Annual Report 2024
Department of Clinical Laboratories
Hiroshi Yamakawa, Masahiro Karibe, Narumi Akimoto, Yuko Adegawa, Momoko Iguchi, Kenta Akie, Michiteru Yamagishi, Atsuko Uno, Masaki Takeda, Takuya Aiba, Mika Narikiyo, Nobuyuki Nakamura, Go Sato, Masayuki Ito, Riko Nakamura, Yuuki Takano, Masako Naito, Mitsunori Tajima, Yui Jinbo, Saki Nakamura, Keisei Nakamura, Kimihiko Kawamura, Yasuharu Hashimoto, Mika Sasanuma, Momoka Sakayori, Haruka Nozaki, Ayaka Takahashi, Madoka Nakayama, Mana Shimamura, Fubuki Omoya, Keiko Nakai, Ryota Utsumi, Atsuko Suzuki, Kozue Saito, Momoko Murata, Nodoka Tanago, Sayuri Shibayama, Tomomi Wachi, Masayuki Sukegawa, Takaki Kobayashi, Katsuhiro Sato, Kentaro Yamada, Yuichiro Yazaki, Shota Oishi, Yuuma Furuya, Hikari Sekiguchi, Jun Ikoma, Masahiro Inoue, Aiko Kimura, Ikumi Komatsuda, Keiko Katsuno, Yumi Takeuchi, Ayumi Iwaya, Aya Koike, Tomoe Katakura, Kaori Toyama, Ayaka Ohashi, Yoriko Furusawa, Michiko Iida, Izumi Suzuki, Sachiko Fukuda, Megumi Yamaguchi, Mariko Kinoshita, Reiji Hoshi, Yui Muto, Konomi Nakazawa, Yukie Nakatani, Yuta Arai, Mizuki Kuniya, Hiromi Kimura, Haruhiko Oda, Kengo Oshima, Yutaka Yoshida, Misaki Yoda, Ayumi Takematsu, Megumi Tateishi, Megumi Shimada, Raika Miyahira, Kazuma Kougo, Eriko Nakayama, Yoshiko Oda
Introduction
In delivering the best possible cancer treatment and prevention to all people through collaboration with society, the Department of Clinical Laboratories is committed to “providing the best cancer treatment,” “research, development and dissemination of new medical care,” and “supporting Japan's efforts through education and training.” We carry out activities to improve medical care and disseminate information domestically and internationally. We provide the best clinical tests for patients at the Clinical Laboratory of Hospital East through three pillars: medical support, research, and education.
The Team and What We Do
The total number of clinical tests in FY2024 was 4,864,536, an increase of approximately 2.8% from the previous year, and is increasing year by year (Table 1).
Table 1. Number of clinical tests performed in FY2024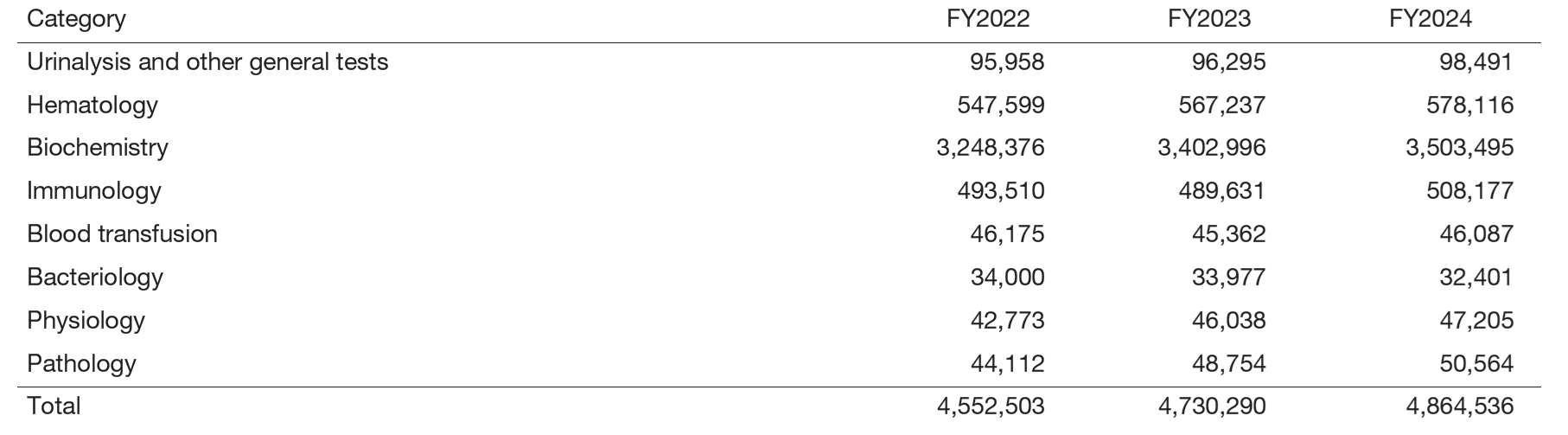

The number of clinical trial tests was 116% for sample processing (PK), 115% for electrocardiogram tests, and 110% for pathology specimen preparation compared to the previous year (Table 2).
Table 2. Number of clinical trial tests performed in FY2024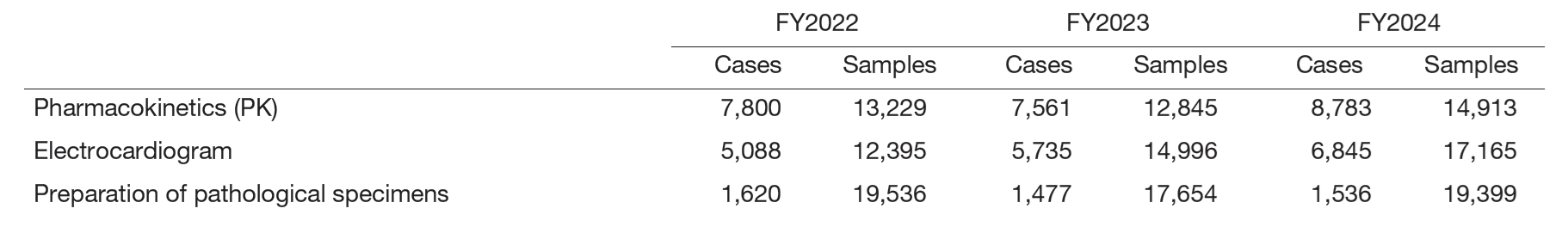

In August 2022, the facility obtained CAP accreditation (College of American Pathologists).
By obtaining CAP accreditation, the facility's clinical test data can be statistically compared with laboratories around the world, and by recognizing the internationally mainstream testing methods, the facility provides more accurate test data.
Research Activities
Active introduction of LBC (Liquid-based cytology)
The introduction of LBC has resulted in shorter screening times, improved cell collection rates, and reduced technical errors. The introduction of LBC has reduced the number of cases determined to be false positives
Education
We actively support employees in obtaining certified qualifications to improve their academic knowledge and skills.
In fiscal year 2024, one staff member passed the certified clinical genetic chromosome technologist examination, three passed the second-class clinical laboratory technologist (pathology, hematology, and immunoserology) examination, and one passed the first-class electrocardiogram certification examination.
Since October 2022, one employee has been job-rotated to the Department of Medical Information to gain experience in another field. This has produced positive results, as the system can now be built smoothly within the testing department and network problems can be handled smoothly.
Eight clinical technologist trainees have been accepted from four facilities.
Future Prospects
As a clinical testing facility that has acquired the international standard ISO15189 certification and CAP accreditation (College of American Pathologists), the facility provides support for medical treatment and clinical research at the Hospital East. In the future, the facility will strengthen collaboration with other departments at the Hospital East and the Exploratory Oncology Research & Clinical Trial Center, and build a system that will enable it to participate in the development of self-regulated tests (LDTs). The facility will contribute to the realization of Hospital East's vision of “providing the world's best cancer medical care and creating new world-class cancer medical care.”
