Annual Report 2023
Team Makinoshima
Hideki Makinoshima, Sho Tabata
Introduction
It is thought that the breakdown of homeostasis, which maintains a constant state of the body, causes carcinogenesis and cancer progression. Beyond these homeostatic mechanisms, metabolites that induce carcinogenesis and metabolic mechanisms specific to cancer cells are overproduced or decreased within an individual, and the metabolites themselves contribute to the development and malignancy of cancer. We have formulated a hypothesis and are continuing our research. Through these studies, we aim to elucidate the true nature of cancer from the perspective of metabolism.
The Team and What We Do
In FY2023, the aim was to identify metabolites and metabolic pathways unique to cancer, and to discover new drugs that target them. In collaborative research between Keio University's Institute for Advanced Biosciences and the National Cancer Center, samples were collected from patient-derived samples, cancer cell lines, cancer stromal cells, and mouse models, and cancer-related metabolome analysis was conducted. Through joint research with the National Cancer Center Research Institute, the National Cancer Center Hospital East, and Yamagata University Faculty of Medicine, we constructed metabolite profiles using metabolome analysis of clinical samples. Furthermore, in joint research with companies and commissioned research, we conducted research based on metabolome analysis with Ajinomoto Co., Inc., Otsuka Pharmaceutical Co., Ltd., JA Kushibiki Agricultural and Industrial Federation, and Midori Service. In drug discovery research targeting metabolic pathways characteristic of cancer, we investigated the control mechanisms of nucleic acid biosynthetic pathways. Furthermore, we proposed the possibility of a new treatment method that inhibits the novel biosynthesis pathway of purine nucleic acids with an antifolate and the recycling pathway with a new drug. In order to elucidate the control mechanism of nucleic acid metabolism in small cell lung cancer, we clarified the importance of the purine base recycling pathway of nucleic acids and published a paper.
Research Activities
We are studying the control mechanisms of novel biosynthetic and recycling pathways for nucleic acid metabolism, including purine and pyrimidine nucleotides, which are the raw materials for DNA and RNA synthesis. We are conducting research on nucleic acid metabolism in cancer, focusing on allosteric control and feedback mechanisms, which are important in metabolic research. Two metabolic pathways are known for the biosynthesis of purine nucleotides: a de novo synthesis pathway and a salvage pathway (Figure 1).
In the history of anticancer drug development, novel biosynthetic pathways related to folate metabolism have been used as drug targets. Using small cell lung cancer cell lines, we revealed that cancer cells preferentially utilize recycling pathways. Furthermore, antifolates suppress purine base biosynthesis by inhibiting a new synthesis pathway, but the existence of a recycling pathway has revealed a drug resistance mechanism that reduces drug efficacy during treatment. (Figure 1).
Figure 1. Purine nucleotide metabolism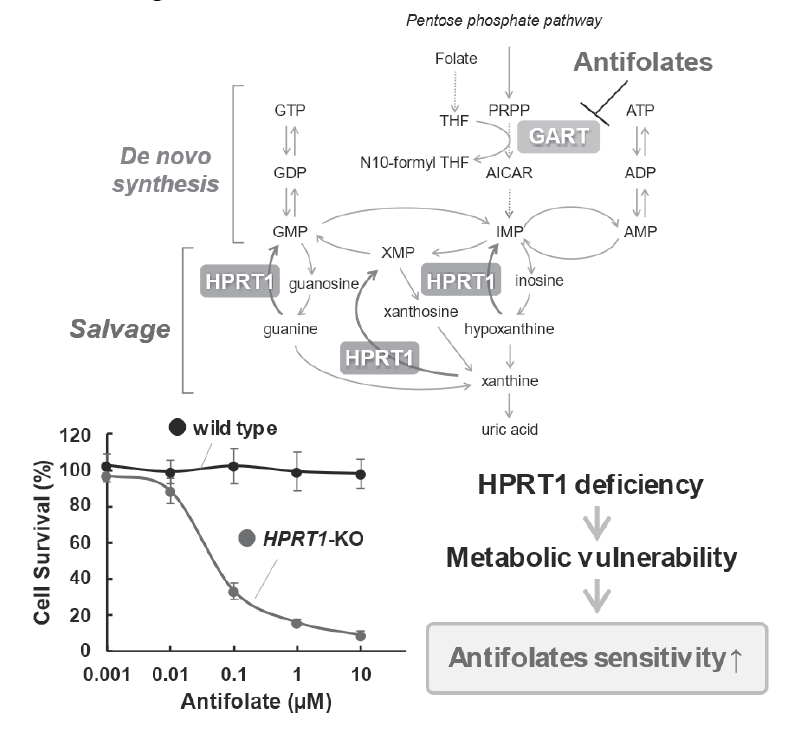

1. Tabata S, Umemura S, Narita M, Udagawa H, Ishikawa T, Tsuboi M, Goto K, Ishii G, Tsuchihara K, Ochiai A, Kobayashi SS, Soga T, Makinoshima H. Metabolic Hallmarks for Purine Nucleotide Biosynthesis in Small Cell Lung Carcinoma. Molecular cancer research, 22:82-93, 2024
2. Hiranuma K, Asami Y, Kato MK, Murakami N, Shimada Y, Matsuda M, Yazaki S, Fujii E, Sudo K, Kuno I, Komatsu M, Hamamoto R, Makinoshima H, Matsumoto K, Ishikawa M, Kohno T, Terao Y, Itakura A, Yoshida H, Shiraishi K, Kato T. Rare FGFR fusion genes in cervical cancer and transcriptome-based subgrouping of patients with a poor prognosis. Cancer medicine, 12:17835-17848, 2023
3. Plasma and Urinary Metabolomic Analysis of Gout and Asymptomatic Hyperuricemia and Profiling of Potential Biomarkers: A Pilot Study. Ohashi Y, Ooyama H, Makinoshima H, Takada T, Matsuo H, Ichida K.Biomedicines. 2024 Jan 27;12(2):300. doi: 10.3390/biomedicines12020300. PMID: 38397902 Free PMC article.
4. In vivo Drug Screening to Identify Anti-metastatic Drugs in Twist1a-ERT2 Transgenic Zebrafish. Nakayama J, Makinoshima H, Gong Z.Bio Protoc. 2023 May 20;13(10):e4673. doi: 10.21769/BioProtoc. 4673. eCollection 2023 May 20.PMID: 37251091 Free PMC article.
Education
Miyu Narita (Research Assistant, Shonai Regional Industry Promotion Center)
Aina Nagamoto (Research Assistant, Shonai Regional Industry Promotion Center)
Shin Goto (Special research student, Keio University Institute for Advanced Biosciences)
Future Prospects
In the next year, we aim to identify control mechanisms and new nucleic acid metabolic pathways unique to cancer, and to develop new treatments that target them. Furthermore, we will promote joint research with companies and obtain new research funds.
