Annual Report 2024
Section of Data Science Strategy
Takashi Yugawa, Hirokazu Fukuda, Katsuya Tsuchihara, Genta Ohno, Akiko Nagai, Yuuta Maruki, Tsuyuko Kawahara
The Team and What We Do
Regarding comprehensive genomic profiling (CGP) tests that are performed for cancer patients under national health insurance in Japan, we promote the primary and secondary utilization of those genomic and clinical data collected at the Center for Cancer Genomics and Advanced Therapeutics (C-CAT) through various data-sharing systems (Figure 1). We perform the following tasks to contribute to the support and development of cancer genomic medicine (CGM):
Figure 1. Medical and Research Use of C-CAT “Real World Data”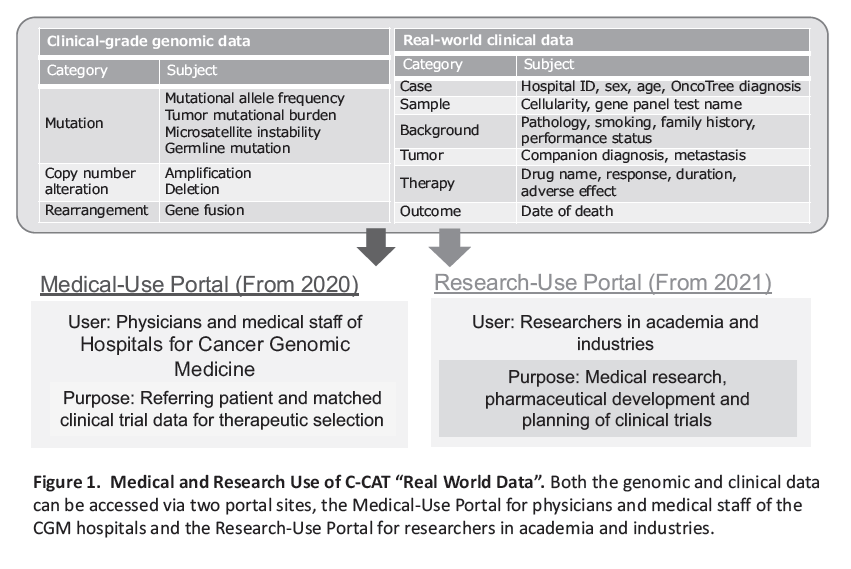

1) Modification and operation of the “C-CAT Medical-Use Portal” to support medical treatment at the CGM hospitals.
2) Modification and operation of the “C-CAT Research-Use Portal” to promote academic research and development of pharmaceuticals, etc.
3) Modification and operation of an additional cloud-based system, “C-CAT CALICO (CALculation & Investigation ClOud),” which enables users to analyze raw sequence data to examine novel genetic alterations that are not included in the reports provided by the registered testing companies.
4) Secretariat management of the C-CAT Data Utilization Review Board to examine the suitability of applications for secondary utilization.
5) Administrative procedures related to the conclusion of contracts for new projects and to change or extension requests for ongoing projects, as well as project management in response to performance reporting requirements.
6) Public relations activities about CGM and CGP tests for patients and the general public.
Research Activities
- The system was modified to improve the search capability and operability of the “C-CAT Medical-Use Portal” to ensure its stable operation.
- Regarding the “C-CAT Research-Use Portal,” we focused on improving the accuracy of aggregated data and enhancing portal functionality, while prioritizing usability in operation.
- In FY2024, the C-CAT Data Utilization Review Board were held five times, and 31 new applications from academic and industrial institutions in Japan (including Japanese branches of foreign pharmaceutical companies) were approved to utilize the C-CAT data. Additionally, five applications for research plan change were consulted and approved for continuous data utilization by the C-CAT Data Utilization Review Board.
- Regarding the “C-CAT CALICO,” operations commenced for existing users of the “C-CAT Research-Use Portal” within academia, with usage beginning for 10 projects. Furthermore, to address user needs, an upgraded version of the cloud environment was constructed and underwent trial operation. An information session for industry was held to expand the scope of eligible applicants for usage requests starting next fiscal year.
- To facilitate the overseas provision of the C-CAT data, revised versions of the ICF model documents were distributed to all Hospitals for Cancer Genomic Medicine, and each hospital has started the change procedures. Additionally, we have created English versions of the Licensing Agreement for the Use of C-CAT Data and the C-CAT Data Secondary Use Policy.
- We updated the content of the patient and public-facing website “CGM and CGP tests” while continuing outreach activities via X (formerly Twitter). These efforts widely disseminated information about cancer genomic medicine, C-CAT's initiatives, the achievement of 100,000 registered cases (Figure 2), and outcomes from utilizing the C-CAT data such as academic papers as well.
Figure 2. Data accumulation in C-CAT has exceeded 100,000 cases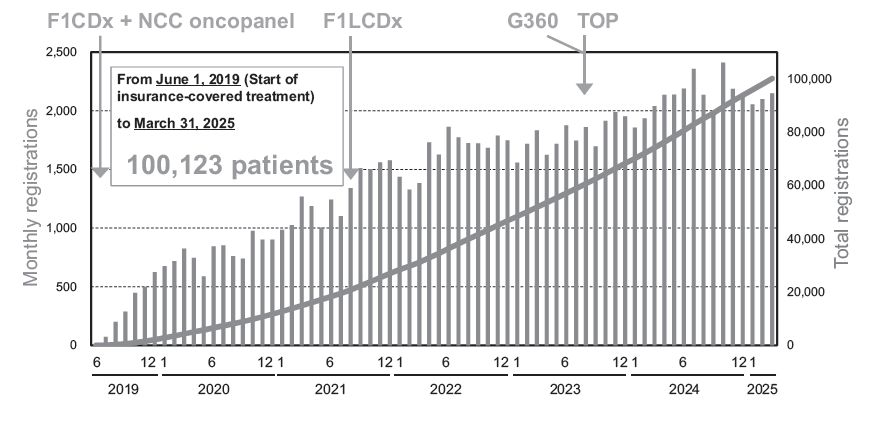

Education
Through hosting visits from academic and medical institutions both domestically and internationally, we have worked to cultivate talent in this field.
Future Prospects
We aim to further increase the number of users through data expansion and system updates to ensure the usefulness of the search portal and cloud system, leading to the creation of new medicine. To facilitate the overseas provision of the C-CAT data, we will conduct final preparations for both the system and operations through ongoing discussions with the Ministry of Health, Labour and Welfare. We will enhance content on our website and X regarding C-CAT initiatives to promote broader societal awareness.
