Annual Report 2024
Department of Head and Neck Medical Oncology
Makoto Tahara, Susumu Okano, Tomohiro Enokida, Takao Fujisawa, Nobukazu Tanaka, Yuta Hoshi, Takuma Kishida, Ryo Kuboki
Introduction
The Department of Head and Neck Medical Oncology is dedicated to the clinical management of patients with head and neck cancer (HNC) and to research on anticancer drugs for HNC.
Our mission is to:
1. deliver the best evidence-based treatments;
2. promote the importance of supportive care in HNC management;
3. accelerate the approval of new drugs and reduce drug lag through active participation in global clinical trials;
4. develop cutting-edge therapeutic strategies; and
5. train the next generation of experts in head and neck medical oncology.
The Team and What We Do
Our department is responsible for drug therapy in head and neck cancer. Clinical practice is carried out by four staff members, one physician-scientist, two residents, and one short-term resident.
Drug therapy for head and neck cancer is not limited to recurrent or metastatic disease; it is also an established standard treatment, in combination with radiotherapy or surgery, for cases requiring functional preservation or those deemed unresectable. Treatment strategies are discussed at multidisciplinary head and neck conferences to provide the most appropriate care for each patient (Table 1).
Table 1. Number of patients by site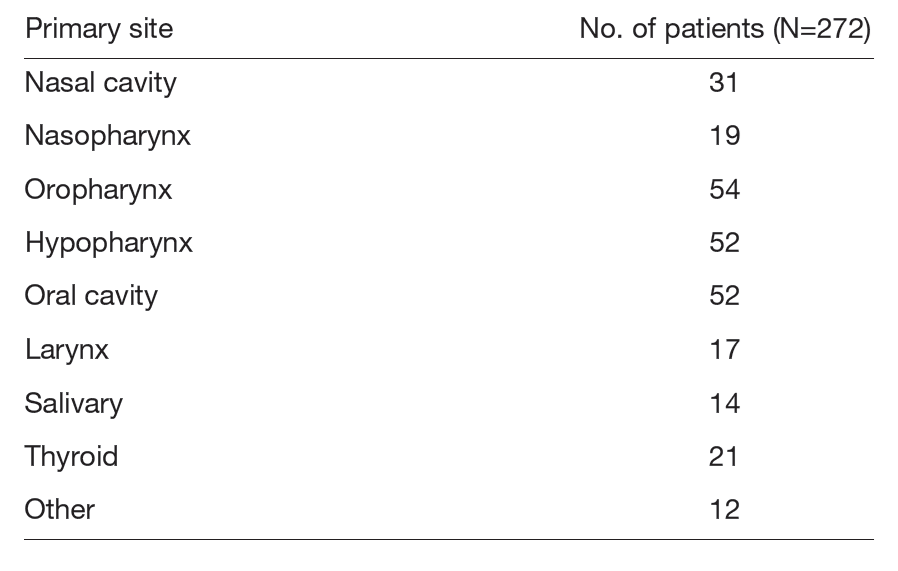

Our department is also highly engaged in drug development, participating in international collaborative clinical trials as well as investigator-initiated trials for rare cancers. On the clinical side, we actively promote translational research, as outlined in Table 2.
Table 2. Number of patients by procedure

Research Activities
1 COLLECT Study (Differentiated Thyroid Cancer, DTC)
In a prospective observational study of lenvatinib for radioactive iodine-refractory DTC, patients in the planned drug holiday group (n=73) showed significantly better OS, TTF, TTS, and PFS compared with those without scheduled breaks (n=180). This provided prospective evidence supporting the benefit of planned drug holidays.
2 Retrospective Study on Lenvatinib Dose Re-escalation for DTC
Because lenvatinib blood concentrations vary widely among individuals, dose reductions may prevent patients from reaching effective levels. In cases of disease progression, we examined the efficacy of dose re-escalation. The median post-escalation treatment duration was 9.9 months, median OS after escalation was 20.4 months, and median OS from treatment initiation was 6.2 years—demonstrating favorable outcomes. This suggests that re-escalation at progression may be a viable treatment option.
3 Induction Chemotherapy (IC-PCE) for Unresectable Locoregional Recurrent HNSCC
We retrospectively compared 42 patients with unresectable locoregional recurrent HNSCC after surgery, who had not undergone prior radiation and were eligible for cisplatin. The IC-PCE group (n=27) achieved an ORR of 55.6% and a CRT completion rate of 88.9%. Compared with the CRT-only group (n=15), ORR and 2-year PFS were superior, with a significant benefit in oral cancer (37.5% vs. 0%, p=0.015). These findings suggest the efficacy of IC-PCE, particularly in oral cancer.
4 ICI Monotherapy for Recurrent/Metastatic Olfactory Neuroblastoma (R/M ONB)
We analyzed 11 patients with R/M ONB. The ICI group (n=6) showed significantly prolonged OS compared with the non-ICI group (n=5) (median not reached vs. 6.4 months, p=0.035). These results indicate the potential efficacy of ICI monotherapy in R/M ONB.
Clinical Trials
The patient enrollment for the following investigator-initiated clinical trials has been completed:
1 A phase 2 study of nivolumab plus lenvatinib for unresectable anaplastic thyroid cancer, and
2 A phase 2 study of darolutamide for androgen receptor–positive recurrent or metastatic salivary gland carcinoma.
To facilitate the timely approval of new drugs and reduce drug lag, we have also participated in global phase trials, including those involving immune checkpoint inhibitors.
Education
We provide education not only to medical staff within our institute but also to those outside, through programs such as the Seminar of the Japan Society of Supportive Care for Patients with Head and Neck Cancer. In addition, our department continuously accepts trainees.
Future Prospects
We anticipate that ongoing and planned clinical trials will help transform the standard of care for head and neck cancer. Our biomarker analyses, the result of collaborative efforts, are expected to contribute to the development of new treatment strategies. Furthermore, the success of our educational programs—aimed at increasing the number of medical oncologists engaged in head and neck cancer treatment—will ultimately lead to improved patient outcomes and quality of survival.
List of papers published in 2024
Journal
1. Tanaka N, Enokida T, Okano S, Fujisawa T, Tanaka H, Onaga R, Hoshi Y, Kishida T, Wada A, Sato M, Takeshita N, Fujisawa T, Motegi A, Zenda S, Akimoto T, Tahara M. Induction chemotherapy and concurrent chemoradiotherapy with cisplatin for T4 nasoethmoidal squamous cell carcinoma: The value of paclitaxel, carboplatin and cetuximab (PCE). Oral oncology, 163:107235, 2025
2. Fujisawa T, Motegi A, Hirata H, Zenda S, Hojo H, Nakamura M, Oyoshi H, Tomizawa K, Zhou Y, Fukushi K, Kageyama SI, Enokida T, Okano S, Tahara M, Shinozaki T, Hayashi R, Matsuura K, Akimoto T. Long-term outcomes of patients with oral cavity cancer receiving postoperative radiotherapy after salvage neck dissection for cervical lymph node recurrence. Head & neck, 46:541-551, 2024
3. Hirai T, Naito Y, Koyama S, Nakanishi Y, Masuhiro K, Izumi M, Kuge T, Naito M, Mizuno Y, Yamaguchi Y, Kang S, Yaga M, Futami Y, Nojima S, Nishide M, Morita T, Kato Y, Tsuda T, Takemoto N, Kinugasa-Katayama Y, Aoshi T, Villa JK, Yamashita K, Enokida T, Hoshi Y, Matsuura K, Tahara M, Takamatsu H, Takeda Y, Inohara H, Kumanogoh A. Sema6D forward signaling impairs T cell activation and proliferation in head and neck cancer. JCI insight, 9:e166349, 2024
4. Kiyota N, Tahara M, Homma A. Current status and future perspective of postoperative treatment for locally advanced squamous cell carcinoma of the head and neck. Japanese journal of clinical oncology, 54:613-619, 2024
5. Machiels JP, Tao Y, Licitra L, Burtness B, Tahara M, Rischin D, Alves G, Lima IPF, Hughes BGM, Pointreau Y, Aksoy S, Laban S, Greil R, Burian M, Hetnał M, Delord JP, Mesía R, Taberna M, Waldron JN, Simon C, Grégoire V, Harrington KJ, Swaby RF, Zhang Y, Gumuscu B, Bidadi B, Siu LL. Pembrolizumab plus concurrent chemoradiotherapy versus placebo plus concurrent chemoradiotherapy in patients with locally advanced squamous cell carcinoma of the head and neck (KEYNOTE-412): a randomised, double-blind, phase 3 trial. The Lancet. Oncology, 25:572-587, 2024
6. Oaknin A, Moore K, Meyer T, López-Picazo González J, Devriese LA, Amin A, Lao CD, Boni V, Sharfman WH, Park JC, Tahara M, Topalian SL, Magallanes M, Molina Alavez A, Khan TA, Copigneaux C, Lee M, Garnett-Benson C, Wang X, Naumann RW. Nivolumab with or without ipilimumab in patients with recurrent or metastatic cervical cancer (CheckMate 358): a phase 1-2, open-label, multicohort trial. The Lancet. Oncology, 25:588-602, 2024
7. Tahara M, Takami H, Ito Y, Okamoto T, Sugitani I, Sugino K, Takahashi S, Takeyama H, Tsutsui H, Hara H, Mitsuma A, Yamashita H, Ohashi Y, Imai T. A Prospective Cohort Study Exploring the Effect of Lenvatinib Planned Drug Holidays in Treatment of Differentiated Thyroid Cancer. Thyroid, 34:566-574, 2024
8. Okano S. Immunotherapy for head and neck cancer: Fundamentals and therapeutic development. Auris, nasus, larynx, 51:684-695, 2024
9. Imamura Y, Kiyota N, Tahara M, Kodaira T, Hayashi R, Nishino H, Asada Y, Mitani H, Iwae S, Nishio N, Onozawa Y, Hanai N, Ohkoshi A, Hara H, Monden N, Nagaoka M, Minami S, Kitabayashi R, Sasaki K, Homma A. Risk prediction model for cisplatin-induced acute kidney injury in patients with head and neck cancer receiving chemoradiotherapy: A re-analysis of a phase II/III JCOG1008 trial. Oral oncology, 154:106868, 2024
10. Hoshi Y, Enokida T, Tamura S, Nakashima T, Okano S, Fujisawa T, Sato M, Wada A, Tanaka H, Takeshita N, Tanaka N, Onaga R, Kishida T, Uryu H, Sakashita S, Asakage T, Tahara M. Efficacy of anti-PD-1 monotherapy for recurrent or metastatic olfactory neuroblastoma. Frontiers in oncology, 14:1379013, 2024
11. Katada C, Yokoyama T, Watanabe A, Hara H, Yoshii T, Fujii H, Yamaguchi H, Nakajima TE, Izawa N, Ando T, Nomura M, Kojima T, Yamashita K, Kawakami S, Ishiyama H, Inoue Y, Sakamoto Y, Sasaki H, Ishikawa H, Hosokawa A, Hamamoto Y, Muto M, Tahara M, Koizumi W. Optimizing Organ-Preservation Strategies Through Chemotherapy-Based Selection in Esophageal Squamous Cell Carcinoma: Results From the CROC Multi-Institutional Phase 2 Clinical Trial. International journal of radiation oncology, biology, physics, 120:1353-1362, 2024
12. Sato M, Enokida T, Fujisawa T, Okano S, Takeshita N, Tanaka N, Tanaka H, Motegi A, Zenda S, Shinozaki T, Matsuura K, Hayashi R, Akimoto T, Tahara M. Induction chemotherapy with paclitaxel, carboplatin, and cetuximab (PCE) followed by chemoradiotherapy for unresectable locoregional recurrence after curative surgery in patients with squamous cell carcinoma of the head and neck. Frontiers in oncology, 14:1420860, 2024
13. Hashimoto T, Nakamura Y, Fujisawa T, Imai M, Shibuki T, Iida N, Ozaki H, Nonomura N, Morizane C, Iwata H, Okano S, Yamagami W, Yamazaki N, Kadowaki S, Taniguchi H, Ueno M, Boku S, Oki E, Komatsu Y, Yuki S, Makiyama A, Otsuka T, Hara H, Okano N, Nishina T, Sakamoto Y, Miki I, Kobayashi S, Yuda J, Kageyama SI, Nagamine M, Sakashita S, Sakamoto N, Yamashita R, Koga Y, Bando H, Ishii G, Kuwata T, Park WY, Ohtsu A, Yoshino T. The SCRUM-MONSTAR Cancer-Omics Ecosystem: Striving for a Quantum Leap in Precision Medicine. Cancer discovery, 14:2243-2261, 2024
14. Onaga R, Enokida T, Okano S, Fujisawa T, Tanaka N, Hoshi Y, Kishida T, Tanaka H, Sato M, Takeshita N, Kuboki R, Nishino H, Ito M, Tahara M. Clinical impact of a dose-escalation strategy for lenvatinib in differentiated thyroid cancer. International journal of clinical oncology, 29:1435-1443, 2024
15. Shimizu H, Kodaira T, Kiyota N, Hayashi R, Nishino H, Asada Y, Mitani H, Hirayama Y, Onozawa Y, Nishio N, Hanai N, Ohkoshi A, Hara H, Monden N, Nagaoka M, Minami S, Fujii T, Tanaka K, Homma A, Yoshimoto S, Oridate N, Omori K, Ueda T, Okami K, Uemura H, Shiga K, Nakahira M, Asakage T, Saito Y, Sasaki K, Kitabayashi R, Ishikura S, Nishimura Y, Tahara M. Incidence and risk factors associated with the development of hypothyroidism after postoperative chemoradiotherapy for head and neck cancer patients with high-risk features: Supplementary analysis of JCOG1008. Oral oncology, 157:106976, 2024
16. Motegi A, Kageyama SI, Kashima Y, Hirata H, Hojo H, Nakamura M, Fujisawa T, Enokida T, Tahara M, Matsuura K, Zenda S. Detection of HPV DNA in Saliva of Patients with HPV-Associated Oropharyngeal Cancer Treated with Radiotherapy. Current oncology (Toronto, Ont.), 31:4397-4405, 2024
17. Imamura Y, Kiyota N, Tahara M, Kodaira T, Hayashi R, Nishino H, Asada Y, Mitani H, Iwae S, Nishio N, Onozawa Y, Hanai N, Ohkoshi A, Hara H, Monden N, Nagaoka M, Minami S, Kitabayashi R, Sasaki K, Homma A. Effect of acute kidney injury and overall survival in patients with postoperative head and neck cancer who received chemoradiotherapy with cisplatin: A supplementary analysis of the phase II/III trial of JCOG1008. Cancer medicine, 13:e70235, 2024
18. Oridate N, Takahashi S, Tanaka K, Shimizu Y, Fujimoto Y, Matsumoto K, Yokota T, Yamazaki T, Takahashi M, Ueda T, Hanai N, Yamaguchi H, Hara H, Yoshizaki T, Yasumatsu R, Nakayama M, Shiga K, Fujii T, Mitsugi K, Takahashi K, Nohata N, Gumuscu B, Lerman N, Tahara M. First-line pembrolizumab with or without chemotherapy for recurrent or metastatic head and neck squamous cell carcinoma: 5-year follow-up of the Japanese population of KEYNOTE‑048. International journal of clinical oncology, 29:1825-1839, 2024
