Annual Report 2024
Department of Hepatobiliary and Pancreatic Oncology
Masafumi Ikeda, Shuichi Mitsunaga, Hiroshi Imaoka, Mitsuhito Sasaki, Kazuo Watanabe, Tomoyuki Satake, Taro Shibuki, Kanae Inoue, Tomonao Taira, Shota Yamaguchi, Go Igarashi
Introduction
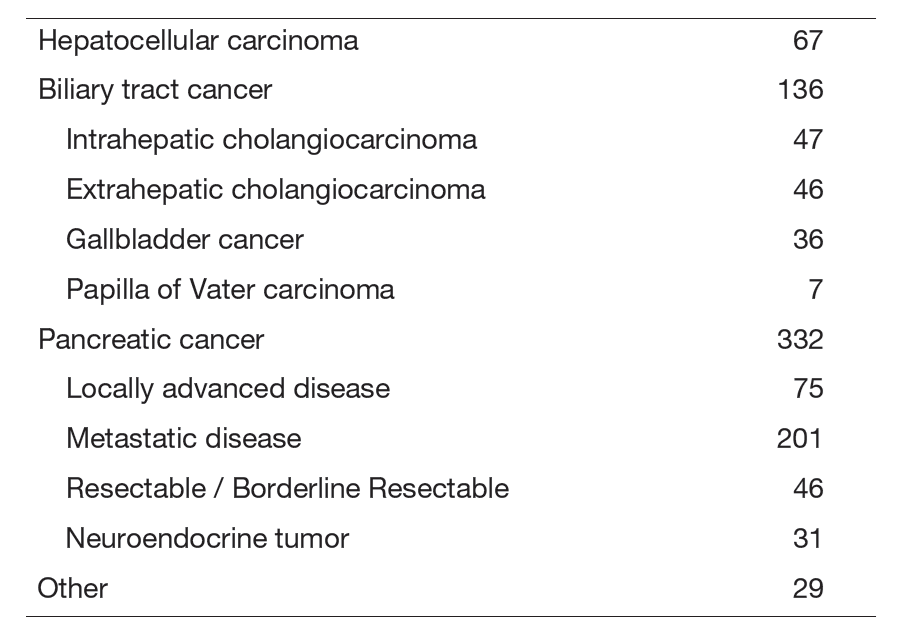
The Department of Hepatobiliary and Pancreatic Oncology is responsible for the diagnosis and treatment of patients with hepatic, biliary, pancreatic cancers and neuroendocrine neoplasms as well as interventional management by endoscopic or percutaneous procedures (Table 1). Our goal is to provide high-quality cancer treatment with adequate palliative care, and to develop novel and effective treatments and procedures through well-designed clinical trials and research.
The Team and What We Do
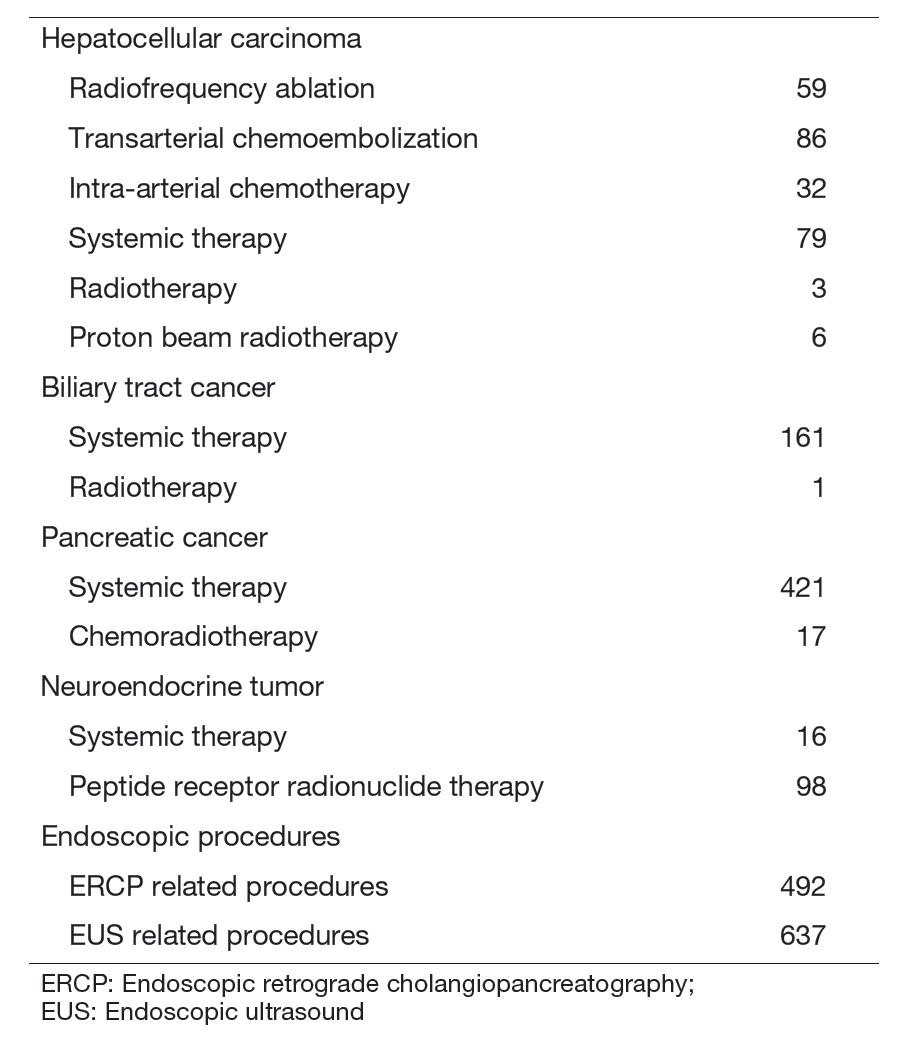
Our department consists of 7 staff oncologists and 3 residents, with an average of 36 beds in the hospital. These doctors are grouped into two teams and each team determines daily treatment plans for each admitted patient, and all doctors discuss the overall treatment strategy for all admitted patients in our department once a week. The major treatment strategies for individual patients are discussed in weekly tumor board conferences attended by medical oncologists, surgeons, radiologists, radiation oncologists, and pharmacists. We are also responsible for endoscopic abdominal ultrasonographic examinations, endoscopic or percutaneous ultrasound-guided biopsies of abdominal masses, local ablative therapy for liver tumors, endoscopic or percutaneous biliary and abscess drainage, and obstructive jaundice stenting, etc (Table 2).
Research Activities
Hepatocellular carcinoma (HCC)
In Japan, systemic therapy for HCC is evolving with the optimization of molecular targeted agents and immunotherapies, along with tailored regimen selection based on liver function, comorbidities, and treatment lines. The phase II trial of lenvatinib plus hepatic arterial infusion chemotherapy with cisplatin for unresectable HCC (LEOPARD) demonstrated favorable antitumor efficacy and tolerability without the use of immune checkpoint inhibitors or bevacizumab, and development has begun for its use as neoadjuvant therapy in borderline resectable cases. Additionally, a multicenter phase II study confirmed the tolerability of atezolizumab plus bevacizumab in patients with Child-Pugh class B liver function. An analysis from 2021-2022 reported an increased financial burden due to the rising use of costly combination regimens, highlighting the need to balance survival benefits with cost-effectiveness. Looking ahead, individualizing treatment sequencing based on imaging and biomarkers, establishing combination strategies originating from Japan, and evaluating their cost-effectiveness will be key priorities for the field.
Biliary tract cancer (BTC)
A Japan-U.S. joint analysis demonstrated that anti-HER2 therapy provides significant survival benefits in HER2-amplified cases, highlighting the growing importance of treatment selection based on molecular profiling. In adjuvant S-1 therapy, recurrence risk stratification based on 5-FU metabolism-related genes has been proposed, suggesting the potential for personalized medicine. A post-hoc analysis of the JCOG1113 trial showed no inter-institutional differences in outcomes between Gemcitabine (Gem) + Cisplatin (GC) and Gem + S-1 (GS) regimens, confirming the external validity of the results. In a comparison of biliary drainage techniques for hilar cholangiocarcinoma, the stent-in-stent approach demonstrated longer patency and time to re-intervention under systemic chemotherapy and was recommended as the preferred method. Additionally, participation in an international collaborative study of GC plus durvalumab therapy provided real-world clinical data. Together, these findings represent major advances toward both the personalization and the standardization of BTC treatment, spanning adjuvant therapy, targeted therapy, and supportive care.
Pancreatic cancer (PC)
The retrospective analyses have shown that nal-IRI plus 5-FU/LV offers superior efficacy compared with S-1 as a second-line treatment for metastatic disease, providing a new option for standard therapy selection. In first-line treatment, the efficacy of NALIRIFOX compared with Gem + nab-paclitaxel (GnP) has been reported, and a detailed protocol for a domestic phase II trial aiming for regulatory approval in Japan has been published. A secondary analysis of a comparative trial of mFOLFIRINOX versus GnP for locally advanced PC revealed that Depth of Response may serve as a prognostic indicator. On the diagnostic front, serum circulating miRNA signatures and urinary CD276 fragments have shown promise as non-invasive biomarkers for early detection and identification of resectable PC. High HER3 expression suggests a potentially new therapeutic target, and covered metal stents have been shown to be an effective treatment option for tumor bleeding due to duodenal invasion. Even in patients with malignant ascites, GnP retains acceptable efficacy and safety, although prognosis remains poor, highlighting the need for enhanced supportive care. Together, these findings are continuously advancing the evidence base for pancreatic cancer management, spanning diagnosis, first- and second-line therapies, and supportive care.
Others
In pancreatic and gastrointestinal neuroendocrine tumors (NET), a "Treatment Selection MAP" to guide first-line therapy was validated in a multicenter study, demonstrating a significant improvement in progression-free survival when treatment was guided by the MAP. The MAP was shown to align with established prognostic factors and tumor characteristics, contributing to greater consistency in clinical decision-making. Furthermore, practical management strategies not fully covered by current guidelines were organized into a "NEN (NeuroEndocrine Neoplasms) Policy Recommendation" in collaboration with experts, representing an important step toward the standardization of care.
Clinical Trials
One hundred and six clinical trials (sponsored: 56 trials, investigator-initiated: 51 trials) are ongoing, and 10 clinical trials (sponsored: 3 trials, investigator-initiated: 7 trials) are being planned for the upcoming year.
HCC
Over the past year, significant progress has been made in the field of HCC, with numerous clinical trials exploring combinations of immunotherapy, molecular targeted agents, and locoregional treatments. In first-line therapy, phase III trials of combination immunotherapies such as atezolizumab plus bevacizumab plus tiragolumab and relatlimab plus nivolumab plus bevacizumab are ongoing, along with a phase II study of budigalimab plus livmoniplimab. Multimodal strategies are also being investigated, including combinations with TACE (lenvatinib plus pembrolizumab, durvalumab plus tremelimumab), atezolizumab plus bevacizumab for unresectable intermediate-stage disease (REPLACEMENT), and for Child-Pugh B patients (CHALLENGE). Neoadjuvant treatment with the LEOPARD-Neo regimen and biomarker studies such as ctDNA analysis (COSMOS-HCC-01) are also progressing. Furthermore, exploratory trials of novel antibody therapeutics such as CBA-1205, ERY974, and MK-1308A are underway. In real-world practice, all-case surveillance studies of cabozantinib and tremelimumab are accumulating safety and efficacy data, which are expected to contribute to optimizing future treatment strategies.
BTC
Over the past year, significant advances have been made in the treatment strategies for BTC, integrating immunotherapy, molecular targeted agents, and chemotherapy. In first-line treatment, ongoing studies include trastuzumab deruxtecan plus rilvegostomig (DESTINY-BTC-01) and zanidatamab for HER2-positive cases, as well as the GEMINI-Hepatobiliary master protocol exploring novel immunomodulators. Efforts to optimize therapy are also underway, such as neoadjuvant GC+S-1 therapy (JCOG1920) for resectable cases and continuation of triple-agent regimens beyond nine cycles in unresectable disease. In later-line settings, targeted therapies based on molecular alterations, such as pemigatinib and futibatinib for FGFR2 fusion-positive cases and ivosidenib for IDH1-mutant tumors-are rapidly advancing. The potential of immunotherapy is further expanding through studies like MARVEL (radiotherapy plus durvalumab) and the investigator-initiated OPAL trial of nivolumab for occupational BTC. Additionally, ctDNA analyses such as COSMOS-BTC-01 and real-world evaluations of immunotherapy are progressing, laying the foundation for the implementation of precision medicine.
PC
Over the past year, significant progress has been made in the field of PC, with a wide range of clinical trials combining immunotherapy, molecular targeted agents, and novel antibody therapies. In first-line treatment, ongoing studies include combinations with NIS793 or quemliclustat, a trial of zolbetuximab in CLDN18.2-positive patients, and a trial of S-1-based chemoradiotherapy plus nivolumab for locally advanced disease (JCOG1908E). For elderly patients with resectable PC, a neoadjuvant GnP regimen (PRESTIGE) is being conducted. Efforts to develop personalized treatments based on genetic alterations are also advancing, such as AMG193 combination therapy for MTAP-deficient pancreatic cancer and binimetinib for BRAF fusion-positive cases. In diagnostics, ctDNA analyses (COSMOS-PC-01, ARTEMIS-PC) are progressing, and studies on ponsegromab and edralimab are being carried out for cancer cachexia. These advances represent comprehensive progress across diagnosis, treatment, and supportive care for PC.
NET
Over the past year, significant progress has been made in the field of NET, with various clinical studies involving molecular targeted therapies and immunotherapies. A phase III trial (JCOG1901) comparing everolimus monotherapy with everolimus plus lanreotide for unresectable NET is ongoing, as is a comparative study (JCOG2107E) of etoposide plus carboplatin ± durvalumab for neuroendocrine carcinoma. Furthermore, an all-case study of Lutetium-177 DOTATATE, and a phase II study (DAREON-5) of the novel DLL3-targeted antibody BI764532 are also progressing, driving the development of new therapeutic options.
Others
Over the past year, significant progress has been made in molecular profiling and the development of treatments targeting rare subtypes in hepatobiliary, pancreatic cancers, and NET, with genomic studies such as the MASCARPONE trial and MONSTAR-screen underway. In the endoscopy field, a nationwide survey on needle tract seeding after EUS-FNA, analysis of duodenal fluid components, and a comparative study of anti-reflux metal stents have been conducted, leading to advancements in diagnostic and therapeutic techniques.
Education
Our trainees are provided with daily training with group discussions on the daily practice of inpatient and outpatient management. They can learn the indications, administration and management of adverse events from loco-regional treatments to systemic therapy for patients with hepatic, biliary, and pancreatic cancer and the accompanying procedures to undertake diagnosis and interventional management, and provide outpatient care. Trainees conduct some retrospective clinical researches and prospective clinical trials to resolve some clinical questions arising from daily practice. Further, they can make presentations of their research in domestic and overseas meetings and publish papers in English under the instruction of staff physicians.
Future Prospects
Looking ahead, the field of hepatobiliary and pancreatic cancers is entering a pivotal phase. Immunotherapy has become a standard treatment for HCC and BTC, and combination strategies with targeted agents and locoregional therapies continue to evolve. Although the incidence of HCC in Japan is decreasing and that of BTC has plateaued, both remain more common than in other countries. Regarding PC, cytotoxic agents remain the mainstay but have limited efficacy, while the number of patients is rapidly rising, highlighting the urgent need for new therapies.
Japanese researchers are expected to lead this progress by participating in early-phase drug development, conducting domestic trials, and driving international collaborations. Future priorities include optimizing immunotherapy and targeted therapy, advancing multidisciplinary strategies, and integrating precision medicine approaches such as biomarker research, liquid biopsy, and ctDNA analysis. Continued innovation in endoscopic and locoregional techniques will also be essential. Leveraging Japan's strong clinical research infrastructure will be key to creating new treatments and shaping global standards of care.
List of papers published in 2024
Journal
1. Oh DY, Ikeda M, Lee CK, Rojas C, Hsu CH, Kim JW, Shen L, Furuse J, Park JO, Borad M, de Braud F, Bridgewater J, Lee SS, Moehler M, Audhuy F, Osada M, Sato M, Yoo C. Bintrafusp alfa and chemotherapy as first-line treatment in biliary tract cancer: A randomized phase 2/3 trial. Hepatology (Baltimore, Md.), 81:823-836, 2025
2. Muramatsu S, Ogura K, Morizane C, Satake T, Toda Y, Iwata S, Kobayashi E, Higashi T, Ikeuchi M, Kawai A. Statistics of Visceral Sarcoma in Japan: Report From the Population-Based National Cancer Registry (NCR) in Japan. Journal of surgical oncology, 131:274-284, 2025
3. Lo Prinzi F, Salani F, Rimini M, Rizzato MD, Antonuzzo L, Camera S, Satake T, Vandeputte H, Vivaldi C, Pressiani T, Lucchetti J, Kim JW, Abidoye O, Rapposelli IG, Tamberi S, Finkelmeier F, Giordano G, Pircher C, Chon HJ, Braconi C, Pastorino A, Castet F, Tamburini E, Yoo C, Parisi A, Diana A, Scartozzi M, Prager GW, Avallone A, Schirripa M, Kim IH, Perkhofer L, Oneda E, Verrico M, Adeva J, Chan SL, Spinelli GP, Personeni N, Garajova I, Rodriquenz MG, Leo S, Melo Alvim C, Roque R, Fornaro L, De Rosa A, Lavacchi D, Rossari F, Ikeda M, Dekervel J, Niger M, Balsano R, Tonini G, Kang M, Bekaii-Saab T, Viola MG, Silvestro L, Esposito L, Boccaccino A, Himmelsbach V, Landriscina M, Ahcene Djaballah S, Zanuso V, Masi G, Lonardi S, Rimassa L, Casadei-Gardini A. Efficacy of cisplatin-gemcitabine-durvalumab in patients with advanced biliary tract cancer experiencing early vs late disease relapse after surgery: a large real-life worldwide population. The oncologist, 30:oyae256, 2025
4. Okuno T, Morizane C, Mizusawa J, Yanagimoto H, Kobayashi S, Imaoka H, Terashima T, Kawakami H, Sano Y, Okusaka T, Ikeda M, Ozaka M, Miwa H, Todaka A, Shimizu S, Mizuno N, Sekimoto M, Sano K, Tobimatsu K, Katanuma A, Gotoh K, Yamaguchi H, Ishii H, Furuse J, Ueno M. Influence of major hepatectomy on gemcitabine-based chemotherapy for recurrent biliary tract cancer after surgery: a subgroup analysis of JCOG1113. International journal of clinical oncology, 30:83-91, 2025
5. Morizane C, Ueno M, Ioka T, Tajika M, Ikeda M, Yamaguchi K, Hara H, Yabusaki H, Miyamoto A, Iwasa S, Muto M, Takashima T, Minashi K, Komatsu Y, Nishina T, Nakajima TE, Takeno A, Moriwaki T, Furukawa M, Sahara T, Ikezawa H, Nomoto M, Takashima S, Uehara T, Funasaka S, Yashiro M, Furuse J. Tasurgratinib in patients with cholangiocarcinoma or gastric cancer: Expansion part of the first-in-human phase I study. Cancer science, 116:192-203, 2025
6. Imaoka H, Ikeda M, Kobayashi S, Ohba A, Ueno M, Suzuki Y, Tsumura H, Kimura N, Kawaguchi S, Kawamoto Y, Nakachi K, Tsuji K, Kobayashi N, Ashida R, Okano N, Umemoto K, Murohisa G, Hosokawa A, Asagi A, Nebiki H, Suzuki R, Terashima T, Shibata R, Kawata K, Doi T, Ohyama H, Kitano Y, Shioji K, Okuyama H, Naganuma A, Negoro Y, Sakamoto Y, Shimizu S, Morizane C, Ueno M, Furuse J, Nagano H. Efficacy of liposomal irinotecan + 5-FU/LV vs. S-1 in gemcitabine-refractory metastatic pancreatic cancer: a real-world study using inverse probability of treatment weighting. Journal of gastroenterology, 60:356-367, 2025
7. Ogata D, Namikawa K, Nakano E, Fujimori M, Uchitomi Y, Higashi T, Satake T, Morizane C, Yamazaki N, Kawai A. Comprehensive epidemiology of melanoma at all sites: insights from Japan's National Cancer Registry, 2016-2017. International journal of clinical oncology, 30:194-198, 2025
8. Fukushi K, Imaoka H, Ikeda M, Mizusawa J, Morizane C, Okusaka T, Kobayashi S, Sasahira N, Shimizu S, Yamazaki K, Okano N, Miwa H, Hara K, Satoi S, Sano K, Sakai K, Sugimoto R, Nakamura K, Terashima T, Ozaka M, Ueno M. Assessment of heterogeneity according to hospital or medical experience factors in outcomes of chemotherapy for advanced biliary tract cancer: a post-hoc analysis of JCOG1113. Japanese journal of clinical oncology, 55:355-361, 2025
9. Kudo M, Ren Z, Guo Y, Han G, Lin H, Zheng J, Ogasawara S, Kim JH, Zhao H, Li C, Madoff DC, Ghobrial RM, Kawaoka T, Gerolami R, Ikeda M, Kumada H, El-Khoueiry AB, Vogel A, Peng X, Mody K, Dutcus C, Dubrovsky L, Siegel AB, Finn RS, Llovet JM. Transarterial chemoembolisation combined with lenvatinib plus pembrolizumab versus dual placebo for unresectable, non-metastatic hepatocellular carcinoma (LEAP-012): a multicentre, randomised, double-blind, phase 3 study. Lancet (London, England), 405:203-215, 2025
10. Katsuya Y, Ikeda M, Koyama T, Sato J, Okada M, Matsubara N, Kondoh C, Mukohara T, Watanabe K, Kotani D, Ogawa Y, Taoka S, Yamamoto N. A Phase I, First-In-Human Study of CBA-1205, an Anti-DLK1 Monoclonal Antibody, in Patients With Advanced Solid Tumors. Cancer science, 116:1012-1022, 2025
11. Mitsunaga S, Ikeda M, Ueno M, Kobayashi S, Tsuda M, Miki I, Kuwahara T, Hara K, Takayama Y, Matsunaga Y, Hanada K, Shimizu A, Yoshida H, Nomoto T, Takahashi K, Iwamoto H, Iwama H, Hatano E, Nakata K, Nakamura M, Sudo H, Takizawa S, Ochiai A. Robust circulating microRNA signature for the diagnosis and early detection of pancreatobiliary cancer. BMC medicine, 23:23, 2025
12. Araki T, Sonoda Y, Shimokawa M, Otsuka T, Hayashi K, Honda T, Nakao K, Shibuki T, Nakazawa J, Arima S, Miwa K, Okabe Y, Koga F, Ueda Y, Kubotsu Y, Shimokawa H, Takeshita S, Komori A, Nishikawa K, Otsu S, Hosokawa A, Oda H, Sakai T, Arita S, Kawahira M, Taguchi H, Tsuneyoshi K, Kawaguchi Y, Fujita T, Sakae T, Shirakawa T, Mizuta T, Mitsugi K. Relationship between neutropenia caused by nanoliposomal irinotecan/fluorouracil/leucovorin and treatment outcomes in the NAPOLEON-2 study (NN-2301). Scientific reports, 15:3427, 2025
13. Kondo H, Ogura K, Morizane C, Satake T, Iwata S, Toda Y, Muramatsu S, Takemori T, Kobayashi E, Higashi T, Kawai A. Chondrosarcoma in Japan: an analytic study using population-based National Cancer Registry. Japanese journal of clinical oncology, 55:490-497, 2025
14. Ikeda M, Morizane C, Ueno M, Okusaka T, Ishii H, Furuse J. Systemic therapy for hepatocellular carcinoma, from the early to the advanced stage: a Japanese perspective. Japanese journal of clinical oncology, 55:465-476, 2025
15. Morizane C, Ueno M, Ikeda M, Okusaka T, Ishii H, Furuse J. Update for: New developments in systemic therapy for advanced biliary tract cancer. Japanese journal of clinical oncology, 55:210-218, 2025
16. Kobayashi S, Nakamura Y, Hashimoto T, Bando H, Oki E, Karasaki T, Horinouchi H, Ozaki Y, Iwata H, Kato T, Miyake H, Ohba A, Ikeda M, Chiyoda T, Hasegawa K, Fujisawa T, Matsuura K, Namikawa K, Yajima S, Yoshino T, Hasegawa K. Japan society of clinical oncology position paper on appropriate clinical use of molecular residual disease (MRD) testing. International journal of clinical oncology, 30:605-654, 2025
17. Imamura S, Watanabe K, Inoue K, Taira T, Shibuki T, Satake T, Yamaguchi S, Sasaki M, Imaoka H, Mitsunaga S, Ikeda M. Side-by-side versus stent-in-stent technique for stent deployment during systemic chemotherapy in biliary tract cancer patients with malignant hilar biliary obstruction. DEN open, 5:e70075, 2025
18. Shibuki T, Ikeda M, Yokoyama M, Sano Y, Furuse J, Kobayashi S, Ohba A, Todaka A, Horie Y, Shioji K, Kanai M, Nishina T, Kumamoto Y, Fujimori N, Katanuma A, Takayama Y, Tsumura H, Miwa H, Ozaka M, Ueno M. Depth of response in patients with locally advanced pancreatic cancer treated with first-line chemotherapy: A supplementary analysis of JCOG1407. Pancreatology, 25:275-283, 2025
19. Ikeda M, Okusaka T, Ueno M, Ozaka M, Satoi S, Skanji D, Martín-Fernández L, Amellal N, Furuse J. NALIRIFOX in Japanese treatment-naïve patients with metastatic pancreatic adenocarcinoma: an open-label, phase II trial design. Future oncology (London, England), 21:959-965, 2025
20. Toda Y, Ogura K, Morizane C, Satake T, Iwata S, Kobayashi E, Takemori T, Kondo H, Muramatsu S, Higashi T, Kawai A. Prognostic factors and management of elderly sarcoma in Japan: the population-based National Cancer Registry (NCR) in Japan. International journal of clinical oncology, 30:1018-1032, 2025
21. Araki T, Hayashi K, Shimokawa M, Otsuka T, Sonoda Y, Honda T, Shibuki T, Nakazawa J, Arima S, Miwa K, Koga F, Ueda Y, Kubotsu Y, Shimokawa H, Takeshita S, Nishikawa K, Komori A, Otsu S, Hosokawa A, Sakai T, Oda H, Kawahira M, Arita S, Taguchi H, Tsuneyoshi K, Fujita T, Sakae T, Kawaguchi Y, Shirakawa T, Mizuta T, Mitsugi K. Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302). Therapeutic advances in medical oncology, 17:17588359251320768, 2025
22. Fujisawa T, Hojo H, Nakamura M, Makita K, Hirata H, Oyoshi H, Tomizawa K, Zhou Y, Fukushi K, Ikeda M, Zenda S. Clinical Outcomes of Patients Receiving Stereotactic Body Radiotherapy Dose De-escalation for Hepatocellular Carcinoma at the Hepatic Hilum. Anticancer research, 45:1159-1169, 2025
23. Okada M, Suzuki E, Morizane C, Ogawa G, Sano Y, Imaoka H, Kobayashi S, Ikeda M, Okano N, Miwa H, Todaka A, Shimizu S, Mizuno N, Satoi S, Sano K, Tobimatsu K, Katanuma A, Masutomi K, Okusaka T, Ozaka M, Ueno M. Impact of clinically diagnosed liver cirrhosis in patients with intrahepatic cholangiocarcinoma treated with systemic chemotherapy: a subgroup analysis of JCOG1113. Japanese journal of clinical oncology, 55:594-602, 2025
24. Kodama Y, Ueshima K, Moriguchi M, Inaba Y, Yamashita T, Iwamoto H, Ueno M, Ogasawara S, Kuzuya T, Kodama T, Sato Y, Tada T, Tsuchiya K, Nishiofuku H, Yamakado K, Sone M, Ikeda M, Takehara T, Hamano T, Kudo M. Protocol of the IMPACT study: randomized, multicenter, phase 3 study evaluating the efficacy of immunotherapy (Atezolizumab) plus anti-VEGF therapy (Bevacizumab) in combination with transcatheter arterial chemoembolization for unresectable hepatocellular carcinoma. BMC cancer, 25:434, 2025
25. Takemori T, Ogura K, Morizane C, Satake T, Iwata S, Toda Y, Muramatsu S, Kondo H, Kobayashi E, Higashi T, Kawai A. Incidence and site specific characteristics of angiosarcoma in Japan using a population-based national cancer registry from 2016 to 2019. Scientific reports, 15:9960, 2025
26. Hoyek C, Pirozzi A, Zheng B, Lida N, Ramaker R, Abidoye S, Eslinger C, Shibuki T, Wu CSY, Sonbol MB, Ahn DH, Borad M, Ikeda M, Nakamura Y, Rimassa L, Yoshino T, Strickler J, Bekaii-Saab T. CLO25-095: Baseline Genomic Alterations as Predictors of Response to Anti-HER2 Therapy in HER2-Amplified Advanced Colorectal Cancer (aCRC). Journal of the National Comprehensive Cancer Network : JNCCN, 23:CLO25-095, 2025
27. Inoue K, Nakamura Y, Caughey B, Zheng-Lin B, Ueno M, Furukawa M, Kawamoto Y, Itoh S, Umemoto K, Sudo K, Satoh T, Mizuno N, Kajiwara T, Fujisawa T, Bando H, Yoshino T, Strickler JH, Morizane C, Bekaii-Saab T, Ikeda M. Clinicomolecular Profile and Efficacy of Human Epidermal Growth Factor Receptor 2 (HER2)-Targeted Therapy for HER2-Amplified Advanced Biliary Tract Cancer. JCO precision oncology, 9:e2400718, 2025
28. Fujisawa T, Nakamura Y, Bando H, Morizane C, Ikeda M, Nonomura N, Matsubara N, Iwata H, Naito Y, Okano S, Aoki D, Harano K, Yamazaki N, Namikawa K, Ueno M, Kadowaki S, Oki E, Kato K, Komatsu Y, Satoh T, Esaki T, Denda T, Hamaguchi T, Yamazaki K, Matsuhashi N, Yasui H, Satake H, Nishina T, Takahashi N, Goto M, Sunakawa Y, Kato T, Otsuka T, Abutani H, Tukachinsky H, Lee JK, Oxnard GR, Kuramoto N, Horasawa S, Sakamoto Y, Taniguchi H, Yoshino T. Benefits of Combining Circulating Tumor DNA With Tissue and Longitudinal Circulating Tumor DNA Genotyping in Advanced Solid Tumors: SCRUM-Japan MONSTAR-SCREEN-1 Study. JCO precision oncology, 9:e2400283, 2025
29. Hashimoto T, Nakamura Y, Mishima S, Nakayama I, Kotani D, Kawazoe A, Kuboki Y, Bando H, Kojima T, Iida N, Shibuki T, Imai M, Fujisawa T, Nagamine M, Sakamoto N, Kuwata T, Yoshino T, Shitara K. Whole-transcriptome sequencing in advanced gastric or gastroesophageal cancer: A deep dive into its clinical potential. Cancer science, 115:1622-1633, 2024
30. Shirakawa T, Makiyama A, Shimokawa M, Otsuka T, Shinohara Y, Koga F, Ueda Y, Nakazawa J, Otsu S, Komori A, Arima S, Fukahori M, Taguchi H, Honda T, Shibuki T, Nio K, Ide Y, Ureshino N, Mizuta T, Mitsugi K, Akashi K, Baba E. Author Correction: C-reactive protein/albumin ratio is the most significant inflammatory marker in unresectable pancreatic cancer treated with FOLFIRINOX or gemcitabine plus nab-paclitaxel. Scientific reports, 14:5311, 2024
31. Hashimoto T, Nakamura Y, Oki E, Kobayashi S, Yuda J, Shibuki T, Bando H, Yoshino T. Bridging horizons beyond CIRCULATE-Japan: a new paradigm in molecular residual disease detection via whole genome sequencing-based circulating tumor DNA assay. International journal of clinical oncology, 29:495-511, 2024
32. Kawamoto Y, Morizane C, Komatsu Y, Kondo S, Ueno M, Kobayashi S, Furukawa M, Lee L, Satoh T, Sakai D, Ikeda M, Imaoka H, Miura A, Hatanaka Y, Yokota I, Nakamura Y, Yoshino T. Phase II trial of niraparib for BRCA-mutated biliary tract, pancreatic and other gastrointestinal cancers: NIR-B. Future oncology (London, England), 20:1901-1907, 2024
33. Ikeda M, Hijioka S, Ito T, Matsumoto S, Honma Y, Ueno M, Okano N, Aoki T, Furuse J. Multicenter validation study of a treatment selection MAP for pancreatic neuroendocrine tumors. Japanese journal of clinical oncology, 54:880-886, 2024
34. Kodama T, Imajima T, Shimokawa M, Otsuka T, Kawahira M, Nakazawa J, Hori T, Shibuki T, Arima S, Ido A, Miwa K, Okabe Y, Koga F, Ueda Y, Kubotsu Y, Shimokawa H, Takeshita S, Nishikawa K, Komori A, Otsu S, Hosokawa A, Sakai T, Sakai K, Oda H, Kawahira M, Arita S, Honda T, Taguchi H, Tsuneyoshi K, Kawaguchi Y, Fujita T, Sakae T, Shirakawa T, Mizuta T, Mitsugi K. A multicenter retrospective observational NAPOLEON2 study of nanoliposomal irinotecan with fluorouracil and folinic acid in patients with unresectable pancreatic cancer. Scientific reports, 14:12422, 2024
35. Oh DY, He AR, Bouattour M, Okusaka T, Qin S, Chen LT, Kitano M, Lee CK, Kim JW, Chen MH, Suksombooncharoen T, Ikeda M, Lee MA, Chen JS, Potemski P, Burris HA 3rd, Ostwal V, Tanasanvimon S, Morizane C, Zaucha RE, McNamara MG, Avallone A, Cundom JE, Breder V, Tan B, Shimizu S, Tougeron D, Evesque L, Petrova M, Zhen DB, Gillmore R, Gupta VG, Dayyani F, Park JO, Buchschacher GL Jr, Rey F, Kim H, Wang J, Morgan C, Rokutanda N, Żotkiewicz M, Vogel A, Valle JW. Durvalumab or placebo plus gemcitabine and cisplatin in participants with advanced biliary tract cancer (TOPAZ-1): updated overall survival from a randomised phase 3 study. The lancet. Gastroenterology & hepatology, 9:694-704, 2024
36. Kobayashi K, Ogasawara S, Itobayashi E, Okubo T, Itokawa N, Nakamura K, Moriguchi M, Watanabe S, Ikeda M, Kuroda H, Kawaoka T, Hiraoka A, Yasui Y, Kuzuya T, Sato R, Kanzaki H, Koroki K, Inoue M, Nakamura M, Kiyono S, Kanogawa N, Kondo T, Nakamoto S, Ozawa Y, Tsuchiya K, Atsukawa M, Aikata H, Aramaki T, Oka S, Morimoto N, Kurosaki M, Itoh Y, Izumi N, Kato N. Ramucirumab for advanced hepatocellular carcinoma in the current real world: a Japanese single-arm study post-REACH-2 (The R-evolution study). Investigational new drugs, 42:394-404, 2024
37. Imaoka H, Sasaki K, Machida R, Nagano H, Satoi S, Ikeda M, Kobayashi S, Yamashita T, Okusaka T, Ido A, Hatano E, Miwa H, Ueno M, Nakao K, Shimizu S, Kuramochi H, Sakamori R, Tsumura H, Okano N, Shioji K, Shirakawa H, Akutsu N, Tsuji K, Ishii H, Umemoto K, Asagi A, Ueno M. Current status of the cost burden of first-line systemic treatment for patients with advanced hepatocellular carcinoma in Japan, 2021-22. Japanese journal of clinical oncology, 54:1071-1077, 2024
38. Ogura K, Morizane C, Satake T, Iwata S, Toda Y, Muramatsu S, Kobayashi E, Arakawa A, Ogawa C, Kato Y, Higashi T, Kawai A. Statistics of bone sarcoma in Japan: report from the population-based cancer registry in Japan. International journal of clinical oncology, 29:1209-1219, 2024
39. Sakamoto Y, Sakamoto T, Ohba A, Sasaki M, Kondo S, Morizane C, Ueno H, Saito Y, Arai Y, Okusaka T. Efficacy of hemostasis by gastroduodenal covered metal stent placement for hemorrhagic duodenal stenosis due to pancreatobiliary cancer invasion: a retrospective study. Clinical endoscopy, 57:628-636, 2024
40. Mitsunaga S, Okumura N, Takei T, Takao T, Tsubouchi H, Nakata K, Nakamura M, Kitahata Y, Motobayashi H, Ikeda M, Nakazato M. Identification of a urinary CD276 fragment for detecting resectable pancreatic cancer using a C-terminal proteomics strategy. Scientific reports, 14:14207, 2024
41. Ogura K, Morizane C, Satake T, Iwata S, Toda Y, Muramatsu S, Takemori T, Kondo H, Kobayashi E, Katoh Y, Higashi T, Kawai A. Soft-tissue sarcoma in Japan: National Cancer Registry-based analysis from 2016 to 2019. Japanese journal of clinical oncology, 54:1150-1157, 2024
42. Rimini M, Fornaro L, Rizzato MD, Antonuzzo L, Rossari F, Satake T, Vandeputte H, Vivaldi C, Pressiani T, Lucchetti J, Kim JW, Abidoye O, Rapposelli IG, Tamberi S, Finkelmeier F, Giordano G, Nichetti F, Chon HJ, Braconi C, Pirrone C, Castet F, Tamburini E, Yoo C, Parisi A, Diana A, Scartozzi M, Prager GW, Avallone A, Schirripa M, Kim IH, Perkhofer L, Oneda E, Verrico M, Adeva J, Chan SL, Spinelli GP, Personeni N, Garajova I, Rodriquenz MG, Leo S, Salani F, De Rosa A, Lavacchi D, Foti S, Ikeda M, Dekervel J, Niger M, Balsano R, Tonini G, Kang M, Bekaii-Saab T, Esposito L, Boccaccino A, Himmelsbach V, Landriscina M, Djaballah SA, Zanuso V, Masi G, Lonardi S, Rimassa L, Casadei-Gardini A. Durvalumab plus gemcitabine and cisplatin in advanced biliary tract cancer: A large real-life worldwide population. European journal of cancer (Oxford, England : 1990), 208:114199, 2024
43. Hashimoto T, Nakamura Y, Fujisawa T, Imai M, Shibuki T, Iida N, Ozaki H, Nonomura N, Morizane C, Iwata H, Okano S, Yamagami W, Yamazaki N, Kadowaki S, Taniguchi H, Ueno M, Boku S, Oki E, Komatsu Y, Yuki S, Makiyama A, Otsuka T, Hara H, Okano N, Nishina T, Sakamoto Y, Miki I, Kobayashi S, Yuda J, Kageyama SI, Nagamine M, Sakashita S, Sakamoto N, Yamashita R, Koga Y, Bando H, Ishii G, Kuwata T, Park WY, Ohtsu A, Yoshino T. The SCRUM-MONSTAR Cancer-Omics Ecosystem: Striving for a Quantum Leap in Precision Medicine. Cancer discovery, 14:2243-2261, 2024
44. Shibuki T, Otsuka T, Shimokawa M, Nakazawa J, Arima S, Fukahori M, Miwa K, Okabe Y, Koga F, Ueda Y, Kubotsu Y, Makiyama A, Shimokawa H, Takeshita S, Nishikawa K, Komori A, Otsu S, Hosokawa A, Sakai T, Oda H, Kawahira M, Arita S, Honda T, Taguchi H, Tsuneyoshi K, Kawaguchi Y, Fujita T, Sakae T, Nio K, Ide Y, Ureshino N, Shirakawa T, Mizuta T, Mitsugi K. Nanoliposomal irinotecan with fluorouracil and folinic acid, FOLFIRINOX, and S-1 as second-line treatment for unresectable pancreatic cancer after gemcitabine/nab-paclitaxel. Scientific reports, 14:16906, 2024
45. Yamamoto N, Kitano S, Koyama T, Ikeda M, Mizugaki H, Narikiyo T, Yamaguchi Y, Ishida T, Takubo R, Ogami C, Sekiya M, Nakagawa Y, Kuboki Y. Phase I study of the safety and clinical activity of the interleukin-8 inhibitor AMY109 combined with atezolizumab in patients with advanced solid cancers. Journal for immunotherapy of cancer, 12:e009262, 2024
46. Furuse J, Ikeda M, Ueno M, Furukawa M, Morizane C, Takehara T, Nishina T, Todaka A, Okano N, Hara K, Nakai Y, Ohkawa K, Sasaki T, Sugimori K, Yokoyama N, Yamamoto K. A Phase II Placebo-Controlled Study of the Effect and Safety of Nanvuranlat in Patients with Advanced Biliary Tract Cancers Previously Treated by Systemic Chemotherapy. Clinical cancer research, 30:3990-3995, 2024
47. Tezuka S, Ozaka M, Furuse J, Yokoyama M, Uemura K, Sano Y, Nakachi K, Imaoka H, Unno M, Shirakawa H, Shimizu S, Kato N, Kojima Y, Sano K, Kobayashi S, Terashima T, Morizane C, Ikeda M, Ueno M. Early tumor shrinkage as a prognostic predictor in chemotherapy-naïve patients with locally advanced pancreatic cancer treated with modified FOLFIRINOX or gemcitabine plus nab-paclitaxel combination therapy: An exploratory analysis of JCOG1407. Pancreatology, 24:909-916, 2024
48. Ohba A, Morizane C, Kawamoto Y, Komatsu Y, Ueno M, Kobayashi S, Ikeda M, Sasaki M, Furuse J, Okano N, Hiraoka N, Yoshida H, Kuchiba A, Sadachi R, Nakamura K, Matsui N, Nakamura Y, Okamoto W, Yoshino T, Okusaka T. Trastuzumab Deruxtecan in Human Epidermal Growth Factor Receptor 2-Expressing Biliary Tract Cancer (HERB; NCCH1805): A Multicenter, Single-Arm, Phase II Trial. Journal of clinical oncology, 42:3207-3217, 2024
49. Takemori T, Ogura K, Morizane C, Satake T, Iwata S, Toda Y, Muramatsu S, Kondo H, Kobayashi E, Higashi T, Kawai A. Clear cell sarcoma in Japan: an analysis of the population-based cancer registry in Japan. Japanese journal of clinical oncology, 54:1281-1287, 2024
50. Sudo K, Nakamura Y, Ueno M, Furukawa M, Mizuno N, Kawamoto Y, Okano N, Umemoto K, Asagi A, Ozaka M, Ohtsubo K, Shimizu S, Matsuhashi N, Itoh S, Matsumoto T, Satoh T, Okuyama H, Goto M, Hasegawa H, Yamamoto Y, Odegaard JI, Bando H, Yoshino T, Ikeda M, Morizane C. Clinical utility of BRCA and ATM mutation status in circulating tumour DNA for treatment selection in advanced pancreatic cancer. British journal of cancer, 131:1237-1245, 2024
51. Mitsunaga S, Ikeda M, Nomura S, Morizane C, Todaka A, Yamamoto N, Kamata K, Yanagibashi H, Mizuno N, Kawamoto Y, Gotoh K, Shirakawa H, Okano N, Nomura T, Tanaka K, Takahashi A, Yagi S, Ohta K, Takayama Y, Miwa H, Nagano H, Kojima Y, Hisano T, Tahara M, Sakuma Y, Arai H, Nakamura I, Katayama H, Konishi M, Ueno M. 5-Fluorouracil metabolic pathway genes predict recurrence risk following adjuvant S-1 therapy: Results of an ancillary analysis from a phase III trial of resected biliary tract cancer (JCOG1202A1). Journal of hepato-biliary-pancreatic sciences, 31:886-896, 2024
52. Araki T, Kawahira M, Shimokawa M, Otsuka T, Hayashi K, Sonoda Y, Honda T, Nakao K, Shibuki T, Nakazawa J, Arima S, Fukahori M, Miwa K, Koga F, Ueda Y, Kubotsu Y, Makiyama A, Shimokawa H, Takeshita S, Nishikawa K, Komori A, Otsu S, Hosokawa A, Sakai T, Oda H, Arita S, Taguchi H, Tsuneyoshi K, Kawaguchi Y, Fujita T, Sakae T, Nio K, Ide Y, Ureshino N, Shirakawa T, Mizuta T, Mitsugi K. Real-World Analysis of the Correlation between Overall Survival and Progression-Free Survival in Advanced Pancreatic Cancer: Results of NAPOLEON-1 and 2 Studies. Oncology, 1-11, 2024
53. Morizane C, Ueno M, Ikeda M, Sudo K, Hirashima Y, Kuroda M, Ueno S, Okusaka T, Furuse J. A Phase 2 study of nivolumab in combination with modified FOLFIRINOX for metastatic pancreatic cancer. BJC reports, 2:3, 2024
54. Igata Y, Kudo M, Kojima M, Kami S, Aoki K, Satake T, Kobayashi T, Sugimoto M, Kobayashi S, Konishi M, Gotohda N. Conversion surgery after gemcitabine and cisplatin plus durvalumab for advanced intrahepatic cholangiocarcinoma: A case report. World journal of clinical cases, 12:6721-6727, 2024
55. Satake T, Morizane C, Okada M, Nishioka M, Hiraoka N, Nara S, Kakegawa T, Kobayashi M, Koyama K, Esaki M, Okusaka T. Prevalence of HER3 Expression in Pancreatic Cancer Patients Treated With Systemic Chemotherapy. Cancer medicine, 13:e70474, 2024
56. Kudo M, Tsuchiya K, Yamashita T, Koga H, Nakagawa Y, Ikeda M. Reply to the Letter Regarding "Impact of Bevacizumab Being Skipped due to Adverse Events of Special Interest for Bevacizumab in Patients with Unresectable Hepatocellular Carcinoma Treated with Atezolizumab plus Bevacizumab: An Exploratory Analysis of the Phase III IMbrave150 Study". Liver cancer, 13:671-673, 2024
57. Baba S, Kawasaki T, Hirano S, Nakamura T, Asano T, Okazaki R, Yoshida K, Kawase T, Kurahara H, Oi H, Yokoyama M, Kita J, Imura J, Kinoshita K, Kondo S, Okada M, Satake T, Igawa YS, Yoshida T, Yamaguchi H, Ando Y, Mizunuma M, Ichikawa Y, Hida K, Nishihara H, Kato Y. A noninvasive urinary microRNA-based assay for the detection of pancreatic cancer from early to late stages: a case control study. EClinicalMedicine, 78:102936, 2024
