Annual Report 2023
Department of Laboratory Medicine
Hirotaka Matsui, Kuniko Sunami, Wataru Takeda, Naoki Maezawa, Chiaki Hayashi, Shunsuke Tezuka, Yoji Hashimoto, Yasuo Shibuki, Hiroki Kakishima, Tomokazu Suzuki, Chiaki Ikeda, Shuji Ota, Arisa Hanai, Satoshi Ito, Yusuke Okui, Sayaka Takeuchi, Kyosuke Tosawa, Mizuho Fujima , Sachiko Kobayashi, Risa Otsuka, Noriko Takahashi, Saori Nakabayashi, Yuki Kase, Maemi Souma, Tomoe Ito, Kyoko Orihara, Kaori Ueki, Akino Kino, Kana Katsuragi, Ruriko Machida, Haruka Katagiri, Kana Miyajima, Kayo Okauchi, Nanami Sato, Kenta Takehara, Kanako Kanaizuka, Chihiro Yamasaki, Hiroshi Chigira, Kaori Yamaguchi, Kyoko Osanai, Moemi Kasane, Hideya Matsubayashi, Yu Aruga, Misato Tsubokura, Yuka Yasuno, Haruki Mitsuya, Kaho Matsui, Nozomi Shishido, Aya Mikami, Saori Kobayashi, Shingo Nakajima, Saeko Shirahama, Mei Fukuhara, Kumi Nakatani, Hiyori Yatsu, Chika Tokutake, Madoka Kondo, Mayu Takeno, Sakura Ishida, Ayaka Ichikawa, Yusuke Takai, Kazuhiro Yoshida, Daichi Yamada, Motoi Miyakoshi, Takako Takada, Takashi Kubo, Mayuko Kitami, Satoyo Oda, Haruka Hirakochi, Shigeru Tamura, Megumi Masuda, Nanako Fujita, Moe Kato, Takeshi Ogasawara, Yu Kawajiri, Kazui Taniguchi, Rio Yamauchi, Yuka Hirano, Noa Suzuki, Miki Nagasawa, Riho Okada, Kazuya Nakanishi, Haruna Omaru, Yuka Oga, Ritsuko Toyama, Chieko Nozawa, Kinue Tsubokawa, Naoki Komiya, Kozaburo Endo
Introduction
The Department of Laboratory Medicine obtained ISO 15189 accreditation, which is an international standard that specifies requirements for quality and competence specific to clinical laboratories, as of September 2012 and has maintained the certification ever since.
The number of test requests to the department in FY2023 was 7,473,259, an increase of 102.6% from FY2022 (Table 1).
Table 1. Number of clinical tests performed

The Team and What We Do
Following a significant equipment upgrade, the Biochemical and General Testing section has integrated the subsections, including Biochemistry, Immunology, Hematology, and Urinalysis subsections. This aims to improve operational efficiency and handle the increasing demand for specimen tests and clinical trial-related services. The Hematology subsection offers a susceptible analysis of minimal residual diseases in hematopoietic malignancies and examines many trace-amount specimens, such as spinal fluid.
The Microbiology Testing section continuously serves PCR testing for COVID-19. It supports surgical and medical activities in the hospital, which includes contribution to nosocomial infection control through testing for drug-resistant bacteria, aiming for proper use of antimicrobial agents.
The Genetic Testing section offers cancer genome profiling tests, including liquid biopsy-based tests, which were newly covered by insurance in FY2021 and have been conducting cancer genome profiling tests than ever before (Table 2). In addition, the section offers NGS-based testing for companion diagnostics.
The Blood Collection section strives to reduce the waiting time for blood collection by setting a goal of "Eighty-five percent of patients wait less than 20 minutes for blood collection" while prioritizing medical safety.
Table 2. Number of sessions and cancer genomic profiling tests in Expert panel meeting 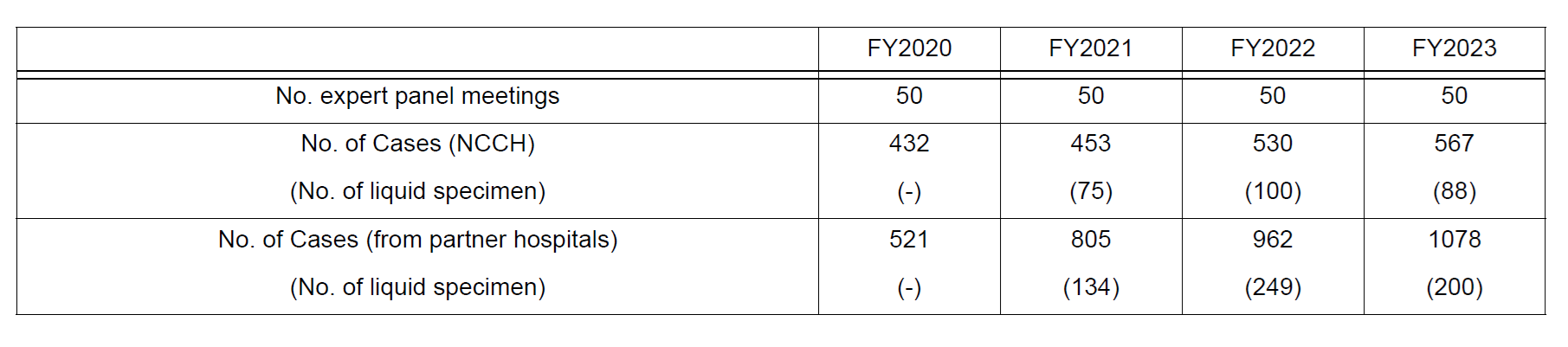

The Blood Transfusion Testing and Cell Processing section contributes to the safe implementation of blood transfusion by preparing a workflow for securing unique products, including blood products for Rh D-negative patients and patients positive for irregular antibodies. Also, the section commits to antigen receptor-expressing T-cell therapy in terms of cell processing.
The Physiology Testing section is actively involved in evaluating cardiotoxicity caused by anticancer drugs and diagnosing hepatic sinusoidal obstruction syndrome/vascular occlusion of the central hepatic vein after hematopoietic stem cell transplantation. The section also cooperates with clinical studies on the relationship between anthracyclines and cardiotoxicity by performing global longitudinal strain measurements.
The Pathology section processes specimens under the guidelines for handling histopathology specimens for genomic medicine and supports companion diagnoses and gene panel tests.
Research Activities
The Department of Laboratory Medicine has conducted basic and clinical research in various fields, especially factors that affect test accuracy. In collaboration with clinical departments and manufacturers, the department is also researching the stability and clinical usefulness of tumor markers, the detection of minimal residual disease in hematopoietic malignancies, and the evaluation of body fluid and blood analysis using an automated blood analysis system. Regarding cancer genome profiling tests, the Genetic Testing section, as the secretariat of the TOP-GEAR project, is conducting clinical research to develop new cancer profiling tests and clinical implementation of whole genome analysis. As for blood transfusion and cell therapy, the department is cooperating with clinical studies conducted by many clinical departments in the hospital, including research on Cell-free and Concentrated Ascites Reinfusion Therapy.
Education
Per ISO 15189 education, training, and training procedures, the department fosters personnel who can reliably provide quality-assured and accurate test results in each of the laboratory sections. The department also actively supports academic activities, including taking qualifying examinations, conference presentations, and writing papers, aiming to train clinical laboratory technicians with professional and educational backgrounds.
Our department actively accepts those who wish to become clinical laboratory technicians as on-site trainees and appeals to them for clinical laboratory work.
Future Prospects
As an ISO 15189-accredited testing facility, the department guarantees the quality and capabilities meeting international standards and promotes cooperation in clinical trials and clinical studies. For biochemical testing, the department is improving the test implementation system to enhance operational efficiency. Additionally, the department is working to increase the personnel who specialize in cell therapy and flow cytometry analyses through professional education to expand these operations' capacity. Regarding cancer genome medicine, the department continues to manage the expert panels in response to the increased number of cancer genome profiling tests. Through carrying out these tasks, the department will establish a clinical laboratory to contribute valuable information from each clinical department and provide it ourselves.
List of papers published in 2023
Journal
1. Ando Y, Shimoi T, Sunami K, Okita N, Nakamura K, Shibata T, Fujiwara Y, Yamamoto N. Progress report of a cross-organ and biomarker-based basket-type clinical trial: BELIEVE Trial. Cancer science, 115:555-563, 2024
2. Shimoi T, Sunami K, Tahara M, Nishiwaki S, Tanaka S, Baba E, Kanai M, Kinoshita I, Shirota H, Hayashi H, Nishida N, Kubo T, Mamesaya N, Ando Y, Okita N, Shibata T, Nakamura K, Yamamoto N. Dabrafenib and trametinib administration in patients with BRAF V600E/R or non-V600 BRAF mutated advanced solid tumours (BELIEVE, NCCH1901): a multicentre, open-label, and single-arm phase II trial. EClinicalMedicine, 69:102447, 2024
3. Watanabe T, Honma Y, Yonemori K, Sunami K, Yoshimoto S, Mori T. High-grade intraductal carcinoma of the parotid gland harboring CTNNA1::ALK rearrangement: Changes in genetic status using genetic testing during treatment with an ALK inhibitor. Head & neck, 46:E26-E31, 2024
4. Shiraishi K, Takahashi A, Momozawa Y, Daigo Y, Kaneko S, Kawaguchi T, Kunitoh H, Matsumoto S, Horinouchi H, Goto A, Honda T, Shimizu K, Torasawa M, Takayanagi D, Saito M, Saito A, Ohe Y, Watanabe SI, Goto K, Tsuboi M, Tsuchihara K, Takata S, Aoi T, Takano A, Kobayashi M, Miyagi Y, Tanaka K, Suzuki H, Maeda D, Yamaura T, Matsuda M, Shimada Y, Mizuno T, Sakamoto H, Yoshida T, Goto Y, Yoshida T, Yamaji T, Sonobe M, Toyooka S, Yoneda K, Masago K, Tanaka F, Hara M, Fuse N, Nishizuka SS, Motoi N, Sawada N, Nishida Y, Kumada K, Takeuchi K, Tanno K, Yatabe Y, Sunami K, Hishida T, Miyazaki Y, Ito H, Amemiya M, Totsuka H, Nakayama H, Yokose T, Ishigaki K, Nagashima T, Ohtaki Y, Imai K, Takasawa K, Minamiya Y, Kobayashi K, Okubo K, Wakai K, Shimizu A, Yamamoto M, Iwasaki M, Matsuda K, Inazawa J, Shiraishi Y, Nishikawa H, Murakami Y, Kubo M, Matsuda F, Kamatani Y, Hamamoto R, Matsuo K, Kohno T. Identification of telomere maintenance gene variations related to lung adenocarcinoma risk by genome-wide association and whole genome sequencing analyses. Cancer communications (London, England), 44:287-293, 2024
5. Mochizuki A, Shiraishi K, Honda T, Higashiyama RI, Sunami K, Matsuda M, Shimada Y, Miyazaki Y, Yoshida Y, Watanabe SI, Yatabe Y, Hamamoto R, Kohno T. Passive Smoking-Induced Mutagenesis as a Promoter of Lung Carcinogenesis. Journal of thoracic oncology, S1556-0864(24)00074-1, 2024
6. Sunami K, Naito Y, Saigusa Y, Amano T, Ennishi D, Imai M, Kage H, Kanai M, Kenmotsu H, Komine K, Koyama T, Maeda T, Morita S, Sakai D, Hirata M, Ito M, Kozuki T, Sakashita H, Horinouchi H, Okuma Y, Takashima A, Kubo T, Hironaka S, Segawa Y, Yakushijin Y, Bando H, Makiyama A, Suzuki T, Kinoshita I, Kohsaka S, Ohe Y, Ishioka C, Yamamoto K, Tsuchihara K, Yoshino T. A Learning Program for Treatment Recommendations by Molecular Tumor Boards and Artificial Intelligence. JAMA oncology, 10:95-102, 2024
7. Takamizawa S, Koyama T, Sunami K, Sudo K, Hirata M, Kubo T, Tao K, Cho H, Narita Y, Kato K, Yamazaki N, Ohe Y, Okusaka T, Matsui Y, Ogawa C, Yonemori K, Yamamoto N. Identification of barriers to implementation of precision oncology in patients with rare cancers. Cancer science, 115:2023-2035, 2024
8. Ishimaru S, Shimoi T, Sunami K, Nakajima M, Ando Y, Okita N, Nakamura K, Shibata T, Fujiwara Y, Yamamoto N. Platform trial for off-label oncology drugs using comprehensive genomic profiling under the universal public healthcare system: the BELIEVE trial. International journal of clinical oncology, 29:89-95, 2024
9. Nakamura W, Hirata M, Oda S, Chiba K, Okada A, Mateos RN, Sugawa M, Iida N, Ushiama M, Tanabe N, Sakamoto H, Sekine S, Hirasawa A, Kawai Y, Tokunaga K, Tsujimoto SI, Shiba N, Ito S, Yoshida T, Shiraishi Y. Assessing the efficacy of target adaptive sampling long-read sequencing through hereditary cancer patient genomes. NPJ genomic medicine, 9:11, 2024
10. Matsui H, Hirata M. Evaluation of the pathogenic potential of germline DDX41 variants in hematopoietic neoplasms using the ACMG/AMP guidelines. International journal of hematology, 119:552-563, 2024
11. Miyamoto H, Kawakami F, Abe S, Sugita H, Matsui H. Comprehensive Cancer Genomic Profiling of Liver Metastasis Led to the Unexpected Identification of Colorectal Cancer. Internal medicine (Tokyo, Japan), 63:63-70, 2024
12. Arai H, Matsui H, Chi S, Utsu Y, Masuda S, Aotsuka N, Minami Y. Germline Variants and Characteristic Features of Hereditary Hematological Malignancy Syndrome. International journal of molecular sciences, 25:652, 2024
13. Tao K, Yamazaki F, Kubo T, Sunami K, Kumamoto T, Arakawa A, Sugiyama M, Watanabe Y, Nakajima M, Shirakawa N, Tanimura K, Koyama T, Hirata M, Sudo K, Tanabe N, Watanabe T, Yoshida T, Kitami M, Yoshida A, Yatabe Y, Nakano Y, Ohira M, Kamijo T, Nakazawa A, Kato M, Ichimura K, Kohno T, Yamamoto N, Hishiki T, Ichikawa H, Ogawa C. Pediatric Precision Medicine at the National Cancer Center Japan: Prospective Genomic Study of Pediatric Patients with Cancer as Part of the TOP-GEAR Project. JCO precision oncology, 7:e2200266, 2023
14. Yoshida T, Yatabe Y, Kato K, Ishii G, Hamada A, Mano H, Sunami K, Yamamoto N, Kohno T. The evolution of cancer genomic medicine in Japan and the role of the National Cancer Center Japan. Cancer biology & medicine, 21:29-44, 2023
15. Oda S, Ushiama M, Nakamura W, Gotoh M, Tanabe N, Watanabe T, Odaka Y, Aoyagi K, Sakamoto H, Nakajima T, Sugano K, Yoshida T, Shiraishi Y, Hirata M. A complex rearrangement between APC and TP63 associated with familial adenomatous polyposis identified by multimodal genomic analysis: a case report. Frontiers in oncology, 13:1205847, 2023
16. Tungalag S, Shinriki S, Hirayama M, Nagamachi A, Kanai A, Inaba T, Matsui H. Ribosome profiling analysis reveals the roles of DDX41 in translational regulation. International journal of hematology, 117:876-888, 2023
17. Tsubokura M, Kojima M, Nakabayashi S, Takahashi N, Takeuchi S, Aruga Y, Ikeda C, Maezawa N, Okazaki K, Uchida S, Watanabe M, Aoki J, Ito A, Tanaka T, Inamoto Y, Kim SW, Matsushita H, Fukuda T. EDTA-induced pseudothrombocytopenia in hematopoietic stem cell donor. Clinical case reports, 11:e7023, 2023
18. Yokomizo-Nakano T, Hamashima A, Kubota S, Bai J, Sorin S, Sun Y, Kikuchi K, Iimori M, Morii M, Kanai A, Iwama A, Huang G, Kurotaki D, Takizawa H, Matsui H, Sashida G. Exposure to microbial products followed by loss of Tet2 promotes myelodysplastic syndrome via remodeling HSCs. The Journal of experimental medicine, 220:e20220962, 2023
19. Shinojima N, Ozono K, Yamamoto H, Abe S, Sasaki R, Tomita Y, Kai A, Mori R, Yamamoto T, Uekawa K, Matsui H, Nosaka K, Matsuzaki H, Komohara Y, Mikami Y, Mukasa A. Lynch syndrome-associated chordoma with high tumor mutational burden and significant response to immune checkpoint inhibitors. Brain tumor pathology, 40:185-190, 2023
20. Usuku H, Yamamoto E, Sueta D, Noguchi M, Fujisaki T, Egashira K, Oike F, Fujisue K, Hanatani S, Arima Y, Takashio S, Kawano Y, Oda S, Kawano H, Matsushita K, Ueda M, Matsui H, Matsuoka M, Tsujita K. Prognostic value of right ventricular global longitudinal strain in patients with immunoglobulin light-chain cardiac amyloidosis. European heart journal open, 3:oead048, 2023
21. Tierens A, Kagotho E, Shinriki S, Seto A, Smith AC, Care M, Maze D, Sibai H, Yee KW, Schuh AC, Kim DDH, Gupta V, Minden MD, Matsui H, Capo-Chichi JM. Biallelic disruption of DDX41 activity is associated with distinct genomic and immunophenotypic hallmarks in acute leukemia. Frontiers in oncology, 13:1153082, 2023
22. Nishimura A, Yokoyama K, Naruto T, Yamagishi C, Imamura T, Nakazono H, Kimura S, Ito M, Sagisaka M, Tanaka Y, Piao J, Namikawa Y, Yanagimachi M, Isoda T, Kanai A, Matsui H, Isobe T, Sato-Otsubo A, Higuchi N, Takada A, Okuno H, Saito S, Karakawa S, Kobayashi S, Hasegawa D, Fujisaki H, Hasegawa D, Koike K, Koike T, Rai S, Umeda K, Sano H, Sekinaka Y, Ogawa A, Kinoshita A, Shiba N, Miki M, Kimura F, Nakayama H, Nakazawa Y, Taga T, Taki T, Adachi S, Manabe A, Koh K, Ishida Y, Takita J, Ishikawa F, Goto H, Morio T, Mizutani S, Tojo A, Takagi M. Myeloid/natural killer (NK) cell precursor acute leukemia as a distinct leukemia type. Science advances, 9:eadj4407, 2023
23. Hirayama M, Shinriki S, Matsui H. R-loops in normal and malignant hematopoiesis. Frontiers in Hematology, 2:1297657, 2023
24. Itoyama M, Ohara A, Yokoyama K, Yamamoto S, Kato K, Mori T, Igaki H, Nakano E, Yamazaki N, Sunami K, Nishigori C, Honma Y. Successful Use of Anti-PD-1 Antibody to Treat Multiple Metastatic Carcinomas in a Patient with Xeroderma Pigmentosum: Case Report and Literature Review. Clinical Images and Case Reports Journal, 5:312, 2023
