Annual Report 2024
Department of Genetic Medicine and Services
Takeshi Kuwata, Katsuya Tsuchihara, Toru Mukohara, Kiwamu Akagi, Mitsuho Imai, Akiko Nakayama, Sachiyo Mimaki, Yumie Hiraoka, Miki Aitani, Kenichi Harano, Shingo Matsumoto, Yuki Matsubara, Kyoko Toju, Nobuyuki Nakamura, Izumi Miki, Kaede Baba, Hiroki Sato, Yoshiko Onuma
Introduction
The Department of Genetic Medicine and Services was newly established in May 2016 to handle genetic as well as genomic testing and related issues, including genetic counseling, conducted in the National Cancer Center Hospital East (NCCHE).
The Team and What We Do
The aim of the Department of Genetic Medicine and Services is to deal with the following subjects:
1) Genetic counseling
2) Genetic/genomic testing
3) Ethical issues related to genetic/genomic medicine
4) Educating people on genetic/genomic medicine
5) Regulating the storage and usage of genetic/genomic information
6) Other matters related to genetic/genomic medicine
Accordingly, members of the department with various specialties were assembled, including medical doctors, nurses, genetic counselors and medical technicians from different clinical departments or research laboratories.
To conduct precision oncology, our hospital is designated as a Cancer Genome Medical Core Hospital in Japan and cooperates with 12 affiliated hospitals, especially for providing cancer genome profiling (CGP) tests. Under the national health care services, by March 2025, we performed 469 CGP tests and conducted expert panels for 462 patients, including 526 patients in affiliated hospitals (Figure 1).
The Outpatient Genetic Counseling Clinic provides genetic counseling and genetic testing for cancer patients and their relatives with a familial history and/or specific features suspected for familial cancers. From April 2024 to March 2025, 96 new clients visited the clinic, and a total of 215genetic counseling sessions were held and 52 genetic testing sessions were provided. Consequently, by March 2025, a total of 905 clients had visited the clinic (Figures 2 and 3).
Fig 1. Number of CGP cases reviewed in expert panels 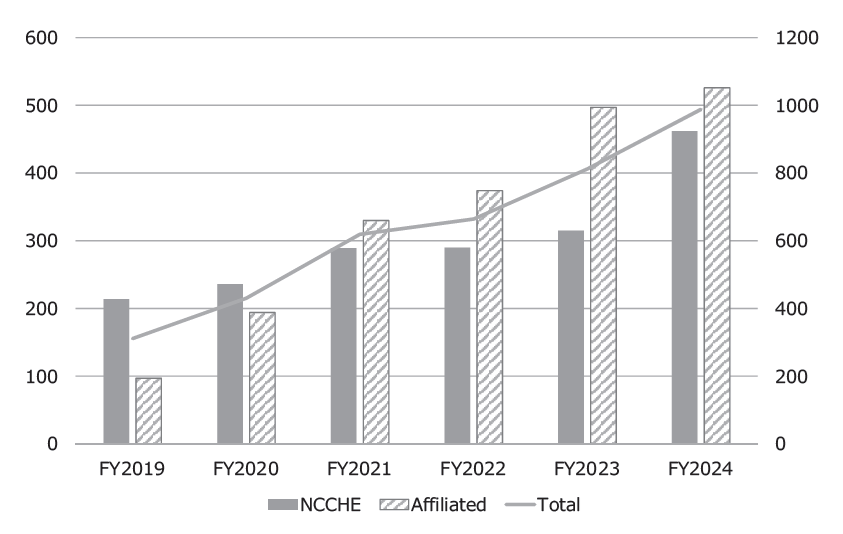

Fig 2. Number of genetic counseling sessions provided 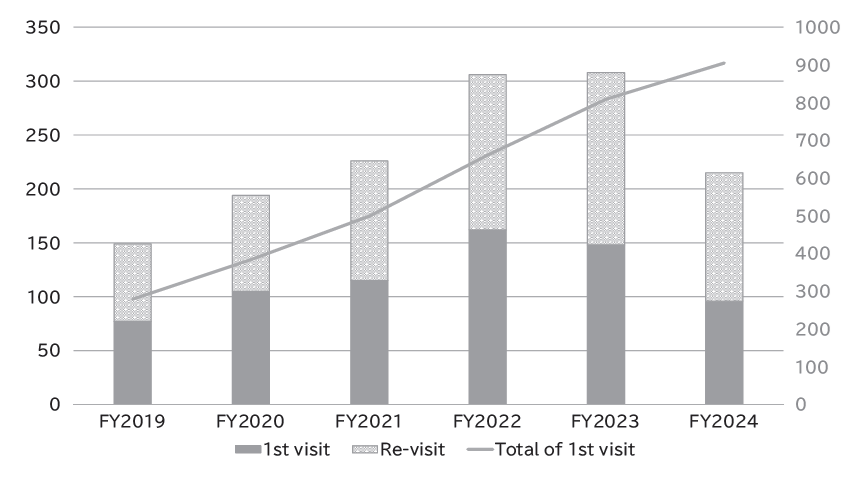

Fig 3. Breakdown of genetic counseling sessions in FY2024 (N=96)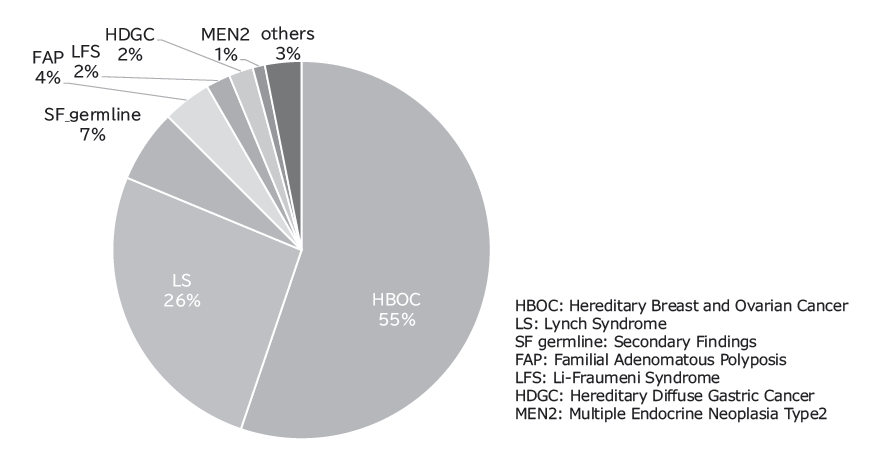

Research Activities
Our Outpatient Genetic Counseling Clinic is participating in an observational study to evaluate the utility of confirmation of germline mutations in patients with solid tumors and relatives (BRANCH study, UMIN000046085), and provides genetic testing.
Clinical Trials
Our Outpatient Genetic Counseling Clinic supports and provides genetic counseling for patients willing to participate in clinical trials where genetic tests are required.
Education
We educated medical doctors wishing to become board-certified geneticists by attending the Outpatient Genetic Counseling Clinic. We have also provided intramural educational seminars for medical doctors and medical staff to familiarize them with genetic and genomic medicine and provide precision medicine for all cancer patients and their families visiting our hospital.
As a Cancer Genome Medical Core Hospital, we also provide seminars and lectures for educating doctors as well as medical staff working at affiliated hospitals.
Future Prospects
We will continue to provide genetic counseling and genetic testing for possible familial cancer patients/families. For precision oncology, we will conduct CGP tests under the national health care system and participate in several clinical trials using new technologies including whole genome/exome sequences. We will also continue to provide education programs for medical doctors and medical staff working in our hospital as well as affiliated hospitals. Our current aim is to establish a genomic testing pipeline from research to clinic to accelerate the development of medical and diagnostic devices for genome medicine.
List of papers published in 2024
Journal
1. Konno Y, Mayama M, Takehara K, Yokoyama Y, Suzuki J, Susumu N, Harano K, Nakagawa S, Nakanishi T, Yamagami W, Yoshihara K, Nomura H, Okamoto A, Aoki D, Watari H. Prognostic impact of the number of resected pelvic nodes in endometrial cancer: Japanese Gynecologic Oncology Group Study JGOG2043 post hoc analysis. Journal of gynecologic oncology, 36:e3, 2025
2. Okazawa-Sakai M, Sakai SA, Hyodo I, Horasawa S, Sawada K, Fujisawa T, Yamamoto Y, Boku S, Hayasaki Y, Isobe M, Shintani D, Hasegawa K, Egawa-Takata T, Ito K, Ihira K, Watari H, Takehara K, Yagi H, Kato K, Chiyoda T, Harano K, Nakamura Y, Yamashita R, Yoshino T, Aoki D. Gut microbiome associated with PARP inhibitor efficacy in patients with ovarian cancer. Journal of gynecologic oncology, 36:e38, 2025
3. Iwata H, Naito Y, Hattori M, Yoshimura A, Yonemori K, Aizawa M, Mori Y, Yoshimitsu J, Umeyama Y, Mukohara T. Safety and pharmacokinetics of vepdegestrant in Japanese patients with ER+ advanced breast cancer: a phase 1 study. International journal of clinical oncology, 30:72-82, 2025
4. Abe H, Kuwata T, Kushima R, Ushiku T. Nationwide survey on HER2 and PD-L1 testing practices in gastric cancer across Japan. Gastric cancer, 28:294-300, 2025
5. Nagata H, Kinoshita T, Sakashita S, Kojima M, Taki T, Kuwata T, Yura M, Shitara K, Ishii G, Sakamoto N. Area of Residual Tumor Measurement After Preoperative Chemotherapy as an Objective and Quantitative Method for Predicting the Prognosis of Gastric Cancer: A Single-Center Retrospective Study. World journal of surgery, 49:717-726, 2025
6. Imai M, Nakamura Y, Shin S, Okamoto W, Kato T, Esaki T, Kato K, Komatsu Y, Yuki S, Masuishi T, Nishina T, Sawada K, Sato A, Kuwata T, Yamashita R, Fujisawa T, Bando H, Ock CY, Fujii S, Yoshino T. Artificial Intelligence-Powered Human Epidermal Growth Factor Receptor 2 and Tumor Microenvironment Analysis in Human Epidermal Growth Factor Receptor 2-Amplified Metastatic Colorectal Cancer: Exploratory Analysis of Phase II TRIUMPH Trial. JCO precision oncology, 9:e2400385, 2025
7. Konno Y, Mayama M, Takehara K, Yokoyama Y, Suzuki J, Susumu N, Harano K, Nakagawa S, Nakanishi T, Yamagami W, Yoshihara K, Nomura H, Okamoto A, Aoki D, Watari H. Prognostic significance of para-aortic node metastasis in endometrial cancer: Japanese Gynecologic Oncology Group Study JGOG2043 post hoc analysis. Journal of gynecologic oncology, 2025
8. Takahashi K, Uozumi R, Mukohara T, Hayashida T, Iwabe M, Iihara H, Kusuhara-Mamishin K, Kitagawa Y, Tsuchiya M, Kitahora M, Nagayama A, Kosaka S, Asano-Niwa Y, Seki T, Ohnuki K, Suzuki A, Ono F, Futamura M, Kawazoe H, Nakamura T. Proton Pump Inhibitors and Cyclin-Dependent Kinase 4/6 Inhibitors in Patients With Breast Cancer. The oncologist, 29:e741-e749, 2024
9. Doi T, Takahashi S, Aoki D, Yonemori K, Hara H, Hasegawa K, Takehara K, Harano K, Yunokawa M, Nomura H, Shimoi T, Horie K, Ogasawara A, Okame S. A first-in-human phase I study of TAS-117, an allosteric AKT inhibitor, in patients with advanced solid tumors. Cancer chemotherapy and pharmacology, 93:605-616, 2024
10. Kuwata T. Molecular classification and intratumoral heterogeneity of gastric adenocarcinoma. Pathology international, 74:301-316, 2024
11. Bardia A, Krop IE, Kogawa T, Juric D, Tolcher AW, Hamilton EP, Mukohara T, Lisberg A, Shimizu T, Spira AI, Tsurutani J, Damodaran S, Papadopoulos KP, Greenberg J, Kobayashi F, Zebger-Gong H, Wong R, Kawasaki Y, Nakamura T, Meric-Bernstam F. Datopotamab Deruxtecan in Advanced or Metastatic HR+/HER2- and Triple-Negative Breast Cancer: Results From the Phase I TROPION-PanTumor01 Study. Journal of clinical oncology, 42:2281-2294, 2024
12. Kakuwa T, Watanabe T, Niino M, Kawata A, Satoh T, Matsumura N, Yokoyama Y, Kawana K, Hirashima Y, Kyo S, Yasuda M, Harano K, Machida H, Tokunaga H, Kaneuchi M, Tabata T, Kobayashi Y, Nagase S, Katabuchi H, Mikami M, Yamamoto Y, Rikitake R, Ichinose Y, Higashi T. Quality of care measurement for patients with ovarian cancer in Japan. The journal of obstetrics and gynaecology research, 50:1182-1191, 2024
13. Mukohara T, Park YH, Sommerhalder D, Yonemori K, Hamilton E, Kim SB, Kim JH, Iwata H, Yamashita T, Layman RM, Mita M, Clay T, Chae YS, Oakman C, Yan F, Kim GM, Im SA, Lindeman GJ, Rugo HS, Liyanage M, Saul M, Le Corre C, Skoura A, Liu L, Li M, LoRusso PM. Inhibition of lysine acetyltransferase KAT6 in ER(+)HER2(-) metastatic breast cancer: a phase 1 trial. Nature medicine, 30:2242-2250, 2024
14. Yamamoto N, Kuboki Y, Harano K, Koyama T, Kondo S, Hagiwara A, Suzuki N, Fujikawa E, Toyoizumi K, Mukai M, Doi T. A phase 1/1b, open-label, dose-escalation study of PD-1 inhibitor, cetrelimab alone and in combination with FGFR inhibitor, erdafitinib in Japanese patients with advanced solid tumors. Investigational new drugs, 42:376-385, 2024
15. Mukohara T, Park YH, Sommerhalder D, Yonemori K, Hamilton E, Kim SB, Kim JH, Iwata H, Yamashita T, Layman RM, Mita M, Clay T, Chae YS, Oakman C, Yan F, Kim GM, Im SA, Lindeman GJ, Rugo HS, Liyanage M, Saul M, Le Corre C, Skoura A, Liu L, Li M, LoRusso PM. Author Correction: Inhibition of lysine acetyltransferase KAT6 in ER(+)HER2(-) metastatic breast cancer: a phase 1 trial. Nature medicine, 30:2371, 2024
16. Hashimoto T, Nakamura Y, Fujisawa T, Imai M, Shibuki T, Iida N, Ozaki H, Nonomura N, Morizane C, Iwata H, Okano S, Yamagami W, Yamazaki N, Kadowaki S, Taniguchi H, Ueno M, Boku S, Oki E, Komatsu Y, Yuki S, Makiyama A, Otsuka T, Hara H, Okano N, Nishina T, Sakamoto Y, Miki I, Kobayashi S, Yuda J, Kageyama SI, Nagamine M, Sakashita S, Sakamoto N, Yamashita R, Koga Y, Bando H, Ishii G, Kuwata T, Park WY, Ohtsu A, Yoshino T. The SCRUM-MONSTAR Cancer-Omics Ecosystem: Striving for a Quantum Leap in Precision Medicine. Cancer discovery, 14:2243-2261, 2024
17. Fassan M, Kuwata T, Matkowskyj KA, Röcken C, Rüschoff J. Claudin-18.2 Immunohistochemical Evaluation in Gastric and Gastroesophageal Junction Adenocarcinomas to Direct Targeted Therapy: A Practical Approach. Modern pathology, 37:100589, 2024
18. Colombo N, Biagioli E, Harano K, Galli F, Hudson E, Antill Y, Choi CH, Rabaglio M, Marmé F, Marth C, Parma G, Fariñas-Madrid L, Nishio S, Allan K, Lee YC, Piovano E, Pardo B, Nakagawa S, McQueen J, Zamagni C, Manso L, Takehara K, Tasca G, Ferrero A, Tognon G, Lissoni AA, Petrella M, Laudani ME, Rulli E, Uggeri S, Barretina Ginesta MP. Atezolizumab and chemotherapy for advanced or recurrent endometrial cancer (AtTEnd): a randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet. Oncology, 25:1135-1146, 2024
19. Fukuda M, Mukohara T, Kuwata T, Sunami K, Naito Y. Efficacy of Trametinib in Neurofibromatosis Type 1-Associated Gastrointestinal Stromal Tumors: A Case Report. JCO precision oncology, 8:e2300649, 2024
20. Kitaoka T, Harada K, Sakashita S, Kojima M, Taki T, Kuwata T, Kinoshita T, Futakuchi M, Ishii G, Sakamoto N. Quantification of Gremlin 1 throughout the tumor stroma using whole slide imaging and its clinicopathological significance in gastric cancer. Virchows Archiv, 485:1107-1116, 2024
21. Aoki D, Tabata T, Yanagida S, Nakamura T, Kondo E, Hamanishi J, Harano K, Hasegawa K, Hirasawa T, Hori K, Komiyama S, Matsuura M, Nakai H, Nakamura H, Sakata J, Takehara K, Takekuma M, Yokoyama Y, Kase Y, Sumino S, Soeda J, Kato A, Suri A, Okamoto A, Sugiyama T. Niraparib in Japanese patients with heavily pretreated, homologous recombination-deficient ovarian cancer: final results of a multicenter phase 2 study. Journal of gynecologic oncology, 35:e114, 2024
22. Nakao T, Harano K, Wakabayashi M, Naito Y, Tanabe H, Mukohara T. Efficacy of a platinum-based chemotherapy rechallenge for platinum-sensitive recurrence after PARP inhibitor maintenance. Gynecologic oncology reports, 55:101482, 2024
23. Watanabe M, Haeno H, Mimaki S, Tsuchihara K. Multistage carcinogenesis in occupational cholangiocarcinoma: the impact of clonal expansion and risk estimation. Genes and environment, 46:21, 2024
24. Hasegawa S, Shoji Y, Kato M, Elzawahry A, Nagai M, Gi M, Suzuki S, Wanibuchi H, Mimaki S, Tsuchihara K, Totsuka Y. Whole Genome Sequencing Analysis of Model Organisms Elucidates the Association Between Environmental Factors and Human Cancer Development. International journal of molecular sciences, 25:11191, 2024
Book
1. Nakajima H, Mukohara T. Understanding mechanisms of resistance to HER2-targeted therapies in HER2-positive breast cancer. In: Bonavida B (ed), Overcoming Cancers Resistant to HER-2 Antibodies: Breaking Tolerance to Antibody-Mediated Immunotherapy, United Kingdom, Academic Press, pp 45-56, 2024
