict survival in advanced biliary tract cancer. Scientific reports, 13:21548, 2023
17. Satake T, Kondo S, Tanabe N, Mizuno T, Katsuya Y, Sato J, Koyama T, Yoshida T, Hirata M, Yamamoto N. Pathogenic Germline Variants in BRCA1/2 and p53 Identified by Real-world Comprehensive Cancer Genome Profiling Tests in Asian Patients. Cancer research communications, 3:2302-2311, 2023
18. Hijioka S, Sakamoto Y, Nagashio Y, Maruki Y, Okusaka T, Saito Y. Novel and safe plastic stent exchange method after endoscopic ultrasound-guided hepaticogastrostomy with incomplete fistula (side hole method). Endoscopy, 55:E24-E25, 2023
19. Hijioka S, Sakamoto Y, Nagashio Y, Maruki Y, Okusaka T, Saito Y. Troubleshooting for endoscopic ultrasound-guided hepaticogastrostomy stent migration: Additional stenting by the partial stent-in-stent method. Endoscopy, 55:E122-E124, 2023
20. Maehara K, Hijioka S, Kawasaki Y, Tamada K, Okusaka T, Saito Y. A novel triple stenting in the treatment of post-choledochojejunostomy reflux cholangitis. Endoscopy, 55:E191-E193, 2023
21. Takeshita K, Hijioka S, Maehara K, Maruki Y, Nagashio Y, Okusaka T, Saito Y. Bile duct radiofrequency ablation for a residual adenoma after endoscopic papillectomy. Endoscopy, 55:E185-E188, 2023
22. Kawasaki Y, Hijioka S, Takeshita K, Tamada K, Okusaka T, Saito Y. Endoscopic ultrasound-guided choledochojejunostomy using a forward-viewing echoendoscopic saddle-cross technique. Endoscopy, 55:E233-E235, 2023
23. Takeshita K, Hijioka S, Kawasaki Y, Maruki Y, Nagashio Y, Okusaka T, Saito Y. Endoscopic ultrasound-guided hepaticojejunostomy for drainage of the right posterior hepatic duct enabled total liver drainage. Endoscopy, 55:E346-E348, 2023
24. Kawasaki Y, Hijioka S, Nagashio Y, Maruki Y, Ohba A, Takeshita K, Takasaki T, Agarie D, Hagiwara Y, Hara H, Okamoto K, Yamashige D, Kondo S, Morizane C, Ueno H, Mizui T, Takamoto T, Nara S, Ban D, Esaki M, Tamada K, Hiraoka N, Saito Y, Okusaka T. Efficacy of endoscopic ultrasound-guided tissue acquisition for solid pancreatic lesions 20 mm or less in diameter suspected as neuroendocrine tumors or requiring differentiation. Journal of gastroenterology, 58:693-703, 2023
25. Umemoto K, Sunakawa Y, Ueno M, Furukawa M, Mizuno N, Sudo K, Kawamoto Y, Kajiwara T, Ohtsubo K, Okano N, Matsuhashi N, Itoh S, Matsumoto T, Shimizu S, Otsuru T, Hasegawa H, Okuyama H, Ohama H, Moriwaki T, Ohta T, Odegaard JI, Nakamura Y, Bando H, Yoshino T, Ikeda M, Morizane C. Clinical significance of circulating-tumour DNA analysis by metastatic sites in pancreatic cancer. British journal of cancer, 128:1603-1608, 2023
26. Okusaka T, Nakamura M, Yoshida M, Kitano M, Ito Y, Mizuno N, Hanada K, Ozaka M, Morizane C, Takeyama Y. Clinical Practice Guidelines for Pancreatic Cancer 2022 from the Japan Pancreas Society: a synopsis. International journal of clinical oncology, 28:493-511, 2023
27. Yoo C, Javle MM, Verdaguer Mata H, de Braud F, Trojan J, Raoul JL, Kim JW, Ueno M, Lee CK, Hijioka S, Cubillo A, Furuse J, Azad N, Sato M, Vugmeyster Y, Machl A, Bajars M, Bridgewater J, Oh DY, Borad MJ. Phase 2 trial of bintrafusp alfa as second-line therapy for patients with locally advanced/metastatic biliary tract cancers. Hepatology (Baltimore, Md.), 78:758-770, 2023
28. Harai S, Hijioka S, Nagashio Y, Ohba A, Maruki Y, Yamashige D, Hisada Y, Yoshinari M, Kitamura H, Maehara K, Murashima Y, Kawasaki Y, Koga T, Takeshita K, Ueno H, Kondo S, Morizane C, Fukasawa M, Sone M, Saito Y, Enomoto N, Okusaka T. Comparison of 6-mm and 10-mm-diameter, fully-covered, self-expandable metallic stents for distal malignant biliary obstruction. Endoscopy international open, 11:E340-E348, 2023
29. Okusaka T, Morimoto M, Eguchi Y, Nakamura S, Iino S, Kageyama R. A Phase I Study to Investigate the Safety, Tolerability and Pharmacokinetics of Napabucasin Combined with Sorafenib in Japanese Patients with Unresectable Hepatocellular Carcinoma. Drugs in R&D, 23:99-107, 2023
30. Okumura T, Fujii T, Terabayashi K, Kojima T, Takeda S, Kashiwada T, Toriyama K, Hijioka S, Miyazaki T, Yamamoto M, Tanabe S, Shirakawa Y, Furukawa M, Honma Y, Hoshino I, Nabeya Y, Yamaguchi H, Uemoto S, Shimada Y, Matsubara H, Ozawa S, Makuuchi H, Imamura M. MicroRNAs associated with postoperative outcomes in patients with limited stage neuroendocrine carcinoma of the esophagus. Oncology letters, 26:276, 2023
31. Kin T, Shimizu Y, Hijioka S, Hara K, Katanuma A, Nakamura M, Yamada R, Itoi T, Ueki T, Masamune A, Hirono S, Koshita S, Hanada K, Kamata K, Yanagisawa A, Takeyama Y. A comparative study between computed tomography and endoscopic ultrasound in the detection of a mural nodule in intraductal papillary mucinous neoplasm -Multicenter observational study in Japan. Pancreatology, 23:550-555, 2023
32. Takamoto T, Maruki Y, Kondo S. Recent updates in the use of pharmacological therapies for downstaging in patients with hepatocellular carcinoma. Expert opinion on pharmacotherapy, 24:1567-1575, 2023
33. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. ASO Author Reflections: Characteristics of Surgery for Biliary Tract Cancer that Should be Considered During the Design of Future Clinical Trials. Annals of surgical oncology, 30:7384-7385, 2023
34. Kobayashi S, Ikeda M, Nakachi K, Ueno M, Okusaka T, Todaka A, Satoi S, Tomokuni A, Konishi M, Furuse J. A Multicenter Survey on Eligibility for a Randomized Phase III Trial of Adjuvant Chemotherapy for Resected Biliary Tract Cancer (JCOG1202, ASCOT). Annals of surgical oncology, 30:7331-7337, 2023
35. Okusaka T, Saiura A, Shimada K, Ikeda M, Ioka T, Kimura T, Hosokawa J, Takita A, Oba MS. Incidence and risk factors for venous thromboembolism in the Cancer-VTE Registry pancreatic cancer subcohort. Journal of gastroenterology, 58:1261-1271, 2023
36. Oh DY, He AR, Qin S, Chen LT, Okusaka T, Vogel A, Kim JW, Suksombooncharoen T, Lee MA, Kitano M, Burris H, Bouattour M, Tanasanvimon S, McNamara MG, Zaucha R, Avallone A, Tan B, Cundom J, Lee CK, Takahashi H, Ikeda M, Chen JS, Wang J, Makowsky M, Rokutanda N, Żotkiewicz M, Kurland JF, Cohen G, Valle JW. Plain language summary of the TOPAZ-1 study: durvalumab and chemotherapy for advanced biliary tract cancer. Future oncology (London, England), 19:2277-2289, 2023
37. Honma Y, Ikeda M, Hijioka S, Matsumoto S, Ito T, Aoki T, Furuse J. Optimal first-line treatment strategies of systemic therapy for unresectable gastrointestinal neuroendocrine tumors based on the opinions of Japanese experts. Investigational new drugs, 41:777-786, 2023
38. Kitamura H, Morizane C, Tanabe N, Go I, Maruki Y, Ohba A, Nagashio Y, Kondo S, Hijioka S, Ueno H, Yoshida T, Okusaka T. Clinical features of germline BRCA1/2 or ATM pathogenic variant positive pancreatic cancer in Japan. Pancreatology, 23:964-969, 2023
39. Koga T, Hijioka S, Nagashio Y, Maruki Y, Maehara K, Murashima Y, Kawasaki Y, Takeshita K, Yamada N, Yoshinari M, Hisada Y, Harai S, Kitamura H, Kawahara S, Ohba A, Morizane C, Ishida Y, Hirai F, Okusaka T. Prospective clinical trial of EUS-guided choledochoduodenostomy without fistula dilation for malignant distal biliary obstruction. Endoscopic ultrasound, 12:409-416, 2023
40. Llovet JM, Kudo M, Merle P, Meyer T, Qin S, Ikeda M, Xu R, Edeline J, Ryoo BY, Ren Z, Masi G, Kwiatkowski M, Lim HY, Kim JH, Breder V, Kumada H, Cheng AL, Galle PR, Kaneko S, Wang A, Mody K, Dutcus C, Dubrovsky L, Siegel AB, Finn RS. Lenvatinib plus pembrolizumab versus lenvatinib plus placebo for advanced hepatocellular carcinoma (LEAP-002): a randomised, double-blind, phase 3 trial. The Lancet. Oncology, 24:1399-1410, 2023