Annual Report 2024
Department of Medical Oncology
Toru Mukohara, Ako Hosono, Yoichi Naito, Nobuaki Matsubara, Kenichi Harano, Chihiro Kondoh, Chikako Funasaka, Hiromichi Nakajima, Hirofumi Mukai, Nobuyuki Takahashi, Misao Fukuda, Akira Hirota, Mai Shimura, Mao Uematsu
Introduction
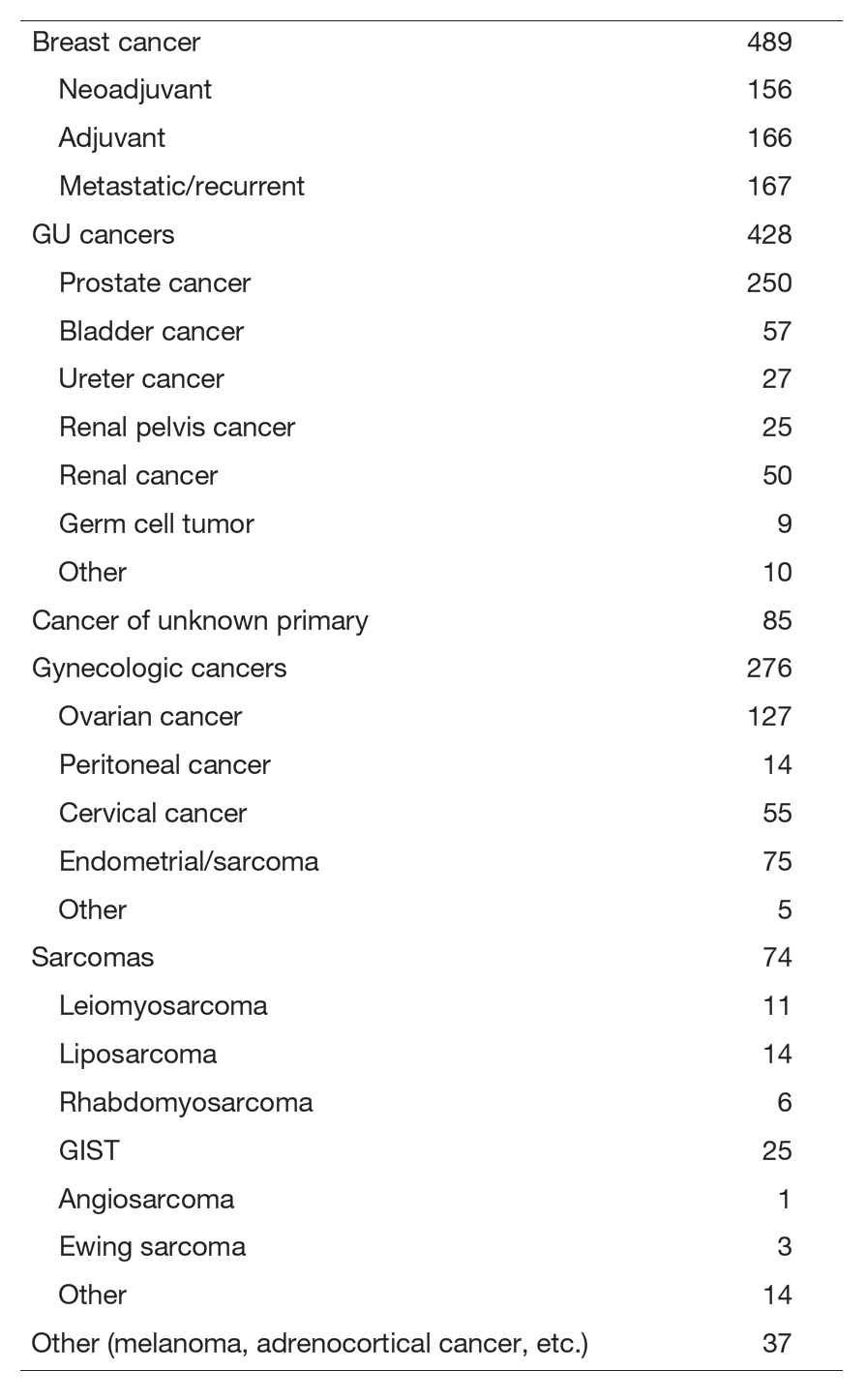
Our department is the only department in the hospital that provides care for cross-organ cancers, mainly breast cancer, urologic cancer, gynecologic cancer, and bone and soft tissue sarcoma. We also provide appropriate treatment for malignancies that are difficult to treat at other facilities, such as cancers of unknown primary, multiple, and rare cancers, utilizing our abundant experience as an oncologist. We also play a central role in conferences that include surgical departments and radiation therapy departments, such as the "Urologic Tumor Conference," "Bone and Soft Tissue Sarcoma Conference," "Gynecologic Tumor Conference," and "Breast Tumor Conference," and implement multidisciplinary treatment.
The Team and What We Do
Our department primarily focuses on breast cancer, urological cancers, gynecological cancers, and bone and soft tissue sarcomas, offering the hospital's only cross-organ cancer treatment program. We also leverage our extensive experience as medical oncologists to provide appropriate treatment for diseases challenging to manage at other facilities, such as cancers of unknown primary origin, multiple cancers, and rare cancers. Furthermore, we play a central role in conferences including the "Urological Oncology Conference," "Bone and Soft Tissue Sarcoma Conference," "Gynecologic Oncology Conference," and "Breast Oncology Conference," playing a central role in multidisciplinary treatment.
Clinically, the department has consistently maintained over 1,100 cases annually since fiscal year 2020 and recorded its highest-ever daily average patient volume. It is also deeply involved in the implementation of cancer gene profiling tests under the national health insurance system. Department Chief, Mukohara served as the Expert Panel Chair. Furthermore, out of the 1,013 cases deliberated by our hospital's Molecular Tumor Board in fiscal year 2024, 427 were handled by our department. In research, we conduct not only retrospective studies but also prospective clinical trials, new drug development trials, and associated translational research (TR).
Research Activities
We conduct not only retrospective studies but also prospective clinical trials, new drug development trials, and associated TR. A randomized, double-blind, physician-initiated trial targeting breast cancer patients receiving paclitaxel therapy, which began enrollment in fiscal year 2021 and aimed to prevent peripheral neuropathy through limb cooling therapy, has been completed. This study secured an oral presentation opportunity at the 2025 ASCO Annual Meeting. We plan to submit the results paper for publication. Additionally, enrollment was completed for a physician-initiated trial evaluating olaparib plus pembrolizumab as neoadjuvant chemotherapy for HRD-positive advanced ovarian cancer. A paper reporting the results, including associated translational research utilizing single-cell RNA sequencing, is currently under submission. As a collaborative study with the new drug development field, a paper reporting the efficacy testing of an ADC formulation using a breast cancer brain metastasis model is also under submission. Furthermore, we have completed an investigator-initiated trial of niraparib plus pimitespib and a collaborative study with a company on circulating tumor cells (CTCs), and are currently preparing reports on these results. Additionally, as a collaborative study with the basic science laboratories, we are conducting efficacy testing of a β-catenin inhibitor and establishing an ex vivo efficacy testing platform using cancer cells present in body cavity fluids.
Clinical Trials
IITs we are leading:
- Phase I Trial of Niraparib and Pimitespib in Patients with Solid Tumors (PI, Naito Y): jRCT2031220179
- A randomized Control trial to Evaluate mitigation of CIPN by limb-cooling apparatus in breast cancer patients who undergo weekly paclitaxel (CECILIA) (PI, Mukohara T): jRCT2032210115
- Phase I clinical study to evaluate the safety and tolerability of intraperitoneal administration of anti-GPC3-CAR expressing iPS cell-derived ILC/NK cells in patients with inoperable advanced recurrent ovarian clear cell carcinoma expressing GPC3 and with peritoneal dissemination (PI, Harano K): jRCT2033200431
- Double-blind, placebo-controlled, randomized, phase III trial of paclitaxel plus carboplatin plus atezolizumab in patients with advanced/ recurrent uterine cancer (PI, Harano K): jRCT2031190013
- Olaparib Monotherapy and Olaparib + Pembrolizumab Combination Therapy for Ovarian Cancer (OLAPem) (PI, Harano K): NCT04417192
Education
The goal of our department's training and education is to cultivate true medical oncologists. We aim for residents and cancer specialists who graduate from our department to be capable not only of providing standard treatment regardless of cancer type, but also of practicing daily clinical work in collaboration with non-physician medical staff. Furthermore, we require and guide residents to conduct research and write research papers addressing clinical questions they identify during their residency. Three English-language papers authored by residents under the guidance of our department staff were published (lead authors: Uematsu, Nakao, Fukuda). Furthermore, five residents had presentation opportunities at conferences such as the Japanese Society of Medical Oncology (JSMO) and the Japan Breast Cancer Society. Additionally, resident Shimura from our department received a group award at the JSMO-RISA workshop hosted by JSMO.
Future Prospects
In clinical practice, we provide high-patient-satisfaction care through multidisciplinary team-based medicine. In education, we advance the training of medical oncologists who possess both organ-transcending clinical capabilities and scientific expertise. In research, we lead development trials from Phase I to Phase III for new drugs. Furthermore, we conduct research with the Common Research and Development Division on preclinical studies focused on predicting drug efficacy and overcoming resistance, aiming to implement physician-initiated clinical trials based on the insights gained. We also continue to promote collaborative research with companies and other academic institutions.
List of papers published in 2024
Journal
1. Konno Y, Mayama M, Takehara K, Yokoyama Y, Suzuki J, Susumu N, Harano K, Nakagawa S, Nakanishi T, Yamagami W, Yoshihara K, Nomura H, Okamoto A, Aoki D, Watari H. Prognostic impact of the number of resected pelvic nodes in endometrial cancer: Japanese Gynecologic Oncology Group Study JGOG2043 post hoc analysis. Journal of gynecologic oncology, 36:e3, 2025
2. Shore ND, Broder MS, Barata PC, Crispino T, Fay AP, Lloyd J, Mellado B, Matsubara N, Pfanzelter N, Schlack K, Sieber P, Soares A, Dalglish H, Niyazov A, Shaman S, Zielinski MA, Chang J, Agarwal N. Expert Consensus Recommendations on the Management of Treatment-emergent Adverse Events Among Men with Prostate Cancer Taking Poly-ADP Ribose Polymerase Inhibitor + Novel Hormonal Therapy Combination Therapy. European urology oncology, 8:94-104, 2025
3. Tsuchiya K, Akisue T, Ehara S, Kawai A, Kawano H, Hiraga H, Hosono A, Hutani H, Morii T, Morioka H, Nishida Y, Oda Y, Ogose A, Shimose S, Yamaguchi T, Yamamoto T, Yoshida M. Japanese orthopaedic association (JOA) clinical practice guideline on the management of primary malignant bone tumors - Secondary publication. Journal of orthopaedic science, 30:1-17, 2025
4. Okazawa-Sakai M, Sakai SA, Hyodo I, Horasawa S, Sawada K, Fujisawa T, Yamamoto Y, Boku S, Hayasaki Y, Isobe M, Shintani D, Hasegawa K, Egawa-Takata T, Ito K, Ihira K, Watari H, Takehara K, Yagi H, Kato K, Chiyoda T, Harano K, Nakamura Y, Yamashita R, Yoshino T, Aoki D. Gut microbiome associated with PARP inhibitor efficacy in patients with ovarian cancer. Journal of gynecologic oncology, 36:e38, 2025
5. O'Donnell PH, Loriot Y, Csoszi T, Matsubara N, Shin SJ, Park SH, Atduev V, Gumus M, Karaca SB, Grivas P, de Wit R, Castellano DE, Powles T, Vuky J, Zhao Y, O'Hara K, Okpara CE, Franco S, Homet Moreno B, Żołnierek J, Siefker-Radtke AO. Efficacy and safety of pembrolizumab in patients with advanced urothelial carcinoma deemed potentially ineligible for platinum-containing chemotherapy: Post hoc analysis of KEYNOTE-052 and LEAP-011. Cancer, 131:e35601, 2025
6. Iwata H, Naito Y, Hattori M, Yoshimura A, Yonemori K, Aizawa M, Mori Y, Yoshimitsu J, Umeyama Y, Mukohara T. Safety and pharmacokinetics of vepdegestrant in Japanese patients with ER+ advanced breast cancer: a phase 1 study. International journal of clinical oncology, 30:72-82, 2025
7. Mamtani R, Matsubara N, Pino AM, Herranz UA, Şendur MAN, Gravis G, Huillard O, Lee HJ, Gafanov R, Joly F, Bedke J, Sella A, Chang YH, Imai K, Moreno BH, Xu JZ, Alva A, Powles T. Outcomes by Retrospective Eligibility for Maintenance Therapy of Patients With Advanced Urothelial Carcinoma: Post Hoc Analysis of the Phase 3 KEYNOTE-361 Trial. Clinical genitourinary cancer, 23:102248, 2025
8. Powles T, Csőszi T, Loriot Y, Matsubara N, Geczi L, Cheng SY, Fradet Y, Alva A, Oudard S, Vulsteke C, Morales-Barrera R, Fléchon A, Gunduz S, Liu CC, Moreno BH, Bavle A, Özgüroğlu M. Cisplatin- or Carboplatin-Based Chemotherapy Plus Pembrolizumab in Advanced Urothelial Cancer: Exploratory Analysis From the Phase 3 KEYNOTE-361 Study. Clinical genitourinary cancer, 23:102261, 2025
9. Bando H, Naito Y, Yamada T, Fujisawa T, Imai M, Sakamoto Y, Saigusa Y, Yamamoto K, Tomioka Y, Takeshita N, Sunami K, Futamura M, Notake C, Aoki S, Okano K, Yoshino T. A prospective study comparing highly qualified Molecular Tumor Boards with AI-powered software as a medical device. International journal of clinical oncology, 30:172-179, 2025
10. Matsubara N, Miyake H, Uemura H, Mizokami A, Kikukawa H, Kosaka T, Nishimura K, Nakamura M, Kobayashi K, Komaru A, Mori Y, Toyoizumi S, Hori N, Umeyama Y, Uemura H. Phase 3 Study of Talazoparib Plus Enzalutamide Versus Placebo Plus Enzalutamide as First-Line Treatment in Patients With Metastatic Castration-Resistant Prostate Cancer: TALAPRO-2 Japanese Subgroup Analysis. Cancer medicine, 14:e70333, 2025
11. Misawa S, Denda T, Kodama S, Suzuki T, Naito Y, Kogawa T, Takada M, Hino A, Shiosakai K, Kuwabara S. One-year incidence of chemotherapy-induced peripheral neuropathy in oxaliplatin- or taxane-based chemotherapy: a multicenter, prospective registry study (MiroCIP study). Expert opinion on pharmacotherapy, 26:335-344, 2025
12. Fizazi K, Azad AA, Matsubara N, Carles J, Fay AP, De Giorgi U, Joung JY, Fong PCC, Voog E, Jones RJ, Shore ND, Dunshee C, Zschäbitz S, Oldenburg J, Ye D, Lin X, Healy CG, Di Santo N, Laird AD, Zohren F, Agarwal N. Talazoparib plus enzalutamide versus placebo plus enzalutamide for patients with advanced prostate cancer and changes in specific DNA repair genes: a plain language summary of the results from the TALAPRO-2 study. Future oncology (London, England), 21:637-652, 2025
13. de Bono JS, He M, Shi Z, Nowicka M, Bracarda S, Sternberg CN, Chi KN, Olmos D, Sandhu S, Massard C, Matsubara N, Chen G, Bienz NS, Canter D, Wongchenko M, Sweeney C. Final Overall Survival and Molecular Data Associated with Clinical Outcomes in Patients Receiving Ipatasertib and Abiraterone in the Phase 3 IPATential150 Trial. European urology, 87:672-682, 2025
14. Konno Y, Mayama M, Takehara K, Yokoyama Y, Suzuki J, Susumu N, Harano K, Nakagawa S, Nakanishi T, Yamagami W, Yoshihara K, Nomura H, Okamoto A, Aoki D, Watari H. Prognostic significance of para-aortic node metastasis in endometrial cancer: Japanese Gynecologic Oncology Group Study JGOG2043 post hoc analysis. Journal of gynecologic oncology, 2025
15. Matsubara N, Osawa T, Abe T, Oya M, Nishimoto K, Iwahori T, Tsuchiya H, Murota M, Yoshida M, Tatematsu Y, Nakano Y, Eto M, Nonomura N. FGFR2/3 Gene Alterations and Clinical Outcomes in Advanced/Metastatic Urothelial Cancer in Japan: MONSTAR-SCREEN Database Study. Cancer science, 116:1424-1432, 2025
16. Taniguchi H, Uehara K, Ishikawa T, Okochi O, Akazawa N, Okuda H, Hasegawa H, Shiozawa M, Kataoka M, Satake H, Shimura T, Kondoh C, Kuramochi H, Matsumoto T, Takegawa N, Yamaguchi T, Nagase M, Nakamura M, Takano N, Fujita H, Watanabe T, Nishina T, Sakamoto Y, Moriwaki T, Ohori H, Nakanishi M, Kito Y, Utsunomiya S, Ishikawa T, Manaka D, Matsuoka H, Suto T, Arai T, Shinzaki S, Funakoshi T, Nakayama G, Negoro Y, Tsuji Y, Makiyama A, Takuma K, Arimoto A, Shinozaki K, Mishima A, Masuishi T. BRAF V600E and Non-V600E Mutations in RAS Wild-Type Metastatic Colorectal Cancer: Prognostic and Therapeutic Insights from a Nationwide, Multicenter, Observational Study (J-BROS). Cancers, 17:399, 2025
17. Suzuki K, Matsuyama H, Matsubara N, Kazama H, Ueno F, Uemura H. Current Evidence on Cabazitaxel for Prostate Cancer Therapy: A Narrative Review. International journal of urology, 32:475-487, 2025
18. Naito Y, Iwagami S, Doi T, Takahashi T, Kurokawa Y. Pimitespib in patients with advanced gastrointestinal stromal tumors in Japan: an expanded access program. International journal of clinical oncology, 30:935-943, 2025
19. Shiota M, Matsubara N, Kato T, Eto M, Osawa T, Abe T, Shinohara N, Nishimoto K, Yasumizu Y, Tanaka N, Oya M, Fujisawa T, Horasawa S, Nakamura Y, Yoshino T, Nonomura N. Genomic characterization of metastatic patterns in prostate cancer using circulating tumor DNA data from the SCRUM-Japan MONSTAR SCREEN project. The journal of liquid biopsy, 7:100282, 2025
20. Petrylak DP, Ratta R, Matsubara N, Korbenfeld E, Gafanov R, Mourey L, Todenhöfer T, Gurney H, Kramer G, Bergman AM, Zalewski P, De Santis M, Armstrong AJ, Gerritsen W, Pachynski R, Byun SS, Retz M, Levesque E, McDermott R, Bracarda S, Manneh R, Levartovsky M, Li XT, Schloss C, Poehlein CH, Fizazi K. Pembrolizumab Plus Docetaxel Versus Docetaxel for Previously Treated Metastatic Castration-Resistant Prostate Cancer: The Randomized, Double-Blind, Phase III KEYNOTE-921 Trial. Journal of clinical oncology, 43:1638-1649, 2025
21. Yamanaka T, Ukita J, Xue D, Kondoh C, Honda S, Noguchi M, Yonejima Y, Nonogaki K, Takemura K, Kizawa R, Yamaguchi T, Tanabe Y, Suyama K, Ogaki K, Miura Y. Artificial intelligence system for predicting hand-foot skin reaction induced by vascular endothelial growth factor receptor inhibitors. Scientific reports, 15:9843, 2025
22. Jones RH, Fizazi K, James ND, Tammela TL, Matsubara N, Priou F, Beuzeboc P, Lesimple T, Bono P, Kataja V, Garcia JA, Protheroe A, Shore N, Aspegren J, Joensuu H, Kuss I, Fiala-Buskies S, Vjaters E. Safety and tolerability of long-term treatment with darolutamide in patients with metastatic castration-resistant prostate cancer. Prostate cancer and prostatic diseases, 27:786-789, 2024
23. Fizazi K, Azad AA, Matsubara N, Carles J, Fay AP, De Giorgi U, Joung JY, Fong PCC, Voog E, Jones RJ, Shore ND, Dunshee C, Zschäbitz S, Oldenburg J, Ye D, Lin X, Healy CG, Di Santo N, Laird AD, Zohren F, Agarwal N. First-line talazoparib with enzalutamide in HRR-deficient metastatic castration-resistant prostate cancer: the phase 3 TALAPRO-2 trial. Nature medicine, 30:257-264, 2024
24. Dorff T, Horvath LG, Autio K, Bernard-Tessier A, Rettig MB, Machiels JP, Bilen MA, Lolkema MP, Adra N, Rottey S, Greil R, Matsubara N, Tan DSW, Wong A, Uemura H, Lemech C, Meran J, Yu Y, Minocha M, McComb M, Penny HL, Gupta V, Hu X, Jurida G, Kouros-Mehr H, Janát-Amsbury MM, Eggert T, Tran B. A Phase I Study of Acapatamab, a Half-life Extended, PSMA-Targeting Bispecific T-cell Engager for Metastatic Castration-Resistant Prostate Cancer. Clinical cancer research, 30:1488-1500, 2024
25. Takahashi K, Uozumi R, Mukohara T, Hayashida T, Iwabe M, Iihara H, Kusuhara-Mamishin K, Kitagawa Y, Tsuchiya M, Kitahora M, Nagayama A, Kosaka S, Asano-Niwa Y, Seki T, Ohnuki K, Suzuki A, Ono F, Futamura M, Kawazoe H, Nakamura T. Proton Pump Inhibitors and Cyclin-Dependent Kinase 4/6 Inhibitors in Patients With Breast Cancer. The oncologist, 29:e741-e749, 2024
26. Doi T, Takahashi S, Aoki D, Yonemori K, Hara H, Hasegawa K, Takehara K, Harano K, Yunokawa M, Nomura H, Shimoi T, Horie K, Ogasawara A, Okame S. A first-in-human phase I study of TAS-117, an allosteric AKT inhibitor, in patients with advanced solid tumors. Cancer chemotherapy and pharmacology, 93:605-616, 2024
27. Rosenberg JE, Mamtani R, Sonpavde GP, Loriot Y, Duran I, Lee JL, Matsubara N, Vulsteke C, Castellano D, Sridhar SS, Pappot H, Gurney H, Bedke J, van der Heijden MS, Galli L, Keam B, Masumori N, Meran J, O'Donnell PH, Park SH, Grande E, Sengeløv L, Uemura H, Skaltsa K, Campbell M, Matsangou M, Wu C, Hepp Z, McKay C, Powles T, Petrylak DP. Health-related Quality of Life in Patients with Previously Treated Advanced Urothelial Carcinoma from EV-301: A Phase 3 Trial of Enfortumab Vedotin Versus Chemotherapy. European urology, 85:574-585, 2024
28. Hirota S, Tateishi U, Nakamoto Y, Yamamoto H, Sakurai S, Kikuchi H, Kanda T, Kurokawa Y, Cho H, Nishida T, Sawaki A, Ozaka M, Komatsu Y, Naito Y, Honma Y, Takahashi F, Hashimoto H, Udo M, Araki M, Nishidate S. English version of Japanese Clinical Practice Guidelines 2022 for gastrointestinal stromal tumor (GIST) issued by the Japan Society of Clinical Oncology. International journal of clinical oncology, 29:647-680, 2024
29. Bardia A, Krop IE, Kogawa T, Juric D, Tolcher AW, Hamilton EP, Mukohara T, Lisberg A, Shimizu T, Spira AI, Tsurutani J, Damodaran S, Papadopoulos KP, Greenberg J, Kobayashi F, Zebger-Gong H, Wong R, Kawasaki Y, Nakamura T, Meric-Bernstam F. Datopotamab Deruxtecan in Advanced or Metastatic HR+/HER2- and Triple-Negative Breast Cancer: Results From the Phase I TROPION-PanTumor01 Study. Journal of clinical oncology, 42:2281-2294, 2024
30. Kakuwa T, Watanabe T, Niino M, Kawata A, Satoh T, Matsumura N, Yokoyama Y, Kawana K, Hirashima Y, Kyo S, Yasuda M, Harano K, Machida H, Tokunaga H, Kaneuchi M, Tabata T, Kobayashi Y, Nagase S, Katabuchi H, Mikami M, Yamamoto Y, Rikitake R, Ichinose Y, Higashi T. Quality of care measurement for patients with ovarian cancer in Japan. The journal of obstetrics and gynaecology research, 50:1182-1191, 2024
31. Li BT, Meric-Bernstam F, Bardia A, Naito Y, Siena S, Aftimos P, Anderson I, Curigliano G, de Miguel M, Kalra M, Oh DY, Park JO, Postel-Vinay S, Rha SY, Satoh T, Spanggaard I, Michelini F, Smith A, Machado KK, Saura C. Trastuzumab deruxtecan in patients with solid tumours harbouring specific activating HER2 mutations (DESTINY-PanTumor01): an international, phase 2 study. The Lancet. Oncology, 25:707-719, 2024
32. Tan RYC, Ong WS, Lee KH, Park S, Iqbal J, Park YH, Lee JE, Yu JH, Lin CH, Lu YS, Ono M, Ueno T, Naito Y, Onishi T, Lim GH, Tan SM, Lee HB, Koh J, Han W, Im SA, Tan VKM, Phyu N, Wong FY, Tan PH, Yap YS. Outcomes in Nonmetastatic Hormone Receptor-Positive HER2-Negative Pure Mucinous Breast Cancer: A Multicenter Cohort Study. Journal of the National Comprehensive Cancer Network : JNCCN, 22:e237121, 2024
33. Shimura M, Fujikawa H, Yazawa M, Matsumoto Y, Yamada M. An Autopsy Case of Reversible Cerebral Vasoconstriction Syndrome After a Severe Acute Respiratory Syndrome Coronavirus 2 Vaccination. Cureus, 16:e59311, 2024
34. Mukohara T, Park YH, Sommerhalder D, Yonemori K, Hamilton E, Kim SB, Kim JH, Iwata H, Yamashita T, Layman RM, Mita M, Clay T, Chae YS, Oakman C, Yan F, Kim GM, Im SA, Lindeman GJ, Rugo HS, Liyanage M, Saul M, Le Corre C, Skoura A, Liu L, Li M, LoRusso PM. Inhibition of lysine acetyltransferase KAT6 in ER(+)HER2(-) metastatic breast cancer: a phase 1 trial. Nature medicine, 30:2242-2250, 2024
35. Yamamoto N, Kuboki Y, Harano K, Koyama T, Kondo S, Hagiwara A, Suzuki N, Fujikawa E, Toyoizumi K, Mukai M, Doi T. A phase 1/1b, open-label, dose-escalation study of PD-1 inhibitor, cetrelimab alone and in combination with FGFR inhibitor, erdafitinib in Japanese patients with advanced solid tumors. Investigational new drugs, 42:376-385, 2024
36. Mukohara T, Park YH, Sommerhalder D, Yonemori K, Hamilton E, Kim SB, Kim JH, Iwata H, Yamashita T, Layman RM, Mita M, Clay T, Chae YS, Oakman C, Yan F, Kim GM, Im SA, Lindeman GJ, Rugo HS, Liyanage M, Saul M, Le Corre C, Skoura A, Liu L, Li M, LoRusso PM. Author Correction: Inhibition of lysine acetyltransferase KAT6 in ER(+)HER2(-) metastatic breast cancer: a phase 1 trial. Nature medicine, 30:2371, 2024
37. Wada R, Yamamoto T, Iwasaki K, Usui S, Matsubara N, Nomori H. Focal ground glass opacity of the lung in metachronous prostate and gastric cancer: A case report. Molecular and clinical oncology, 21:54, 2024
38. Matsubara N, Miura Y, Nishiyama H, Taoka R, Kojima T, Shimizu N, Hwang J, Ote T, Oyama R, Toyoizumi K, Mukhopadhyay S, Triantos S, Deprince K, Loriot Y. Phase 3 THOR Japanese subgroup analysis: erdafitinib in advanced or metastatic urothelial cancer and fibroblast growth factor receptor alterations. International journal of clinical oncology, 29:1516-1527, 2024
39. Kohjimoto Y, Uemura H, Yoshida M, Hinotsu S, Takahashi S, Takeuchi T, Suzuki K, Shinmoto H, Tamada T, Inoue T, Sugimoto M, Takenaka A, Habuchi T, Ishikawa H, Mizowaki T, Saito S, Miyake H, Matsubara N, Nonomura N, Sakai H, Ito A, Ukimura O, Matsuyama H, Hara I. Japanese clinical practice guidelines for prostate cancer 2023. International journal of urology, 31:1180-1222, 2024
40. Krämer A, Bochtler T, Pauli C, Shiu KK, Cook N, de Menezes JJ, Pazo-Cid RA, Losa F, Robbrecht DG, Tomášek J, Arslan C, Özgüroğlu M, Stahl M, Bigot F, Kim SY, Naito Y, Italiano A, Chalabi N, Durán-Pacheco G, Michaud C, Scarato J, Thomas M, Ross JS, Moch H, Mileshkin L. Molecularly guided therapy versus chemotherapy after disease control in unfavourable cancer of unknown primary (CUPISCO): an open-label, randomised, phase 2 study. Lancet (London, England), 404:527-539, 2024
41. Colombo N, Biagioli E, Harano K, Galli F, Hudson E, Antill Y, Choi CH, Rabaglio M, Marmé F, Marth C, Parma G, Fariñas-Madrid L, Nishio S, Allan K, Lee YC, Piovano E, Pardo B, Nakagawa S, McQueen J, Zamagni C, Manso L, Takehara K, Tasca G, Ferrero A, Tognon G, Lissoni AA, Petrella M, Laudani ME, Rulli E, Uggeri S, Barretina Ginesta MP. Atezolizumab and chemotherapy for advanced or recurrent endometrial cancer (AtTEnd): a randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet. Oncology, 25:1135-1146, 2024
42. Fukuda M, Mukohara T, Kuwata T, Sunami K, Naito Y. Efficacy of Trametinib in Neurofibromatosis Type 1-Associated Gastrointestinal Stromal Tumors: A Case Report. JCO precision oncology, 8:e2300649, 2024
43. Yamamoto Y, Yukami H, Yamaguchi T, Ohori H, Nagasu S, Kagawa Y, Sugimoto N, Sonoda H, Yamazaki K, Takashima A, Okuyama H, Hasegawa H, Kondo C, Baba E, Matsumoto T, Kawamoto Y, Kataoka M, Shindo Y, Ishikawa T, Esaki T, Kito Y, Sato T, Funakoshi T, Yamaguchi T, Shimada Y, Moriwaki T. Real-world outcomes of FOLFOXIRI plus bevacizumab in patients with metastatic colorectal cancer: the JSCCR-TRIPON study. International journal of clinical oncology, 29:1878-1886, 2024
44. Aoki D, Tabata T, Yanagida S, Nakamura T, Kondo E, Hamanishi J, Harano K, Hasegawa K, Hirasawa T, Hori K, Komiyama S, Matsuura M, Nakai H, Nakamura H, Sakata J, Takehara K, Takekuma M, Yokoyama Y, Kase Y, Sumino S, Soeda J, Kato A, Suri A, Okamoto A, Sugiyama T. Niraparib in Japanese patients with heavily pretreated, homologous recombination-deficient ovarian cancer: final results of a multicenter phase 2 study. Journal of gynecologic oncology, 35:e114, 2024
45. Nakao T, Harano K, Wakabayashi M, Naito Y, Tanabe H, Mukohara T. Efficacy of a platinum-based chemotherapy rechallenge for platinum-sensitive recurrence after PARP inhibitor maintenance. Gynecologic oncology reports, 55:101482, 2024
46. Shiota M, Matsubara N, Kato T, Eto M, Osawa T, Abe T, Shinohara N, Nishimoto K, Yasumizu Y, Tanaka N, Oya M, Fujisawa T, Horasawa S, Nakamura Y, Yoshino T, Nonomura N. Prediction of undetectable circulating tumor DNA by comprehensive genomic profiling assay in metastatic prostate cancer: the SCRUM-Japan MONSTAR SCREEN project. World journal of urology, 42:526, 2024
47. Naito Y, Nakamura S, Kawaguchi-Sakita N, Ishida T, Nakayama T, Yamamoto Y, Masuda N, Matsumoto K, Kogawa T, Sudo K, Shimomura A, Lai C, Zhang D, Iwahori Y, Gary D, Huynh D, Iwata H. Preliminary results from ASCENT-J02: a phase 1/2 study of sacituzumab govitecan in Japanese patients with advanced solid tumors. International journal of clinical oncology, 29:1684-1695, 2024
48. Fléchon A, Morales-Barrera R, Powles T, Alva A, Özgüroğlu M, Csöszi T, Loriot Y, Rodriguez-Vida A, Géczi L, Cheng SY, Fradet Y, Oudard S, Vulsteke C, Gunduz S, Mamtani R, Yu EY, Montesa Pino A, Anido U, Sendur MAN, Gravis G, Révész J, Kostorov V, Huillard O, Ma J, Rajasagi M, Vajdi A, Lunceford J, Cristescu R, Imai K, Homet Moreno B, Matsubara N. Association of Tumor Mutational Burden and PD-L1 with the Efficacy of Pembrolizumab with or without Chemotherapy versus Chemotherapy in Advanced Urothelial Carcinoma. Clinical cancer research, 30:5353-5364, 2024
49. Azad AA, Fizazi K, Matsubara N, Saad F, De Giorgi U, Joung JY, Fong PCC, Jones RJ, Zschäbitz S, Oldenburg J, Shore ND, Dunshee C, Carles J, Fay AP, Lin X, DeAnnuntis L, Di Santo N, Zielinski MA, Agarwal N. Talazoparib plus enzalutamide in metastatic castration-resistant prostate cancer: Safety analyses from the randomized, placebo-controlled, phase III TALAPRO-2 study. European journal of cancer (Oxford, England : 1990), 213:115078, 2024
50. Hashimoto T, Nakamura Y, Komatsu Y, Yuki S, Takahashi N, Okano N, Hirano H, Ohtsubo K, Ohta T, Oki E, Nishina T, Yasui H, Kawakami H, Esaki T, Machida N, Doi A, Boku S, Kudo T, Yamamoto Y, Kanazawa A, Denda T, Goto M, Iida N, Ozaki H, Shibuki T, Imai M, Fujisawa T, Bando H, Naito Y, Yoshino T. Different efficacy of tyrosine kinase inhibitors by KIT and PGFRA mutations identified in circulating tumor DNA for the treatment of refractory gastrointestinal stromal tumors. BJC reports, 2:54, 2024
Book
1. Nakajima H, Mukohara T. Understanding mechanisms of resistance to HER2-targeted therapies in HER2-positive breast cancer. In: Bonavida B (ed), Overcoming Cancers Resistant to HER-2 Antibodies: Breaking Tolerance to Antibody-Mediated Immunotherapy, United Kingdom, Academic Press, pp 45-56, 2024
