Annual Report 2024
Department of Thoracic Oncology
Yuichiro Ohe, Noboru Yamamoto, Hidehito Horinouchi, Yasushi Goto, Tatsuya Yoshida, Yusuke Okuma, Yuki Shinno, Ken Masuda, Yuji Matsumoto, Yukiko Igawa, Akiko Tateishi, Yoh Yamaguchi, Yoshihiro Masui
Introduction
Lung cancer is the leading cause of cancer death in Japan and worldwide. The incidence of lung cancer in Japan is still increasing, especially in the elderly. The Department of Thoracic Oncology provides care for patients with primary lung cancer, mediastinal tumors, and pleural tumors. The goals of the department are to provide treatment with the highest quality and to establish new effective treatments against lung cancer and other thoracic malignancies through innovative clinical and translational research. To assist our patients through multidisciplinary care, the staff members of the department work closely with thoracic surgeons, radiation oncologists, pathologists, pharmacists, clinical research coordinators, and psychiatrists who have expertise in these areas. The department includes nine staff physicians. Moreover, residents and trainees from other institutions have joined the Thoracic Oncology Program.
The Team and What We Do
The staff physicians attend outpatient services for thoracic diseases, and the department has approximately 50 beds in the hospital. Inpatient care is carried out by five teams. Each team consists of one staff physician and one or two residents and/or trainee doctors. Case conferences are scheduled every Monday and Tuesday morning. Protocol conference and journal club are scheduled every Monday afternoon and Thursday morning, respectively.
A total of 542 new patients started treatment in 2024, and their backgrounds and initial treatments of these patients are shown in Tables 1 and 2. The initial treatments were chemotherapy in 288 cases, adjuvant chemotherapy after surgery in 59, chemoradiotherapy in 59, curative radiotherapy in 3, and supportive care including palliative radiotherapy in 41. The survival rates of lung cancer patients treated in 2014-2018 in our department are shown in Table 3.
Table 1. Number of patients in 2024

Table 2. Type of procedure in 2024

Table 3. Survival rates of lung cancer patients treated in 2014-2018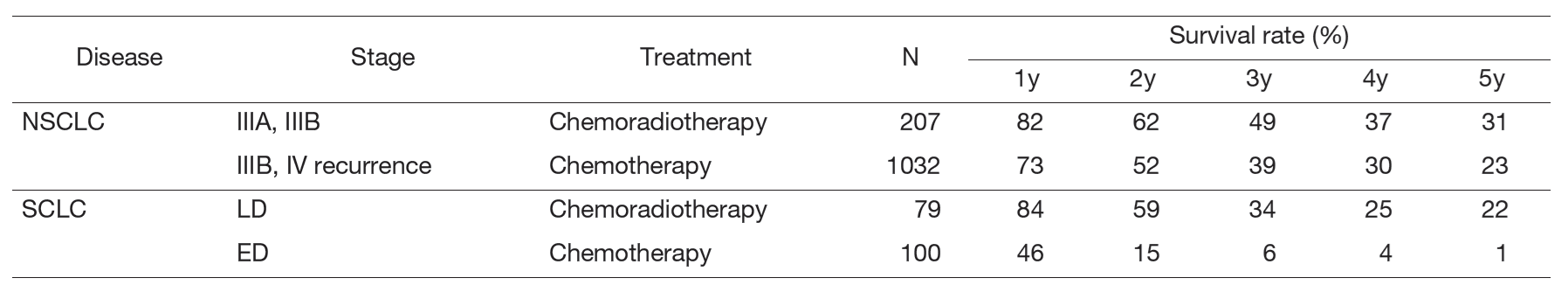

Research Activities
In fiscal year 2024, our department achieved notable progress in both clinical and translational research with a focus on lung cancer. Clinically, we actively participated in international collaborations and JCOG trials, including adjuvant alectinib for resected ALK-positive disease (NEJM), perioperative immunotherapy for resectable NSCLC (CheckMate 77T and KEYNOTE-671; NEJM/Lancet), crizotinib for MET-altered tumors in Japanese patients, and JCOG2007 evaluating optimization of immune checkpoint inhibitor-based regimens. These efforts directly contributed to establishing standards of care. We also published practice-driven studies from Japan addressing real-world issues such as the impact of antibiotic-induced dysbiosis on chemo-immunotherapy outcomes and pharmacokinetic analyses of pembrolizumab in older adults.
For rare and refractory malignancies, we reported long-term outcomes of lenvatinib for thymic carcinoma, real-world therapeutic selections, and case series demonstrating the antitumor efficacy of corticosteroids in refractory thymoma—findings that help inform treatment guidance. Further, we advanced precision oncology through investigations of treatment effects in tumors with uncommon drivers such as EGFR and HER2, characterization of compound mutations and immune resistance mechanisms, and discovery research using TCR repertoire profiling and multi-omics analyses, which were published as translational bridgework in international journals.
We also contributed on the health-policy front by reporting national data on high-cost therapies within the JCOG Lung Cancer Group and co-authoring a society position paper on the appropriate clinical use of molecular residual disease (MRD) testing—linking frontline practice with policy. In addition, our participation in global trials of next-generation agents for small-cell lung cancer (e.g., tarlatamab, telisotuzumab vedotin) supported ongoing therapeutic development.
Collectively, these outputs appeared in leading journals—including Lung Cancer, Journal of Clinical Oncology, Clinical Cancer Research, Molecular Cancer, the New England Journal of Medicine, and The Lancet—highlighting growing international recognition of our program. We also delivered presentations at major international meetings (ASCO, ESMO, WCLC), further underscoring our department’s role in shaping contemporary lung cancer care.
Education
In 2024, 2 chief residents, 6 residents, and 4 trainee doctors joined the department. A monthly research conference is held to discuss the clinical and translational research conducted by young doctors.
Future Prospects
In lung cancer, advances in perioperative immunotherapy and molecular targeted therapy are expected to further improve outcomes, with biomarker-driven treatment optimization being the key to future progress. For rare malignancies such as thymic carcinoma and mesothelioma, international collaborations and accumulation of real-world data are essential to establish standards of care, while novel targeted agents and immunotherapies hold promise. Moving forward, integrated efforts spanning basic science, clinical practice, and health policy will be crucial to generate robust evidence and maximize patient benefit.
List of papers published in 2024
Journal
1. Sekino Y, Hishida T, Yoshioka H, Wakabayashi M, Mitome N, Shiono S, Kenmotsu H, Nosaki K, Aokage K, Horinouchi H, Fukuda H, Ohe Y, Watanabe SI. Protocol summary of a randomized phase III study: comparing systemic therapy with and without debulking surgery (primary tumour resection) for clinical stage IVA (cT1-2bN0-1M1a) non-small cell lung cancer with radiologically undetermined pleural dissemination JCOG2103 (DEBULK-LUNG). Japanese journal of clinical oncology, 55:176-182, 2025
2. Okuma Y. New territories: perioperative chemoimmunotherapy in early-stage NSCLC. The Lancet. Respiratory medicine, 13:102-103, 2025
3. Horinouchi H, Chang CS, Shaw J, Archangelidi O, Balasubramanian A, Pundole X. Real-world treatment patterns and survival in extensive stage small-cell lung cancer in Japan. Japanese journal of clinical oncology, 55:383-390, 2025
4. Horinouchi H, Murakami H, Harada H, Sobue T, Kato T, Atagi S, Kozuki T, Tokito T, Oizumi S, Seike M, Ohashi K, Mio T, Sone T, Iwao C, Iwane T, Koto R, Tsuboi M. Real-world status of multimodal treatment of Stage IIIA-N2 non-small cell lung cancer in Japan: Results from the SOLUTION study, a non-interventional, multicenter cohort study. Lung cancer (Amsterdam, Netherlands), 199:108027, 2025
5. Hatano H, Yoshida T, Higashiyama R, Torasawa M, Uehara Y, Ohe Y. EGFR, TP53, and CUL3 Triple Mutation in Non-Small Cell Lung Cancer and its Potentially Poor Prognosis: A Case Report and Database Analysis. Thoracic cancer, 16:e15523, 2025
6. Fukui T, Mamesaya N, Takahashi T, Kishi K, Yoshizawa T, Tokito T, Azuma K, Morikawa K, Igawa S, Okuma Y, Yamanaka Y, Hosokawa S, Kasai T, Masubuchi K, Nakamichi S, Aga M, Sasaki J, Kada A, Saito AM, Naoki K, Okamoto H. A Prospective Phase II Trial of First-Line Osimertinib for Patients With EGFR Mutation-Positive NSCLC and Poor Performance Status (OPEN/TORG2040). Journal of thoracic oncology, 20:665-675, 2025
7. Masuda K, Yoshida T, Motoi N, Shinno Y, Matsumoto Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N, Watanabe SI, Hoshino T, Yatabe Y, Ohe Y. Schlafen 11 Expression in Patients With Small Cell Lung Cancer and Its Association With Clinical Outcomes. Thoracic cancer, 16:e15529, 2025
8. Shirasawa M, Yoshida T, Matsutani T, Takeyasu Y, Goto N, Yagishita S, Kitano S, Kuroda H, Hida T, Kurata T, Ohe Y. Diversity of TCR repertoire predicts recurrence after CRT followed by durvalumab in patients with NSCLC. NPJ precision oncology, 9:17, 2025
9. Hayato S, Hamuro L, Shimizu T, Yonemori K, Nishio S, Yunokawa M, Yoshida T, Nishio M, Matsumoto K, Takehara K, Hasegawa K, Kozuki T, Hirashima Y, Kato H, Miura T, Nomoto M, Zhao Y, Zhu L, Yasuda S. Pharmacokinetic and Exposure-Response Modeling Support Body Surface Area-Based Dosing of Farletuzumab Ecteribulin in Japanese Patients with Solid Tumors. Journal of clinical pharmacology, 65:751-762, 2025
10. Lee KH, Lee JS, Sugawara S, Kang JH, Kim HR, Inui N, Hida T, Yoshida T, Tanaka H, Yang CT, Inoue T, Nishio M, Ohe Y, Tamura T, Yamamoto N, Yu CJ, Akamatsu H, Takahashi S, Nakagawa K. First-line nivolumab plus platinum chemotherapy and bevacizumab for advanced nonsquamous non-small cell lung cancer: A 3-year follow-up of the phase 3 randomized TASUKI-52 trial. Lung cancer (Amsterdam, Netherlands), 201:108109, 2025
11. Fujii H, Okuma Y, Hirata M, Shinno Y, Yoshida T, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. EGFR-Mutated Lung Adenocarcinoma With Li-Fraumeni Syndrome: The Imperative for Germline Testing in Patients With a Family History, a Case Report. JTO clinical and research reports, 6:100691, 2025
12. Okuma Y. Treatment strategy for EGFR mutated NSCLC. The Lancet. Respiratory medicine, 13:292-294, 2025
13. Kobayashi S, Nakamura Y, Hashimoto T, Bando H, Oki E, Karasaki T, Horinouchi H, Ozaki Y, Iwata H, Kato T, Miyake H, Ohba A, Ikeda M, Chiyoda T, Hasegawa K, Fujisawa T, Matsuura K, Namikawa K, Yajima S, Yoshino T, Hasegawa K. Japan society of clinical oncology position paper on appropriate clinical use of molecular residual disease (MRD) testing. International journal of clinical oncology, 30:605-654, 2025
14. Camidge DR, Sugawara S, Kondo M, Kim HR, Ahn MJ, Yang JCH, Han JY, Hochmair MJ, Lee KH, Delmonte A, Kudou K, Asato T, Hupf B, Vranceanu F, Fram RJ, Ohe Y, Popat S. Efficacy and safety of brigatinib in patients with ALK TKI-naive advanced ALK+ NSCLC: Integrated analysis of the ALTA-1L and J-ALTA trials. Lung cancer (Amsterdam, Netherlands), 201:108424, 2025
15. Nishinakamura H, Shinya S, Irie T, Sakihama S, Naito T, Watanabe K, Sugiyama D, Tamiya M, Yoshida T, Hase T, Yoshida T, Karube K, Koyama S, Nishikawa H. Coactivation of innate immune suppressive cells induces acquired resistance against combined TLR agonism and PD-1 blockade. Science translational medicine, 17:eadk3160, 2025
16. Takeuchi T, Horinouchi H, Takasawa K, Mukai M, Masuda K, Shinno Y, Okuma Y, Yoshida T, Goto Y, Yamamoto N, Ohe Y, Miyake M, Watanabe H, Kusumoto M, Aoki T, Nishimura K, Hamamoto R. A series of natural language processing for predicting tumor response evaluation and survival curve from electronic health records. BMC medical informatics and decision making, 25:85, 2025
17. Satoh H, Okuma Y, Shinno Y, Masuda K, Matsumoto Y, Yoshida T, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. Evolving treatments and prognosis in Stage IV non-small cell lung cancer: 20 years of progress of novel therapies. Lung cancer (Amsterdam, Netherlands), 202:108453, 2025
18. Nogami N, Umemura S, Kozuki T, Zenke Y, Ohtani J, Ishii M, Han S, Noguchi K, Horinouchi H. A phase 1 study of pembrolizumab plus ipilimumab as first-line treatment in Japanese patients with advanced non-small-cell lung cancer. Respiratory investigation, 63:296-302, 2025
19. Hakozaki T, Tanaka K, Shiraishi Y, Sekino Y, Mitome N, Okuma Y, Aiba T, Utsumi T, Tanizaki J, Azuma K, Hara S, Morita R, Niho S, Yokoyama T, Toyozawa R, Horinouchi H, Okamoto I, Hosomi Y, Ohe Y. Gut Microbiota in Advanced NSCLC Receiving Chemoimmunotherapy: An Ancillary Biomarker Study From the Phase III Trial JCOG2007 (NIPPON). Journal of thoracic oncology, 2025
20. Cho BC, Johnson M, Bar J, Schaefer E, Yoh K, Zer A, Moskovitz M, Lee SH, Moreno V, de Miguel M, Okuma Y, Kim JH, Lee CH, Peguero J, Ansell P, Biesdorf C, Saab R, Freise KJ, Ramies D, Jeng EE, Camidge DR. A phase 1b study of cofetuzumab pelidotin monotherapy in patients with PTK7-expressing recurrent non-small cell lung cancer. Lung cancer (Amsterdam, Netherlands), 202:108492, 2025
21. Torasawa M, Yoshida T, Shiraishi K, Yagishita S, Ono H, Uehara Y, Miyakoshi J, Tateishi A, Igawa YS, Higashiyama RI, Mochizuki A, Masuda K, Matsumoto Y, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Hamamoto R, Yamamoto N, Watanabe SI, Yatabe Y, Takahashi K, Kohno T, Ohe Y. Implications of EGFR expression on EGFR signaling dependency and adaptive immunity against EGFR-mutated lung adenocarcinoma. Lung cancer (Amsterdam, Netherlands), 202:108494, 2025
22. Kinoshita F, Tanzawa S, Misumi T, Yoshioka H, Miyauchi E, Ninomiya K, Murata Y, Takeshita M, Yamaguchi M, Sugawara S, Kawashima Y, Hashimoto K, Mori M, Miyanaga A, Hayashi A, Tanaka H, Honda R, Nojiri M, Sato Y, Yamamoto K, Masuda K, Kozuki T, Kawamura T, Suzuki T, Yamaguchi T, Asada K, Tetsumoto S, Tanaka H, Watanabe S, Umeda Y, Yamaguchi K, Kuyama S, Tsuruno K, Misumi Y, Kuraishi H, Yoshihara K, Nakao A, Kubo A, Yokoyama T, Watanabe K, Seki N. Skin disorder within 30 days is a favorable prognostic factor in patients with lung squamous cell carcinoma treated with necitumumab plus gemcitabine and cisplatin: a sub-analysis of the NINJA study. Therapeutic advances in medical oncology, 17:17588359241312503, 2025
23. Kitadai R, Okuma Y, Shibata T, Kohno T, Koyama T. Tissue-agnostic target profiles and treatment efficacy in cancer patients: Insights from the C-CAT clinicogenomic repository. European journal of cancer (Oxford, England : 1990), 220:115380, 2025
24. Miyakoshi J, Yoshida T, Kashima J, Shirasawa M, Torasawa M, Matsumoto Y, Masuda K, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Shiraishi K, Kohno T, Yamamoto N, Yatabe Y, Suzuki T, Ohe Y. Clinical significance of inter-assay discrepancy in PD-L1 evaluation for the efficacy of pembrolizumab in advanced NSCLC with high PD-L1 expression. Lung cancer (Amsterdam, Netherlands), 191:107788, 2024
25. Wu YL, Dziadziuszko R, Ahn JS, Barlesi F, Nishio M, Lee DH, Lee JS, Zhong W, Horinouchi H, Mao W, Hochmair M, de Marinis F, Migliorino MR, Bondarenko I, Lu S, Wang Q, Ochi Lohmann T, Xu T, Cardona A, Ruf T, Noe J, Solomon BJ. Alectinib in Resected ALK-Positive Non-Small-Cell Lung Cancer. The New England journal of medicine, 390:1265-1276, 2024
26. Niho S, Sato J, Satouchi M, Itoh S, Okuma Y, Mizugaki H, Murakami H, Fujisaka Y, Kozuki T, Nakamura K, Machida R, Ohe Y, Tamai T, Ikezawa H, Yamamoto N. Long-term follow-up and exploratory analysis of lenvatinib in patients with metastatic or recurrent thymic carcinoma: Results from the multicenter, phase 2 REMORA trial. Lung cancer (Amsterdam, Netherlands), 191:107557, 2024
27. Tamura K, Okuma Y, Nomura S, Fukuda A, Masuda K, Matsumoto Y, Shinno Y, Yoshida T, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. Efficacy and safety of chemoimmunotherapy in advanced non-small cell lung cancer patients with antibiotics-induced dysbiosis: a propensity-matched real-world analysis. Journal of cancer research and clinical oncology, 150:216, 2024
28. Torasawa M, Yoshida T, Shiraishi K, Goto N, Ueno T, Ichikawa H, Yagishita S, Kohsaka S, Goto Y, Yatabe Y, Hamada A, Mano H, Ohe Y. Rapid Response to Lenvatinib and Disease Flare After Discontinuation in a Patient With Thymic Carcinoma Harboring KIT Exon 11 Mutation: A Case Report. JTO clinical and research reports, 5:100657, 2024
29. Tian T, Li Y, Li J, Xu H, Fan H, Zhu J, Wang Y, Peng F, Gong Y, Du Y, Yan X, He X, Cali Daylan AE, Pircher A, Neibart SS, Okuma Y, Hong MH, Huang M, Lu Y. Immunotherapy for patients with advanced non-small cell lung cancer harboring oncogenic driver alterations other than EGFR: a multicenter real-world analysis. Translational lung cancer research, 13:861-874, 2024
30. Cascone T, Awad MM, Spicer JD, He J, Lu S, Sepesi B, Tanaka F, Taube JM, Cornelissen R, Havel L, Karaseva N, Kuzdzal J, Petruzelka LB, Wu L, Pujol JL, Ito H, Ciuleanu TE, de Oliveira Muniz Koch L, Janssens A, Alexandru A, Bohnet S, Moiseyenko FV, Gao Y, Watanabe Y, Coronado Erdmann C, Sathyanarayana P, Meadows-Shropshire S, Blum SI, Provencio Pulla M. Perioperative Nivolumab in Resectable Lung Cancer. The New England journal of medicine, 390:1756-1769, 2024
31. Nosaki K, Yoh K, Toyozawa R, Horinouchi H, Morise M, Ohashi K, Murakami H, Satouchi M, Sakakibara-Konishi J, Yano S, Okumura F, Matsumoto S, Shimokawa M, Seto T, Goto K. Phase 2 trial of crizotinib in Japanese patients with advanced NSCLC harboring a MET gene alteration: a Co-MET study. International journal of clinical oncology, 29:1142-1151, 2024
32. Tamura A, Inaba Higashiyama R, Yoshida T, Satozono Y, Ohe Y. Response to dabrafenib plus trametinib on a rare BRAF mutation (V600_W604 deletion-insertion R) in an advanced non-small cell lung cancer patient. Thoracic cancer, 15:1454-1456, 2024
33. Fukuda A, Okuma Y. From Rarity to Reality: Osimertinib's Promising Horizon in Treating Uncommon EGFR Mutations in Non-Small Cell Lung Cancer. Clinical cancer research, 30:3128-3136, 2024
34. Tsuboi M, Murakami H, Harada H, Sobue T, Kato T, Atagi S, Tokito T, Mio T, Adachi H, Kozuki T, Sone T, Seike M, Toyooka S, Kitagawa H, Koto R, Yamazaki S, Horinouchi H. Treatment patterns and clinical outcomes of resectable clinical stage III non-small cell lung cancer in a Japanese real-world setting: Surgery cohort analysis of the SOLUTION study. Thoracic cancer, 15:1541-1552, 2024
35. Camidge DR, Bar J, Horinouchi H, Goldman J, Moiseenko F, Filippova E, Cicin I, Ciuleanu T, Daaboul N, Liu C, Bradbury P, Moskovitz M, Katgi N, Tomasini P, Zer A, Girard N, Cuppens K, Han JY, Wu SY, Baijal S, Mansfield AS, Kuo CH, Nishino K, Lee SH, Planchard D, Baik C, Li M, Ansell P, Xia S, Bolotin E, Looman J, Ratajczak C, Lu S. Telisotuzumab Vedotin Monotherapy in Patients With Previously Treated c-Met Protein-Overexpressing Advanced Nonsquamous EGFR-Wildtype Non-Small Cell Lung Cancer in the Phase II LUMINOSITY Trial. Journal of clinical oncology, 42:3000-3011, 2024
36 .Kaneko S, Takasawa K, Asada K, Shiraishi K, Ikawa N, Machino H, Shinkai N, Matsuda M, Masuda M, Adachi S, Takahashi S, Kobayashi K, Kouno N, Bolatkan A, Komatsu M, Yamada M, Miyake M, Watanabe H, Tateishi A, Mizuno T, Okubo Y, Mukai M, Yoshida T, Yoshida Y, Horinouchi H, Watanabe SI, Ohe Y, Yatabe Y, Saloura V, Kohno T, Hamamoto R. Mechanism of ERBB2 gene overexpression by the formation of super-enhancer with genomic structural abnormalities in lung adenocarcinoma without clinically actionable genetic alterations. Molecular cancer, 23:126, 2024
37. Yagishita S, Yamanaka Y, Kurata T, Watanabe K, Hosomi Y, Horinouchi H, Ohe Y, Nakahara Y, Naoki K, Asao T, Takahashi K, Saeki S, Sakagami T, Nakashima K, Tsubata Y, Fujita Y, Wakui H, Furuta M, Konishi JS, Ohuchi M, Ando Y, Mizugaki H, Hamada A. Multicenter Pharmacokinetic and Pharmacodynamic Study of Pembrolizumab for Non-small-Cell Lung Cancer in Patients Aged 75 Years and Older. Clinical pharmacology and therapeutics, 116:1042-1051, 2024
38. Ou SI, Le X, Nagasaka M, Reungwetwattana T, Ahn MJ, Lim DWT, Santos ES, Shum E, Lau SCM, Lee JB, Calles A, Wu F, Lopes G, Sriuranpong V, Tanizaki J, Horinouchi H, Garassino MC, Popat S, Besse B, Rosell R, Soo RA. Top 20 EGFR+ NSCLC Clinical and Translational Science Papers That Shaped the 20 Years Since the Discovery of Activating EGFR Mutations in NSCLC. An Editor-in-Chief Expert Panel Consensus Survey. Lung Cancer (Auckland, N.Z.), 15:87-114, 2024
39. Tanaka T, Goto Y, Masuda K, Shinno Y, Matsumoto Y, Okuma Y, Yoshida T, Horinouchi H, Yamamoto N, Ohe Y. The remarkable antitumor efficacy of corticosteroid treatment in patients with refractory thymomas. Respiratory investigation, 62:766-772, 2024
40. Blechter B, Wang X, Shi J, Shiraishi K, Choi J, Matsuo K, Chen TY, Dai J, Hung RJ, Chen K, Shu XO, Kim YT, Choudhury PP, Williams J, Landi MT, Lin D, Zheng W, Yin Z, Zhou B, Wang J, Seow WJ, Song L, Chang IS, Hu W, Chien LH, Cai Q, Hong YC, Kim HN, Wu YL, Wong MP, Richardson BD, Li S, Zhang T, Breeze C, Wang Z, Bassig BA, Kim JH, Albanes D, Wong JY, Shin MH, Chung LP, Yang Y, An SJ, Zheng H, Yatabe Y, Zhang XC, Kim YC, Caporaso NE, Chang J, Man Ho JC, Kubo M, Daigo Y, Song M, Momozawa Y, Kamatani Y, Kobayashi M, Okubo K, Honda T, Hosgood HD, Kunitoh H, Watanabe SI, Miyagi Y, Nakayama H, Matsumoto S, Horinouchi H, Tsuboi M, Hamamoto R, Goto K, Ohe Y, Takahashi A, Goto A, Minamiya Y, Hara M, Nishida Y, Takeuchi K, Wakai K, Matsuda K, Murakami Y, Shimizu K, Suzuki H, Saito M, Ohtaki Y, Tanaka K, Wu T, Wei F, Dai H, Machiela MJ, Su J, Kim YH, Oh IJ, Fun Lee VH, Chang GC, Tsai YH, Che KY, Huang MS, Su WC, Chen YM, Seow A, Park JY, Kweon SS, Chen KC, Gao YT, Qian B, Wu C, Lu D, Liu J, Schwartz AG, Houlston R, Spitz MR, Gorlov IP, Wu X, Yang P, Lam S, Tardon A, Chen C, Bojesen SE, Johansson M, Risch A, Bickeböller H, Ji BT, Wichmann HE, Christiani DC, Rennert G, Arnold S, Brennan P, McKay J, Field JK, Davies MPA, Shete SS, Le Marchand L, Liu G, Andrew A, Kiemeney LA, Zienolddiny-Narui S, Grankvist K, Johansson M, Cox A, Taylor F, Yuan JM, Lazarus P, Schabath MB, Aldrich MC, Jeon HS, Jiang SS, Sung JS, Chen CH, Hsiao CF, Jung YJ, Guo H, Hu Z, Burdett L, Yeager M, Hutchinson A, Hicks B, Liu J, Zhu B, Berndt SI, Wu W, Wang J, Li Y, Choi JE, Park KH, Sung SW, Liu L, Kang CH, Wang WC, Xu J, Guan P, Tan W, Yu CJ, Yang G, Loon Sihoe AD, Chen Y, Choi YY, Kim JS, Yoon HI, Park IK, Xu P, He Q, Wang CL, Hung HH, Vermeulen RCH, Cheng I, Wu J, Lim WY, Tsai FY, Chan JKC, Li J, Chen H, Lin HC, Jin L, Liu J, Sawada N, Yamaji T, Wyatt K, Li SA, Ma H, Zhu M, Wang Z, Cheng S, Li X, Ren Y, Chao A, Iwasaki M, et al. (複数名のため一部抜粋して記載。詳細は掲載先をご参照ください). Stratifying Lung Adenocarcinoma Risk with Multi-ancestry Polygenic Risk Scores in East Asian Never-Smokers. medRxiv : the preprint server for health sciences, 2024.06.26.24309127, 2024
41. Murata S, Horinouchi H, Morishita M, Kaku S, Shinno Y, Okuma Y, Yoshida T, Goto Y, Yamamoto N, Kashihara T, Okuma K, Kusumoto M, Ohe Y. Impact of Durvalumab on the Duration and Complexity of Corticosteroid Therapy for Pneumonitis After Chemoradiotherapy. Clinical lung cancer, 25:e369-e378.e3, 2024
42. Koyama T, Yonemori K, Shimizu T, Sato J, Kondo S, Sudo K, Yoshida T, Katsuya Y, Imaizumi T, Enomoto M, Seki R, Yamamoto N. Phase I Study of Simlukafusp Alfa (FAP-IL2v) with or without Atezolizumab in Japanese Patients with Advanced Solid Tumors. Cancer research communications, 4:2349-2358, 2024
43. Watanabe K, Sasaki K, Machida R, Shimizu J, Yamane Y, Tamiya M, Saito S, Takada Y, Yoh K, Yoshioka H, Murakami H, Kitazono S, Goto Y, Horinouchi H, Ohe Y. High-cost treatments for advanced lung cancer in Japan (Lung Cancer Study Group of the Japan Clinical Oncology Group). Japanese journal of clinical oncology, 54:1084-1092, 2024
44. Shiraishi Y, Nomura S, Sugawara S, Horinouchi H, Yoneshima Y, Hayashi H, Azuma K, Hara S, Niho S, Morita R, Yamaguchi M, Yokoyama T, Yoh K, Kurata T, Okamoto H, Okamoto M, Kijima T, Kasahara K, Fujiwara Y, Murakami S, Kanda S, Akamatsu H, Takemoto S, Kaneda H, Kozuki T, Ando M, Sekino Y, Fukuda H, Ohe Y, Okamoto I. Comparison of platinum combination chemotherapy plus pembrolizumab versus platinum combination chemotherapy plus nivolumab-ipilimumab for treatment-naive advanced non-small-cell lung cancer in Japan (JCOG2007): an open-label, multicentre, randomised, phase 3 trial. The Lancet. Respiratory medicine, 12:877-887, 2024
45. Dowlati A, Hummel HD, Champiat S, Olmedo ME, Boyer M, He K, Steeghs N, Izumi H, Johnson ML, Yoshida T, Bouchaab H, Borghaei H, Felip E, Jost PJ, Gadgeel S, Chen X, Yu Y, Martinez P, Parkes A, Paz-Ares L. Sustained Clinical Benefit and Intracranial Activity of Tarlatamab in Previously Treated Small Cell Lung Cancer: DeLLphi-300 Trial Update. Journal of clinical oncology, 42:3392-3399, 2024
46. Asada K, Kaneko S, Takasawa K, Shiraishi K, Shinkai N, Shimada Y, Takahashi S, Machino H, Kobayashi K, Bolatkan A, Komatsu M, Yamada M, Miyake M, Watanabe H, Tateishi A, Mizuno T, Okubo Y, Mukai M, Yoshida T, Yoshida Y, Horinouchi H, Watanabe SI, Ohe Y, Yatabe Y, Kohno T, Hamamoto R. Multi-omics and clustering analyses reveal the mechanisms underlying unmet needs for patients with lung adenocarcinoma and identify potential therapeutic targets. Molecular cancer, 23:182, 2024
47. Fujimoto N, Akamatsu A, Honda C, Aoki M, Ohe Y. Real-world safety of nivolumab in patients with malignant pleural mesothelioma in Japan: post-marketing surveillance study. Japanese journal of clinical oncology, 54:1321-1328, 2024
48. Higashiyama RI, Yoshida T. A Response to the Letter to the Editor: Safety Implications of Switching Pembrolizumab Dosage From 200 mg Every 3 Weeks to 400 mg Every 6 Weeks in Patients With Advanced NSCLC. Journal of thoracic oncology, 19:e33-e34, 2024
49. Furuta M, Horinouchi H, Yokota I, Yamaguchi T, Itoh S, Fukui T, Iwashima A, Sugisaka J, Miura Y, Tanaka H, Miyawaki T, Yokouchi H, Miura K, Saito R, Saito G, Kamoshida T, Uchinami Y, Kato T, Kobayashi K, Asahina H. Durvalumab after chemoradiotherapy for locoregional recurrence of completely resected non-small-cell lung cancer (NEJ056). Cancer science, 115:3705-3717, 2024
50. Spicer JD, Garassino MC, Wakelee H, Liberman M, Kato T, Tsuboi M, Lee SH, Chen KN, Dooms C, Majem M, Eigendorff E, Martinengo GL, Bylicki O, Rodríguez-Abreu D, Chaft JE, Novello S, Yang J, Arunachalam A, Keller SM, Samkari A, Gao S. Neoadjuvant pembrolizumab plus chemotherapy followed by adjuvant pembrolizumab compared with neoadjuvant chemotherapy alone in patients with early-stage non-small-cell lung cancer (KEYNOTE-671): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet (London, England), 404:1240-1252, 2024
51. Igawa Y, Yoshida T, Makihara R, Torasawa M, Tateishi A, Matsumoto Y, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. Association between lorlatinib blood concentration and adverse events and clinical impact of dose modification. Lung cancer (Amsterdam, Netherlands), 196:107954, 2024
52. Maggie Liu SY, Jin ZY, Deng JY, Zhong SM, Ahn MJ, Horinouchi H, Li Y, Wu YL. Drug development and evidence for lung cancer targeted therapy in Eastern Asia. The Lancet regional health. Western Pacific, 49:101090, 2024
53. Sanomachi T, Yoshida T, Higashiyama RI, Satozono Y, Matsunaga S, Kanemitsu Y, Ohe Y. Ileal perforation at the site of peritoneal metastasis with intestinal infiltration by dual EGFR-VEGF pathway inhibition in an EGFR-mutant non-small cell lung cancer patient. International cancer conference journal, 13:374-376, 2024
54. Kato Y, Udagawa H, Matsumoto S, Izumi H, Ohe Y, Kato T, Nishino K, Miyamoto S, Kawana S, Chikamori K, Shingyoji M, Sato Y, Takada Y, Toyozawa R, Azuma K, Tanaka Y, Sakai T, Shibata Y, Sugiyama E, Nosaki K, Zenke Y, Umemura S, Yoh K, Seike M, Goto K. Efficacy of immune checkpoint inhibitors plus platinum-based chemotherapy as 1st line treatment for patients with non-small cell lung cancer harboring HER2 mutations: Results from LC-SCRUM-Asia. Lung cancer (Amsterdam, Netherlands), 197:107992, 2024
55. Hirota R, Sasaki M, Iyama S, Kurihara K, Fukushi R, Obara H, Oshigiri T, Morita T, Nakazaki M, Namioka T, Namioka A, Onodera R, Kataoka-Sasaki Y, Oka S, Takemura M, Ukai R, Yokoyama T, Sasaki Y, Yamashita T, Kobayashi M, Okuma Y, Kondo R, Aichi R, Ohmatsu S, Kawashima N, Ito YM, Kobune M, Takada K, Ishiai S, Ogata T, Teramoto A, Yamashita T, Kocsis JD, Honmou O. Intravenous Infusion of Autologous Mesenchymal Stem Cells Expanded in Auto Serum for Chronic Spinal Cord Injury Patients: A Case Series. Journal of clinical medicine, 13:6072, 2024
56. Tateishi A, Okuma Y, Goto Y, Arakaki M, Igawa YS, Torasawa M, Shinno Y, Yoshida T, Horinouchi H, Yamamoto N, Ohe Y. Key Therapeutic Agents for Thymic Carcinoma in Real-world Clinical Practice. Anticancer research, 44:5501-5513, 2024
57. Yamamoto G, Tanaka K, Kamata R, Saito H, Yamamori-Morita T, Nakao T, Liu J, Mori S, Yagishita S, Hamada A, Shinno Y, Yoshida T, Horinouchi H, Ohe Y, Watanabe SI, Yatabe Y, Kitai H, Konno S, Kobayashi SS, Ohashi A. WEE1 confers resistance to KRAS(G12C) inhibitors in non-small cell lung cancer. Cancer letters, 611:217414, 2024
58. Baba S, Kawasaki T, Hirano S, Nakamura T, Asano T, Okazaki R, Yoshida K, Kawase T, Kurahara H, Oi H, Yokoyama M, Kita J, Imura J, Kinoshita K, Kondo S, Okada M, Satake T, Igawa YS, Yoshida T, Yamaguchi H, Ando Y, Mizunuma M, Ichikawa Y, Hida K, Nishihara H, Kato Y. A noninvasive urinary microRNA-based assay for the detection of pancreatic cancer from early to late stages: a case control study. EClinicalMedicine, 78:102936, 2024
59. Uehara Y, Takeyasu Y, Yoshida T, Tateishi A, Torasawa M, Hosomi Y, Masuda K, Shinno Y, Matsumoto Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N, Ohe Y. Real-world outcomes of treatment strategy between first-line osimertinib, first/second-generation EGFR-TKIs followed by osimertinib and without osimertinib in advanced EGFR-mutant NSCLC. ESMO Real World Data and Digital Oncology, 5:100058, 2024
60. Kitadai R, Okuma Y, Kashima J. Gingival Metastasis of ALK Rearranged Non-Small Cell Lung Cancer. Case reports in oncology, 12:171-177, 2019
