Center for Research Administration and Support (CRAS)
Teruhiko Yoshida
(Research Administration Section)
Nobuko Ushirozawa, Katsuya Sawadaishi, Yoshimitu Sakamoto, Kiyokazu Kawashima, Shugo Nemoto, Ayako Mesawa, Yuichi Amanuma, Fumie Suzuki, Kiyoka Nagayama, Yuko Yamada, Miki Ito, Sawako Nakayama, Emi Nobuhara, Chinatu Watanabe
(Research Alliance and Intellectual Property Section)
Genta Ohno, Yukari Nakayama, Ryota Kosaka, Junko Kaneko, Ayuri Bakuya
(Research administrators)
Kazunori Aoki, Kazuhiko Aoyagi, Kazuya Tuchihara
(Human Research Protection Section)
Noriko Yamashita, Yayoi Ofuji, Kuniko Takahashi, Norie Enomoto, Yoko Yokota, Satoko Arai, Keiko Koide, Tetsuya Ueda, Sanae Kawada, Megumi Teshirogi, Masahiko Ozaki, Yuki Harada, Takahiro Sakai, Hayato Kamata, Miwa Kanehira , Yumika Yoshikawa, Sayaka Yamamoto, Yoko Matsubuchi
(Biostatistics Division)
Taro Shibata, Aya Kuchiba, Junki Mizusawa, Masashi Wakabayashi, Gakuto Ogawa, Ryunosuke Machida, Ryo Sadachi, Ryo Kitabayashi, Masayuki Yokoyama, Yuki Konda, Naomi Konishi, Kohei Uemura, Akihiro Hirakawa, Shogo Nomura, Riku Kajikawa, Shogo Ando, Yuya Ishikawa, Seiichiro Yamamoto, Suga Yamagami, Yuko Minami, Shintaro Iwamoto, Ryo Kanzaka
(Bioethics Division)
Tsunakuni Ikka, Haruka Nakada, Yoko Saito, Tomoko Seki
Introduction
In the annual report each year, the whole history of the Center for Research Administration and Support (CRAS) is reviewed since its establishment on July 16, 2014. The starting members of the CRAS were approximately 160 staff, who together provided diverse functions and specialties, ranging from research fund administration, alliances with the private sector, intellectual properties, clinical research coordination and data management, monitoring and audit, biostatistics support, secretariat services for research ethics (IRB), and COI committees.
The background and purpose of the creation of the CRAS was explained by Dr. Tomomitsu Hotta, the President of the National Cancer Center (NCC), in NCC News 2014 Vol. 5 No. 3 (in Japanese). Briefly, since its foundation in 1962, the NCC has added several new segments and organizations to evolve as a comprehensive cancer center. Because each segment needed its own research infrastructure, support activities in the NCC had become fragmented and scattered with the possibility of gaps and redundancies. Dr. Hotta launched extensive NCC-wide discussion with the help of the Strategic Planning Bureau and assembled the “NCC New Vision” in FY 2014, in which he proposed integration and communication for various research support functions in the NCC. The CRAS was created in response to the FY 2014 Vision.
In FY 2015, the NCC Hospital (NCCH) and the NCC Hospital East (NCCHE) were certified as Core Clinical Research Hospitals under the Medical Care Act in August and September, respectively. As a result, the governance of the Research Coordination Division, Research Promotion Division and Regulatory Science Section of the CRAS moved to the Clinical Research Support Offices, which belong to the common departments of each hospital. In January 2017, these divisions and section were officially separated from the CRAS in the NCC organization.
Another major reformation of the CRAS in FY 2017 was the establishment of the Bioethics Division by expanding the former Bioethics Section. The Clinical Trials Act was promulgated on April 14, 2017, and came into effect on April 1, 2018. Despite the general scarcity of human resources in the specialty field, the NCC has been endowed with strong staff, who have been contributing to work not just inside the NCC but also all over Japan with regard to research ethics-related issues including the implementation of the Clinical Trial Act.
In FY 2018, the concept of the RA (research administrator) system in the NCC was discussed in the CRAS and updated in the NCC headquarters.
In FY 2019, under the leadership of the new Chief of Bioethics Division, several important discussions and pieces of rule-making were done in collaboration with the researchers and staff in both campuses of the NCC. For instance, liaison and research manager staff have been assigned in each section of the NCC to facilitate communication between researchers and the research ethics review committee and build a solid base for the research ethics in the research groups.
In FY 2020, the CRAS continued to work in response to directions by the government on research governance, such as the establishment of a basic policy for the data sharing in collaboration with the JH (Japan Health Research Promotion Bureau), an organization officially established in FY 2020 to promote alliance among the 6 National Centers. The CRAS led the coordination of various sections on both campuses to get a more precise and comprehensive picture of the research-related funding and revenue of the NCC.
In FY 2021, the CRAS has made several renovations to support the researchers, such as the introduction of electronic grant application systems, to alleviate their administrative burden in the research activities. Another revision of the CRAS organization scheme was moving the Human Research Protection Section from the Bioethics Division to the Research Administration Division, thereby demarcating the research review function and research support services such as research ethics consultation.
In FY 2022, the CRAS has further evolved electronic grant application system to the integrated database of the public grants and the NCC Research & Development fund, so that the NCC can review and analyze its research portfolio and potentials. The total research funds awarded to the NCC researchers, both from public grants, contract and collaborative research and revenues from intellectual properties, have been increasing and reached over 20 billion yen. The remarkable performance of the NCC is of course attributable primarily to the high research capabilities and motivation of the NCC researchers, the role played by the Research Alliance and Intellectual Property Section has been also crucial, for its excellent strategic advice and support. Along with the avid collaboration with the industrial sectors, institutional COI policy has been drafted in FY 2022 by COI Management Section.
In FY 2023, following the approval of the "Comprehensive Innovation Strategy 2023" by the Cabinet on June 9, 2023, which emphasized the promotion of immediate open access to research outputs, such as publications and their key data, the center began considering the implementation of the "Institutional Repository". Additionally, based on the "Initiatives for Strengthening the Functions of National Research and Development Agencies" (as agreed upon by related ministries on March 29, 2024), discussions were initiated on specific measures to enhance their research capabilities and connect research results to practical applications in society. These measures include "efforts to secure and nurture excellent and diverse human resource," "the promotion of joint research and intellectual property management to facilitate social implementation," and "ensuring and thoroughly implementing research security and integrity." Furthermore, the center considered introducing the PI Salary System (enforced on February 5, 2024), which would allow the payment of personnel costs for principal investigators and others from direct competitive research funds. This system aims to improve researchers' performance and strengthen the research capabilities of institutions. Collaborative research with industrial sectors has been growing to surpass the figures in FY2022 both in the number of the projects, 743, and the amount of the funds received, 5 billion yen. Moreover, one of the NCC-certified ventures has gone public for the first time, initiating its IPO (Initial Public Offering).
(Future Prospects)
The upcoming FY2024 will be the last year of the first decade of the CRAS history. During that period, the biomedical research and technologies have kept evolving, showing a number of groundbreaking progress and transformation in diverse fields of science together with the major changes in the research environment and regulation. For instance, the avid pursuit of the Open Science based on the open-and-close strategy, research security and integrity is a solid aspect of the current global consensus. The NCC aims to maximize its research and social impact based on the deep understanding of the strength and opportunity of both campuses, which together cover basic, clinical, public health and social sciences as a comprehensive cancer center. As an integral part of the NCC research scheme, CRAS will also envision the next era. However, the core concept of the CRAS may stay unchanged to contribute to the NCC mission by promoting organic unity of the NCC as a whole and the CRAS aims to contribute to bringing the various sections together in the NCC.
1. Research Administration Division
1) Research Administration Section
The Research Administration Section is a central office in charge of various administrative work related to research funding including application and reporting. The major external funding sources of the NCC are competitive grants from the government and government-supported agencies, such as the Ministry of Health, Labour and Welfare (MHLW), the Japan Society for the Promotion of Science (JSPS), and the Japan Agency for Medical Research and Development (AMED).
This section also serves as an administrative office for the NCC Research and Development Fund, which is provided directly from the government to the NCC for fulfillment of its mission as the national core institute of cancer control. This section organized seminars regarding research funding and its rules to prevent financial misconduct or research misconduct.
The Guidelines for Management and Audit of Public Research Funds at Research Institutions were updated by the Ministry of Education, Culture, Sports, Science and Technology (MEXT) in February 2021 and adopted by the MHLW in March 2021. This section serves as a compliance promotion office of the Guidelines and has established a new system for research fund administration, which is fully compatible with the new Guidelines.
Based on revision by MEXT of the guidelines for managing and auditing public research funds at research institutions, we formulated an action plan to prevent the improper use of research funds. As part of our enlightenment activities, we distributed newsletters and disseminated items related to the prevention of improper use of research funds. (The items were reported by the compliance promotion officers of each department to each department’s members, in order to raise their awareness related to the prevention of financial misconduct.)
(Future Prospects)
The Section will keep playing a central role in establishing, maintaining and improving the research management system in NCC that complies with the aforementioned government Guidelines for the Management and Audit of Public Research Funds at Research Institutions (MEXT and MHLW). Additionally, the Section's new focus will include the promotion of the "Open Science", which is a global initiative and has been adopted by the "Comprehensive Innovation Strategy 2023" in Japan (June 9, 2023), and “Research Security and Integrity” and the “Institutional Repository.” based on the "Initiatives for Strengthening the Functions of National Research and Development Agencies" (March 29, 2024).
Specifically, Institutional Repository and its national network aim to advance the integration of diverse and scattered information such as research funds, research achievements, research outcomes, and personnel all over Japan. The system will serve as a crucial stepping stone to create an environment capable of collecting and analyzing information, thereby enhancing the information infrastructure and analytical capabilities that contribute to the strategic analysis and planning in a wide variety of research and development fields, including cancer research, and their integrated areas.
2) Research Alliance and Intellectual Property Section
We efficiently build bridges between NCC research and industries by collective management of the collaborative research alliance and the utilization of intellectual properties (IPs) arising therefrom.
1. Alliance with the private sector
We have supported NCC's variety of alliances, including but not limited to establishing worldwide comprehensive collaborative partnerships with major pharmaceutical companies and academic institutions. Currently, 11 comprehensive collaborative alliances are active between the NCC and domestic/overseas industry partners (Figure 1). We have been also supporting a nationwide genomic screening project (SCRUM-Japan “Cancer Genome Screening Project for Individualized Medicine in Japan”) led by the NCC as first challenge in Japan. Nationwide medical institutions and 22 domestic/overseas pharmaceutical companies have joined the Project (Table 1). The collaborative research and research funds that we manage have been increasing year by year. In 2023, the number of collaboration cases reached 743, and the amount of research funds reached 45.01 billion Japanese yen (Figure 2). This is 70 times the size of the figure in 2010, just after the NCC was incorporated.
Figure 1. Comprehensive Research Alliance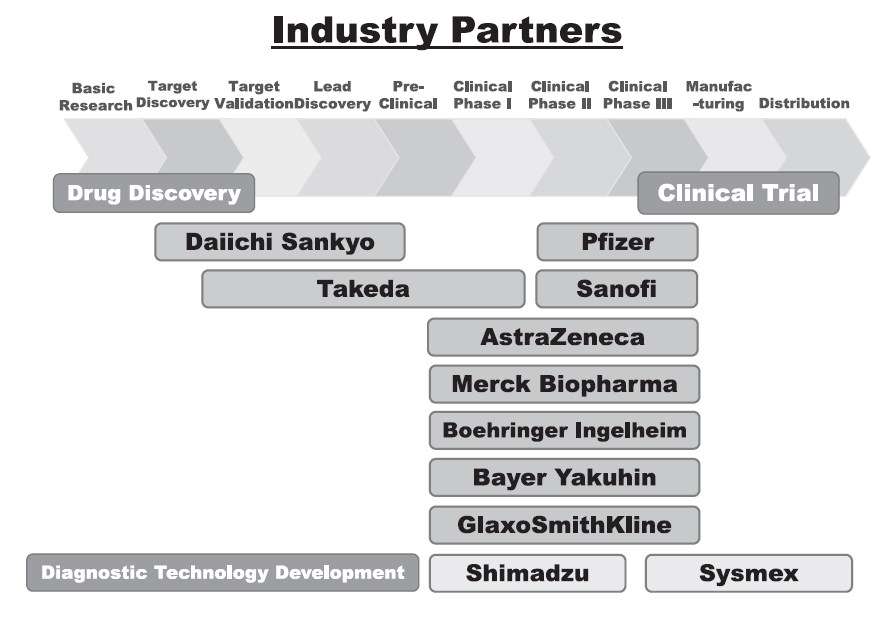

Table 1. Participant Companies to SCRUM-Japan

Figure 2. Collaborative Research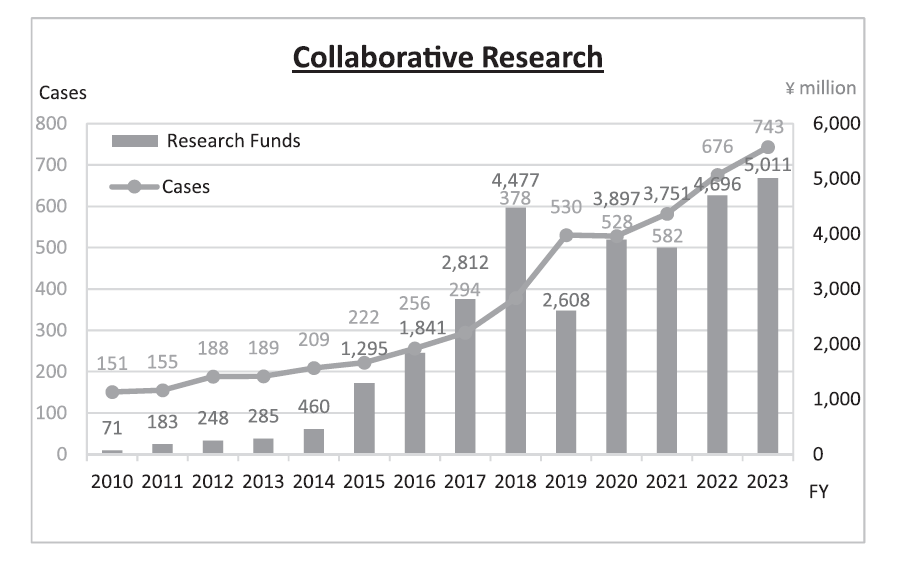

2. Intellectual property management
We encourage, needless to mention the collaborative research results with companies, NCC researchers to license-out IPs derived from their research at early stages to partner companies who can work together towards realization of their research results. Our IP strategy enables us to reduce costs in patent prosecution and to expedite commercialization of our research results, eventually availing the receipt of monetary consideration. On the other hand, for those IPs NCC substantially bears the cost, we constantly review the value of IPs and dismiss those which could not find sponsors within a certain period. By allocating NCC’s financial resources to IPs with commercial values, NCC has been successful in sustaining positive outcome of IP management for years (Figure 3).
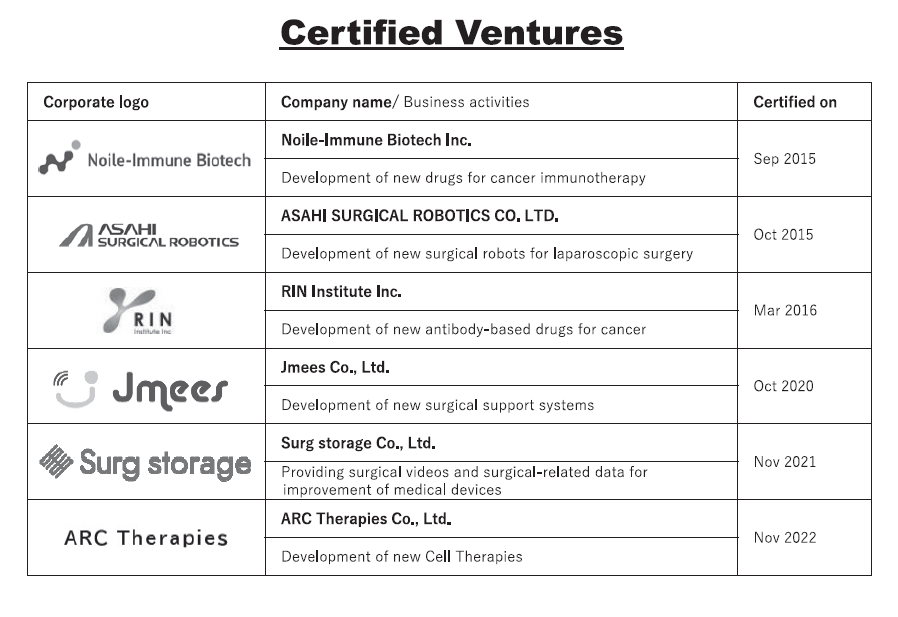
3. Certified ventures
We certify “NCC-launched venture companies” that try to perform application and commercialization of IPs and research results originating in the NCC. As of 2023, we have certified six NCC-launched venture companies (Table 2). In 2023, the first listed company was born from these certified venture companies.
Figure 3. Intellectual Property Income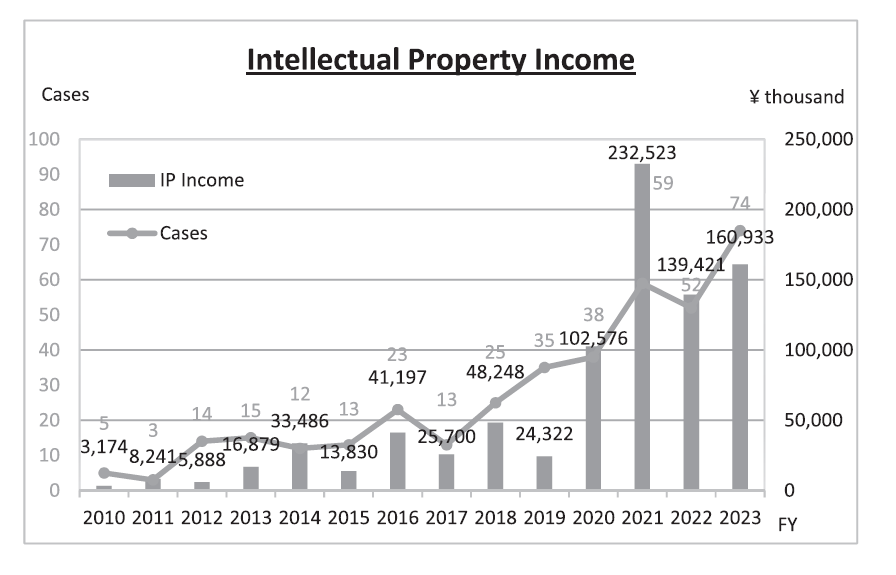

Human resource development and education
We host educational seminars of essential IP knowledge to NCC faculties at least twice a year. Our staff are encouraged to participate in seminars and conferences to update their knowledge on IP laws, regulations, and guidelines as well as scientific technologies, all of which are necessary to promote academic-industrial alliances. From time to time, we consult with experts, such as lawyers and patent attorneys, to solve problems and to gain practical experiences.
(Future Prospects)
Under the NCC’s vision of “commitment for providing the best possible cancer treatment and care through relentless partnership with the community”, we will keep supporting the creation of effective and innovative collaborative research frameworks which develop to fruitful research partnership. We will continue to play key roles in assisting the NCC making decisions on its IP management.
3) Research administrators
Research administrators (RA) have supported to translate research outcomes into practical application based upon a comprehensive alliance with pharmaceutical companies. To promote cooperation with industries and academia, RA has introduced bioresources including PDX library, advanced technologies and recent research findings in the NCC through Tsukiji TR Board activity and BioJapan 2023 exhibition. RA also supported the progress of AMED research projects through the Practical Research for Innovative Cancer Control Management Office (PRIMO), and collaborative research projects of 6 National Centers (NCs) conducted by Japan Health Research Promotion Bureau (JH).
(Future Prospects)
RA promotes the translation of innovative research in the NCC to clinical diagnostic and therapeutic development and to patient care through comprehensive alliances with pharmaceutical companies, Tsukiji TR Board activity, AMED support projects by PRIMO, and cooperation between 6 NCs and JH.
4) Human Research Protection Section
The Human Research Protection Section oversees the administrative operations of various human subject research review committees and handles administrative tasks related to the responsibilities of the head of the research institution regarding ethics review.
Furthermore, the Institutional Review Board/ Independent Committee (IRB/IEC), in accordance with the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices, performs committee secretariat functions in collaboration with the Clinical Trial Secretariat of the Hospital and the Hospital East.
Accomplishments of the reviews by the IRBs at the NCC in FY2023 included 2,043 active research projects and 337 new research plans by the NCC faculty members (include 113 central reviews)
(Future Prospects)
The HRPS will continue working, in cooperation with the NCCH’s and the NCCHE’s Ethical Review Support Sections, to ameliorate the NCC’s whole ethics review processes for more appropriate and efficient ways to be achieved.
The medium- to long-term outlook for our office is to further enhance the expertise and speed of the Ethics Review Committee secretariat functions and provide more advanced ethics review functions. In the current situation where research is diversifying and multiple laws and regulations are amended almost every year, it is important for center researchers to carry out maintenance of the center's internal system without delay and ensure appropriate and prompt review functions. Of course, providing this information to a wide range of external researchers is in response to the needs of the ethical review committees of national cancer centers. In the future, we would like to contribute to supporting other organizations as a national center, such as by hosting training for staff from other organizations' ethical review committee secretariats.
2. Biostatistics Division
Biostatistics Division has a role of responsibility in formulating research hypothesis, study design, analysis, interpretation and publication, especially in JCOG and EPOC clinical trials and the investigator-initiated clinical trials which are led by investigators in the NCC Hospitals. We have also committed to establishing an infrastructure to support the clinical trials in the NCC Hospitals. Furthermore, we have been actively involved in collaborative relationships with the NCC Research Institute and the Institute of Cancer Control.
Furthermore, we have provided biostatistical consultation and expertise, which supports NCC investigators working on basic, translational, clinical and epidemiological researches. We offered advice to about 133 problems for which biostatistical consultation was requested from April 2023 to March 2024.
A survey of biostatistical consultation provided in the NCC were presented as a poster at the 15th Annual Meeting of the Japan Society of Clinical Trials and Research. In statistical research, one research paper, with one of our members as the first author, was accepted in the Journal of Biopharmaceutical Statistics, and two poster presentations were given at the International Biometric Society and the Japanese Joint Statistical Meeting. In addition, one clinical trial related research paper as the first author was accepted in the Cancer Medicine, and poster presentations were given at the European Society for Medical Oncology and Japan Society of Clinical Trials and Research
Education
We provided 13 introductory Biostatistics lectures for investigators in NCC to learn and review the elementary aspects of Biostatistics. We had a cumulative total of 545 participants. In addition, we hosted 3 Biostatistics lectures to cover advanced and important biostatistical side of various application fields and a total of 160 investigators participated. The lectures are open to any applicants from outside institutes.
In addition, we provided an internship program for 17 graduate students from the graduate schools of the University of Tokyo (the Biostatistics and bioinformatics course, Graduate School of Interdisciplinary Information Studies).
In response to the activity of career development and education for biostatisticians by the Japan Health Research Promotion Bureau (JH), the newly established "Young Biostatisticians NC Collaboration Training Pilot Project" was started from fiscal year 2022. Through this initiative, we have admitted two junior biostatisticians, one from the National Center of Neurology and Psychiatry and one from the National Center for Child Health and Development, into on-the-job training (OJT) program on a weekly basis. Notably, both junior biostatisticians obtained the accreditation of a Trial Statistician through the Biometric Society of Japan in fiscal year 2023, based on achievements that encompassed OJT experiences.
In terms of educational outreach to clinical research practitioners nationwide, we have continued to develop and operate the e-learning platform ICRweb (Introduction to Clinical Research: https://www.icrweb.jp), releasing a total of 60 new contents. The user base has grown from 177,014 to 199,022, and we expanded to include 29 new facilities, bringing the total number of contracted institutions to 156. This expansion ensured stable operations independent of research funding. Graduation certificates were issued for 30,933 completions, representing the widespread provision of clinical research education to a diverse range of individuals engaged in clinical research.
Furthermore, as part of the Asian Clinical Trials Network initiative (ATLAS Project) by the National Cancer Center Hospital, an English version of ICRweb was introduced, offering learning opportunities in clinical research in English to not only domestic but also international practitioners. Continuing from the previous year, we further enriched the 6NC Common Educational Platform with lectures not only on clinical research but also animal experimentation and data science, contributing to comprehensive education.
Future Prospects
The NCC has a critical role for providing clinical service, education, conducting research and making policy recommendation/proposal, which are all required to make a decision on the basis of solid and scientific evidence from reliable data and information. In addition, new challenges have emerged, such as the use of Real World Data for new drug application and clinical evaluation for implementation of medical device with artificial intelligence. To adapt flexibly to these developments, the Biostatistics Division is expanding its independent and collaborative research across a range of areas. Recently, we have made a particular effort to build effective relationships with the investigators at the NCC Research Institute and the Institute of Cancer Control. Collaboration on such a wide range of areas have motivated to develop statistical methods to get deeper insight into evidence and public health impact. We are also working on promoting cooperative framework with outside experts in statistics/biostatistics. We are opening up a new methodological research area in which a mathematical approach will serve as a solid basis.
3. Bioethics Division
The Bioethics Division provides education and training on issues related to research ethics and conflicts of interest (COI), mainly in relation to the protection of human subjects, including ethical guidelines for life sciences and medical research involving human subjects, the Pharmaceutical Machine Law, the Clinical Research Law, laws related to regenerative medicine and the Personal Information Protection Law, and the operation of various ethical review committees for human subject research. The following are some of the activities.
- The National Cancer Centre's "Training for new employees" (delivered online in April).
Training on research ethics at the National Cancer Centre's 'Clinical Research Seminar' (15 May, 26 June, 12 July, 1 August, 19 December and 5 March in FY2023).
- Training for Ethical Review Committee and Clinical Trial Review Committee members in 2023 (26 August 2023).
In addition to the provision of these education and training programmes, the Bioethics and Medical Law Section and the COI Management Section, each of which is located in this division, carry out the following activities.
1) Bioethics and Healthcare Law Section
The Office of Bioethics and Medical Law provides research ethics consultation services to researchers, research assistants and research ethics review committee members at various stages from the planning of research to its publication. Unlike ethics review, research ethics consultation is non-binding advice and deals with ethical issues beyond regulatory compliance.
FY2023 results: 123 consultations were received annually. 36 were from researchers and others at the Tsukiji Campus and 87 were from researchers and others at the Kashiwa Campus. 7 cases were related to the Clinical Research Act, 113 cases to ethical guidelines and 27 other cases.
(Future Prospects)
We will continue to provide and enhance our research ethics consultation services. In FY2023, the Ethical Guidelines for Life Sciences and Medical Research Involving Human Subjects were revised, and we will strive to support the smooth implementation of research based on the new guidelines from an ethical perspective. Other research support activities related to the Centre as a whole, in addition to consultations with individual researchers, will be continued as necessary.
We have a mission to support the resolution of ELSI issues in research implementation through consultation services and education and training activities for the entire center and all researchers, and to further develop the systems necessary to achieve the overall mission of the National Cancer Centre while responding to changes in various rules and social conditions.
2) COI management section
Our section serves as the COI management committee secretariat to manage NCC researchers’ COI and to respond to inquiries. The subjects of COI management are physician-initiated clinical trials, some of the clinical research conducted under the Ethical Guidelines for Life Sciences and Medical Research Involving Human Subjects. We recommend that the researchers who are involved in the research project funded by national ministries or authorities disclose their COI despite being subject to COI review. Furthermore, we manage the COI of the members of the research ethics committee and institutional review board. We also handle the COI management required by the Clinical Trials Act.
FY2023 results: The COI management committee reviewed 123 physician-initiated clinical trials (2,488 researchers) and 192 clinical research conducted under the Ethical Guidelines for Life Sciences and Medical Research Involving Human Subjects (2,366 researchers). Furthermore, we verified 167 COI disclosure forms following the Clinical Trials Act. Furthermore, we completed the COI Declaration Management System modification to verify the researchers’ COI disclosure required by the Clinical Trials Act.
(Future Prospects)
We continue to improve the structure of COI review and management. From the second half of 2023, the secretariat members have been renewed and will need to operate more efficiently. In addition, the relevant regulations, which have remained unrevised for many years, will be revised in due course.
Our mission is to address conflicts of interest in the conduct of research through conflict of interest management and education and training activities for researchers throughout the Centre, and we aim to further develop the systems necessary to achieve the overall mission of the National Cancer Centre while responding to changes in various rules and social conditions.
