Annual Report 2024
Department of Medical Oncology
Kan Yonemori, Tatsunori Shimoi, Kazuki Sudo, Tadaaki Nishikawa, Yuki Kojima, Aiko Maejima, Shosuke Kita, Munehiro Ito, Ayumi Saito, Mai Hoshino, Eijiro Nakamura, Hitomi Sumiyoshi-Okuma, Asuka Kawachi,Rui Kitadai, Taro Yamanaka, Yasuhiro Fujiwara, Emi Noguchi,
Introduction
The Department of Medical Oncology provides the most effective treatments using chemotherapy and works on the establishment of new standards of care for adult malignancies including breast cancer, gynecologic cancer, urological cancer, soft-tissue sarcoma, extra-gonadal germ cell tumor, cancer of unknown primary site and other rare types of solid tumors.
We envision becoming a leading medical oncology department, which makes a difference in cancer care in Japan and worldwide. Our mission is to provide patient-centered, state-of-the-art medical care to cancer patients, to develop new effective cancer treatments through clinical and translational research, and to nurture medical oncologists. An evidence-based, research-oriented and multidisciplinary approach is the core value of our practice.
The Team and What We Do
The Department of Medical Oncology conducts clinical, research and educational activities with a focus on pharmacotherapy for breast, uterine, ovarian, urological cancers, malignant soft tissue tumors (e.g. sarcomas), germ cell tumors, cancers of unknown primary, and other rare malignancies (Table 1).
Table 1. New Patients in the Department of Medical Oncology (2024.4-2025.3)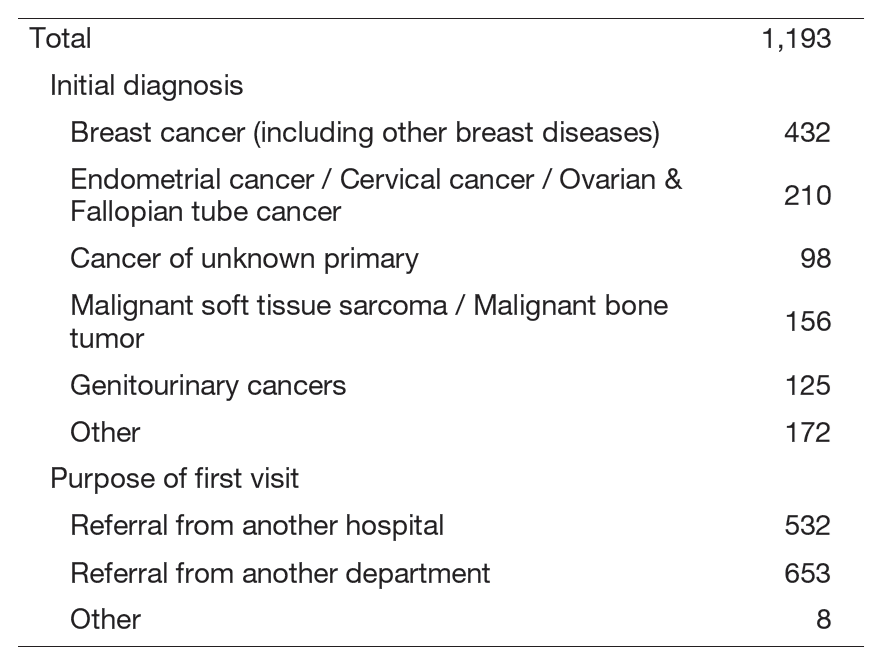

The five basic missions are as follows
- Contribute to the development of new cancer therapies domestically and globally by planning, supervising or participating in clinical trials.
- Promote translational research and strengthen cooperation with other departments, other professions and research centers.
- Lead the world in the development of treatments for breast cancers, gynecological cancers, urological cancers and sarcoma, and in the early clinical development of anti-cancer drugs.
- Educate and train specialists in cancer pharmacotherapy or breast cancer treatment (medical specialists).
- Understand the physical, mental and social suffering of cancer patients and aim for holistic medical care.
In addition to providing the latest evidence-based pharmacotherapy, the department focuses on research activities aimed at building new evidence, focusing on the social environment surrounding cancer care, and developing treatments for rare cancers.
Research Activities
Our research interests extend across a wide range of topics related to treatment and clinical program development. Many of our research programs are secured by public and consignment research grants. We conducted many clinical studies as a primary investigator and participated in additional programs as a co-investigator in research programs secured by competitive public research funds. We published 68 international manuscripts, focusing on early-phase anti-cancer drug development, molecular imaging, drug efficacy studies using patient derived xenografts, translational research, novel chemotherapy against breast and gynecological cancer, novel biomarkers to predict the efficacy and adverse events of anti-cancer drugs and other basic research. We value cancer survivorship as a research theme to develop a comprehensive patient-centered care program.
Clinical Trials
In 2024, we actively enrolled patients in phase I studies (including first in human or global) as well as domestic and international phase II and III studies. Of note we enrolled patients in IIT for rare cancers. We also conducted many types of translational studies (TR) to identify novel biomarkers.
Education
We provide rich educational opportunities to both residents and chief residents through clinical experience as well as research activities. Residents are encouraged to make presentations at local and national conferences. We vigorously support basic, clinical, and translational research conducted by postdoctoral researchers.
Future Prospects
We will continue to establish new standard treatments and propose a near-future model for the clinical management of adult solid tumors, including breast cancer, gynecologic cancer, genitourinary cancer and rare cancer. Moreover, we aim to build a comprehensive program, which includes tumor registry, translational research, clinical trials and patient care in rare adult tumors based on our rich clinical experience. We would also like to improve the efficiency of anti-cancer drug development by coordinating basic and translational research in early-phase clinical trials.
List of papers published in 2024
Journal
1. Yazaki S, Chiba Y, Kojima Y, Yoshida H, Takamizawa S, Kitadai R, Saito A, Kita S, Yamamoto K, Sumiyoshi-Okuma H, Nishikawa T, Sudo K, Shimoi T, Noguchi E, Uno M, Ishikawa M, Kato T, Fujiwara Y, Yonemori K. Folate Receptor Alpha Expression and the Tumor Immune Microenvironment in Patients with Cervical Cancer. International journal of gynecological pathology, 44:104-111, 2025
2. Mizoguchi C, Nishikawa T, Yoshida H, Yasuda M, Kato T, Hasegawa K, Yonemori K. HER2-negative or low expression as an unfavorable prognostic factor in patients with stage I/II uterine carcinosarcoma. Journal of gynecologic oncology, 36:e14, 2025
3. Iwata H, Naito Y, Hattori M, Yoshimura A, Yonemori K, Aizawa M, Mori Y, Yoshimitsu J, Umeyama Y, Mukohara T. Safety and pharmacokinetics of vepdegestrant in Japanese patients with ER+ advanced breast cancer: a phase 1 study. International journal of clinical oncology, 30:72-82, 2025
4. Hirakawa A, Asakawa T, Tokushige K, Ozaki R, Yoshida M, Okuma HS, Saito S, Shimizu Y, Kitabayashi R, Hanazawa R, Sato H, Uemura Y. Planning and Implementing Master Protocol Trials in Japan: Key Considerations of the Japanese Guideline. Clinical pharmacology and therapeutics, 117:627-632, 2025
5. Takahashi Y, Yoshida A, Yoshimoto S, Suzuki S, Kishikawa S, Mitsui A, Ryo E, Kojima Y, Yonemori K, Yatabe Y, Mori T. TFE3-rearranged perivascular epithelioid cell tumors of the head and neck with rare fusion partners: clues to the differential diagnosis between benign and malignant tumors. Diagnostic pathology, 20:7, 2025
6. Okines AFC, Curigliano G, Mizuno N, Oh DY, Rorive A, Soliman H, Takahashi S, Bekaii-Saab T, Burkard ME, Chung KY, Debruyne PR, Fox JR, Gambardella V, Gil-Martin M, Hamilton EP, Monk BJ, Nakamura Y, Nguyen D, O'Malley DM, Olawaiye AB, Pothuri B, Reck M, Sudo K, Sunakawa Y, Van Marcke C, Yu EY, Ramos J, Tan S, Bieda M, Stinchcombe TE, Pohlmann PR. Tucatinib and trastuzumab in HER2-mutated metastatic breast cancer: a phase 2 basket trial. Nature medicine, 31:909-916, 2025
7. Hayato S, Hamuro L, Shimizu T, Yonemori K, Nishio S, Yunokawa M, Yoshida T, Nishio M, Matsumoto K, Takehara K, Hasegawa K, Kozuki T, Hirashima Y, Kato H, Miura T, Nomoto M, Zhao Y, Zhu L, Yasuda S. Pharmacokinetic and Exposure-Response Modeling Support Body Surface Area-Based Dosing of Farletuzumab Ecteribulin in Japanese Patients with Solid Tumors. Journal of clinical pharmacology, 65:751-762, 2025
8. Nishimura R, Sudo K, Kitadai R, Kawachi A, Ito M, Hoshino M, Kita S, Saito A, Kojima Y, Maejima A, Noguchi E, Okuma HS, Koyama T, Shimoi T, Yonemori K. Treatment and Reasons for Choosing Treatment in Breast Cancer Patients Who Underwent Next-Generation Sequencing Test. Oncology, 1-10, 2025
9. Kitadai R, Yazaki S, Kuchiba A, Yamanaka T, Shiino S, Yamauchi C, Harano K, Saito M, Hirotsu Y, Aiba H, Yoshida T, Hamamoto R, Shimizu C, Shimomura A, Kojima Y, Shimoi T, Momozawa Y, Sudo K, Yoshida M, Sunami K, Hori M, Katanoda K, Shimada Y, Yamashita Y, Kogawa T, Murata T, Fujiwara S, Miyagi Y, Nakagomi H, Tachibana K, Omata M, Ohtake T, Suto A, Onishi T, Naito Y, Yamashita T, Yonemori K, Kohno T, Shiraishi K. Germline Pathogenic Variants and Clinical Outcomes in Asian Patients With Breast Cancer. Cancer science, 116:1048-1058, 2025
10. Saito Y, Suzuki S, Sanomachi T, Kato K, Otake H, Nishise Y, Yamada Y, Saito K, Takahashi K, Kumanishi R, Fukui T, Yoshioka T. Pancreatic Mixed Acinar-neuroendocrine Carcinoma in a Patient With a Germline PTEN Variant: A Case Report and Genomic Literature Review. In vivo (Athens, Greece), 39:1173-1181, 2025
11. Nassar AH, Abou Alaiwi S, El Zarif T, Denu R, Macaron W, Abdel-Wahab N, Freeman D, Vasbinder A, Hayeck S, Anderson E, Goodman RS, Johnson DB, Grynberg S, Shapira R, Kwan JM, Woodford R, Long GV, Haykal T, Dent S, Kojima Y, Yonemor K, Tandon A, Trevino A, Akhter N, Yang EH, Hui G, Drakaki A, El-Am E, Kozaily E, Al-Hader A, Bou Farhat E, Babu P, Mittra A, Li M, Jones N, Baena J, Juarez Herrera M, Foderaro S, Nana FA, Kim C, Sackstein P, Parikh K, Desai AP, Smith C, Cortellini A, Pinato DJ, Korolewicz J, Lopetegui-Lia N, Funchain P, Choudhary A, Asnani A, Navani V, Meyers D, Stukalin I, Ocejo Gallegos JA, Trent J, Nusrat S, Malvar C, McKay RR, Neilan TG, Choueiri TK, Naqash AR. Safety and efficacy of immune checkpoint therapy for the treatment of patients with cardiac metastasis: a multicenter international retrospective study. Journal for immunotherapy of cancer, 13:e009364, 2025
12. Hara H, Hijioka S, Yamashige D, Nagashio Y, Komori Y, Kuwada M, Fukuda S, Yagi S, Okamoto K, Agarie D, Chatto M, Okada M, Maruki Y, Morizane C, Ueno H, Saito Y, Yonemori K, Okusaka T. Clinical significance and efficacy of endoscopic ultrasound-guided tissue acquisition for para-aortic lymph node metastasis. Digestive endoscopy, 2025
13. Yamanaka T, Nishikawa T, Yoshida H. Development of Antibody-Drug Conjugates for Malignancies of the Uterine Corpus: A Review. Cells, 14:333, 2025
14. Imai T, Shimoi T, Kawai A, Yonemori K. Diagnosis and treatment of cardiac tumors. Medical oncology (Northwood, London, England), 42:110, 2025
15. Yamanaka T, Ukita J, Xue D, Kondoh C, Honda S, Noguchi M, Yonejima Y, Nonogaki K, Takemura K, Kizawa R, Yamaguchi T, Tanabe Y, Suyama K, Ogaki K, Miura Y. Artificial intelligence system for predicting hand-foot skin reaction induced by vascular endothelial growth factor receptor inhibitors. Scientific reports, 15:9843, 2025
16. Kitadai R, Okuma Y, Shibata T, Kohno T, Koyama T. Tissue-agnostic target profiles and treatment efficacy in cancer patients: Insights from the C-CAT clinicogenomic repository. European journal of cancer (Oxford, England : 1990), 220:115380, 2025
17. Takamizawa S, Koyama T, Sunami K, Sudo K, Hirata M, Kubo T, Tao K, Cho H, Narita Y, Kato K, Yamazaki N, Ohe Y, Okusaka T, Matsui Y, Ogawa C, Yonemori K, Yamamoto N. Identification of barriers to implementation of precision oncology in patients with rare cancers. Cancer science, 115:2023-2035, 2024
18. Leon-Ferre RA, Jonas SF, Salgado R, Loi S, de Jong V, Carter JM, Nielsen TO, Leung S, Riaz N, Chia S, Jules-Clément G, Curigliano G, Criscitiello C, Cockenpot V, Lambertini M, Suman VJ, Linderholm B, Martens JWM, van Deurzen CHM, Timmermans AM, Shimoi T, Yazaki S, Yoshida M, Kim SB, Lee HJ, Dieci MV, Bataillon G, Vincent-Salomon A, André F, Kok M, Linn SC, Goetz MP, Michiels S. Tumor-Infiltrating Lymphocytes in Triple-Negative Breast Cancer. JAMA, 331:1135-1144, 2024
19. Uehara T, Matsuzaki J, Yoshida H, Ogawa Y, Miura J, Fujimiya H, Yamamoto Y, Kawauchi J, Takizawa S, Yonemori K, Sakamoto H, Kato K, Ishikawa M, Ochiya T. Potential utility of pretreatment serum miRNAs for optimal treatment selection in advanced high-grade serous ovarian cancer. Japanese journal of clinical oncology, 54:917-925, 2024
20. Imai T, Kojima Y, Shimoi T, Aiba H, Okuma HS, Saito A, Kita S, Yamamoto K, Maejima A, Nishikawa T, Sudo K, Noguchi E, Yoshida A, Matsui Y, Iwata S, Kobayashi E, Kawai A, Udagawa R, Fujiwara Y, Yonemori K. Predicting Trabectedin Efficacy in Soft Tissue Sarcoma: Inflammatory Biomarker Analysis. Anticancer research, 44:2125-2132, 2024
21. Chiba Y, Kojima Y, Yazaki S, Yoshida H, Takamizawa S, Kitadai R, Saito A, Okuma HS, Nishikawa T, Shimoi T, Sudo K, Noguchi E, Uno M, Ishikawa M, Kato T, Fujiwara Y, Yonemori K. Trop-2 expression and the tumor immune microenvironment in cervical cancer. Gynecologic oncology, 187:51-57, 2024
22. Ozawa R, Nishikawa T, Yoshida H, Shiraishi K, Shimoi T, Kato T, Yonemori K. Unveiling pembrolizumab effectiveness in diverse subtypes of MSI-high endometrial cancers. Journal of gynecologic oncology, 35:e103, 2024
23. Kawai A, Yoshida A, Shimoi T, Kobayashi E, Yonemori K, Ogura K, Iwata S, Toshirou N. Histological diagnostic discrepancy and its clinical impact in bone and soft tissue tumors referred to a sarcoma center. Cancer science, 115:2831-2838, 2024
24. Mukohara T, Park YH, Sommerhalder D, Yonemori K, Hamilton E, Kim SB, Kim JH, Iwata H, Yamashita T, Layman RM, Mita M, Clay T, Chae YS, Oakman C, Yan F, Kim GM, Im SA, Lindeman GJ, Rugo HS, Liyanage M, Saul M, Le Corre C, Skoura A, Liu L, Li M, LoRusso PM. Inhibition of lysine acetyltransferase KAT6 in ER(+)HER2(-) metastatic breast cancer: a phase 1 trial. Nature medicine, 30:2242-2250, 2024
25. Cortés J, Hurvitz SA, Im SA, Iwata H, Curigliano G, Kim SB, Chiu JWY, Pedrini JL, Li W, Yonemori K, Bianchini G, Loi S, Borges GS, Wang X, Bachelot T, Nakatani S, Ashfaque S, Liang Z, Egorov A, Hamilton E. Trastuzumab deruxtecan versus trastuzumab emtansine in HER2-positive metastatic breast cancer: long-term survival analysis of the DESTINY-Breast03 trial. Nature medicine, 30:2208-2215, 2024
26. Shimoi T, Pathadka S, Sekine N, Cai Z, Tanizawa Y, Kawaguchi T, Saji S, Yamashita T. Real-world data on patients with early breast cancer who were prescribed abemaciclib adjuvant therapy in Japan. Future oncology (London, England), 20:2179-2188, 2024
27. Imai T, Shoji H, Hirano H, Matsuguma K, Awatsu T, Hirose T, Okita N, Takashima A, Kato K. BRAF V600E-mutant colorectal cancer with CNS metastases treated successfully with encorafenib, binimetinib and cetuximab. CNS oncology, 13:2347824, 2024
28. Uchihara M, Tanabe A, Kojima Y, Shimoi T, Maeshima AM, Umamoto K, Shimomura A, Shimizu C, Yamazaki Y, Nakamura E, Matsui Y, Takemura N, Miyazaki H, Sudo K, Yonemori K, Kajio H. Immunohistochemical Profiling of SSTR2 and HIF-2α with the Tumor Microenvironment in Pheochromocytoma and Paraganglioma. Cancers, 16:2191, 2024
29. Nagao A, Murakami N, Sakuramachi M, Kashihara T, Takahashi K, Kaneda T, Inaba K, Okuma K, Okamoto H, Nakayama Y, Yonemori K, Igaki H. Role of the gel spacer in safely delivering whole pelvic radiation therapy without central shielding in computed tomography-based image-guided adaptive brachytherapy for uterine cervical cancer patients. Brachytherapy, 23:595-603, 2024
30. Vergote I, González-Martín A, Fujiwara K, Kalbacher E, Bagaméri A, Ghamande S, Lee JY, Banerjee S, Maluf FC, Lorusso D, Yonemori K, Van Nieuwenhuysen E, Manso L, Woelber L, Westermann A, Covens A, Hasegawa K, Kim BG, Raimondo M, Bjurberg M, Cruz FM, Angelergues A, Cibula D, Barraclough L, Oaknin A, Gennigens C, Nicacio L, Teng MSL, Whalley E, Soumaoro I, Slomovitz BM. Tisotumab Vedotin as Second- or Third-Line Therapy for Recurrent Cervical Cancer. The New England journal of medicine, 391:44-55, 2024
31. Birrer M, Li G, Yunokawa M, Lee JY, Kim BG, Oppermann CP, Zhou Q, Nishio S, Okamoto A, Wu X, Mileshkin L, Oaknin A, Ray-Coquard I, Hasegawa K, Jehl G, Vugmeyster Y, Zhang S, Bajars M, Yonemori K. Bintrafusp Alfa for Recurrent or Metastatic Cervical Cancer After Platinum Failure: A Nonrandomized Controlled Trial. JAMA oncology, 10:1204-1211, 2024
32. Nishikawa T, Kakunaga S, Tamura K, Ando M, Ozaki T, Kawai A, Ueda T, Kawasaki M, Tomatsuri S, Okamura N, Kamikura M, Hamada A, Yoshida A, Hirakawa A, Shibata T, Nakamura K, Yonemori K. Efficacy and safety of nivolumab monotherapy in patients with unresectable clear cell sarcoma and alveolar soft part sarcoma (OSCAR Trial/NCCH1510). Cancer, 130:3836-3844, 2024
33. Nakayama T, Niikura N, Yamanaka T, Yamamoto M, Matsuura K, Inoue K, Takahara S, Nomura H, Kita S, Yamaguchi M, Aruga T, Shibata N, Shimomura A, Ozaki Y, Sakai S, Takiguchi D, Takata T, Bastanfard A, Shiosakai K, Tsurutani J. Trastuzumab deruxtecan for the treatment of patients with HER2-positive breast cancer with brain and/or leptomeningeal metastases: an updated overall survival analysis using data from a multicenter retrospective study (ROSET-BM). Breast cancer (Tokyo, Japan), 31:1167-1175, 2024
34. Koyama T, Yonemori K, Shimizu T, Sato J, Kondo S, Sudo K, Yoshida T, Katsuya Y, Imaizumi T, Enomoto M, Seki R, Yamamoto N. Phase I Study of Simlukafusp Alfa (FAP-IL2v) with or without Atezolizumab in Japanese Patients with Advanced Solid Tumors. Cancer research communications, 4:2349-2358, 2024
35. Kitadai R, Shimoi T, Yazaki S, Okuma HS, Hoshino M, Ito M, Saito A, Kita S, Kojima Y, Nishikawa T, Sudo K, Noguchi E, Fujiwara Y, Yoshida M, Yonemori K. Clinicopathological and prognostic features of HER2-null and HER2-low advanced breast cancer treated with eribulin or capecitabine. Breast cancer (Tokyo, Japan), 31:1037-1045, 2024
36. Noguchi E, Yamanaka T, Mukai H, Yamamoto N, Chung CF, Lu YS, Chang DY, Sohn J, Kim GM, Lee KH, Lee SC, Iwasa T, Iwata H, Watanabe K, Jung KH, Tanabe Y, Kang SY, Yasojima H, Aogi K, Tokunaga E, Sim SH, Yap YS, Matsumoto K, Tseng LM, Umeyama Y, Sudo K, Kojima Y, Hata T, Kuchiba A, Shibata T, Nakamura K, Fujiwara Y, Tamura K, Yonemori K. A phase 3 study (PATHWAY) of palbociclib plus tamoxifen in patients with HR-positive/HER2-negative advanced breast cancer. NPJ breast cancer, 10:76, 2024
37. Akechi T, Furukawa TA, Hashimoto H, Harada Y, Ito Y, Furukawa Y, Kitano A, Maeda N, Kojima Y, Tada Y, Watanabe A, Kurata A, Matsubara T, Sakurai N, Uchitomi Y, Okamura M, Fujimori M. Smartphone-based distress screening, information provision, and psychotherapy for reducing psychological distress among AYA cancer survivors: protocol for a fully decentralized multicenter randomized controlled clinical trial. Japanese journal of clinical oncology, 54:1351-1357, 2024
38. Karasawa H, Yasumizu Y, Kosaka T, Shimoi T, Oya M. Efficacy of trametinib in a metastatic urothelial carcinoma patient with a BRAF mutation. IJU case reports, 7:375-378, 2024
39. Ozawa R, Nishikawa T, Yamamoto K, Shimoi T, Ishikawa M, Kato T, Yonemori K. The efficacy and safety of lenvatinib plus pembrolizumab therapy in patients with uterine carcinosarcoma. Gynecologic oncology reports, 55:101479, 2024
40. Jhaveri KL, Lim E, Jeselsohn R, Ma CX, Hamilton EP, Osborne C, Bhave M, Kaufman PA, Beck JT, Manso Sanchez L, Parajuli R, Wang HC, Tao JJ, Im SA, Harnden K, Yonemori K, Dhakal A, Neven P, Aftimos P, Pierga JY, Lu YS, Larson T, Jerez Y, Sideras K, Sohn J, Kim SB, Saura C, Bardia A, Sammons SL, Bacchion F, Li Y, Yuen E, Estrem ST, Rodrik-Outmezguine V, Nguyen B, Ismail-Khan R, Smyth L, Beeram M. Imlunestrant, an Oral Selective Estrogen Receptor Degrader, as Monotherapy and in Combination With Targeted Therapy in Estrogen Receptor-Positive, Human Epidermal Growth Factor Receptor 2-Negative Advanced Breast Cancer: Phase Ia/Ib EMBER Study. Journal of clinical oncology, 42:4173-4186, 2024
41. Yamamoto K, Tanabe Y, Nonogaki K, Watanabe S, Takemura K, Yamanaka T, Kizawa R, Yamaguchi T, Suyama K, Hayashi N, Miura Y. Questionnaire survey of healthcare professionals on taxane-induced nail change in Japan. Supportive care in cancer, 32:647, 2024
42. Bardia A, Hu X, Dent R, Yonemori K, Barrios CH, O'Shaughnessy JA, Wildiers H, Pierga JY, Zhang Q, Saura C, Biganzoli L, Sohn J, Im SA, Lévy C, Jacot W, Begbie N, Ke J, Patel G, Curigliano G. Trastuzumab Deruxtecan after Endocrine Therapy in Metastatic Breast Cancer. The New England journal of medicine, 391:2110-2122, 2024
43. Naito Y, Nakamura S, Kawaguchi-Sakita N, Ishida T, Nakayama T, Yamamoto Y, Masuda N, Matsumoto K, Kogawa T, Sudo K, Shimomura A, Lai C, Zhang D, Iwahori Y, Gary D, Huynh D, Iwata H. Preliminary results from ASCENT-J02: a phase 1/2 study of sacituzumab govitecan in Japanese patients with advanced solid tumors. International journal of clinical oncology, 29:1684-1695, 2024
44. Hasegawa K, Takahashi S, Ushijima K, Okadome M, Yonemori K, Yokota H, Vergote I, Monk BJ, Tewari KS, Fujiwara K, Li J, Jamil S, Paccaly A, Takehara K, Usami T, Aoki Y, Suzuki N, Kobayashi Y, Yoshida Y, Watari H, Seebach F, Lowy I, Mathias M, Fury MG, Oaknin A. Cemiplimab monotherapy in Japanese patients with recurrent or metastatic cervical cancer. Cancer medicine, 13:e70236, 2024
45. Yonemori K, Boni V, Min KG, Meniawy TM, Lombard J, Kaufman PA, Richardson DL, Bender L, Okera M, Matsumoto K, Giridhar KV, García-Sáenz JA, Prenen H, de Speville Uribe BD, Dizon DS, Garcia-Corbacho J, Van Nieuwenhuysen E, Li Y, Estrem ST, Nguyen B, Bacchion F, Ismail-Khan R, Jhaveri K, Banda K. Imlunestrant, an oral selective estrogen receptor degrader, as monotherapy and combined with abemaciclib, in recurrent/advanced ER-positive endometrioid endometrial cancer: Results from the phase 1a/1b EMBER study. Gynecologic oncology, 191:172-181, 2024
46. Tokura M, Ando MM, Kojima Y, Kitadai R, Yazaki S, Atutubo CMN, Li RK, Perez MZ, Gorospe AE, Madrid MA, Ordinario MVC, Imasa MSB, Sudo K, Shimoi T, Suto A, Kohsaka S, Machida R, Sadachi R, Yoshida M, Yatabe Y, Hata T, Nakamura K, Yonemori K, Shiino S. Multicenter Prospective Study in HER2-Positive Early Breast Cancer for Detecting Minimal Residual Disease by Circulating Tumor DNA Analysis With Neoadjuvant Chemotherapy: HARMONY Study. Breast cancer : basic and clinical research, 18:11782234241288671, 2024
