Annual Report 2024
Department of Gastric Surgery
Takaki Yoshikawa, Yukinori Yamagata, Tsutomu Hayashi, Takeyuki Wada, Kenichi Ishizu
Introduction
Our division provides surgical treatment for patients with gastric cancer, adenocarcinoma of esophago-gastric junction, and gastrointestinal stromal tumors (GIST). The aim of our division is to contribute to human health through treatment and research in this field. Our policy to achieve this goal is to (1) provide high quality treatment for patients, (2) efficiently perform clinical tasks, and (3) contribute to developing treatments through clinical trials and clinical research and to provide high quality education.
The Team and What We Do
(1) We conduct daily morning conference to determine the treatment strategy for new patients and those who require any new treatment to provide high quality treatment. All the staff check the endoscopy and CT and determine the tumor staging based on the uniform criteria, and discuss the treatment strategy for each patient based on the problem when applying the standard treatment and the patient's indication to be a candidate for any clinical trial. Surgical morbidity and hospital stay were dramatically improved after introducing the Enhanced Recovery After Surgery program (ERAS) for perioperative care.
(2) We prepared the medical documents for the patients for the uniform explanation and the printed protocol at the conference room and the outpatient clinic to check the applicability to the clinical trial for an efficient clinical task. We also relieve the physicians and residents from the task of preparing the database.
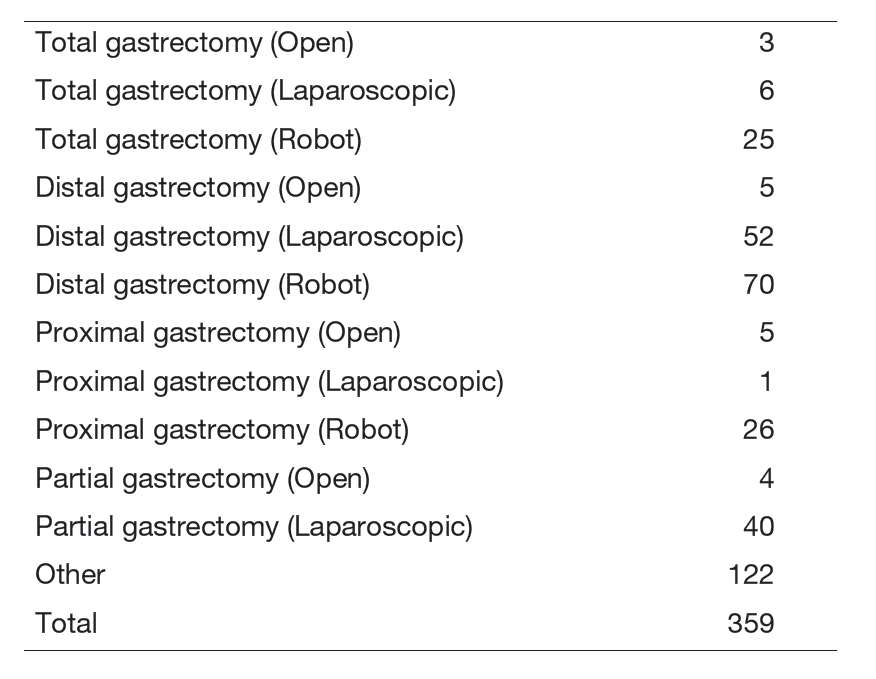
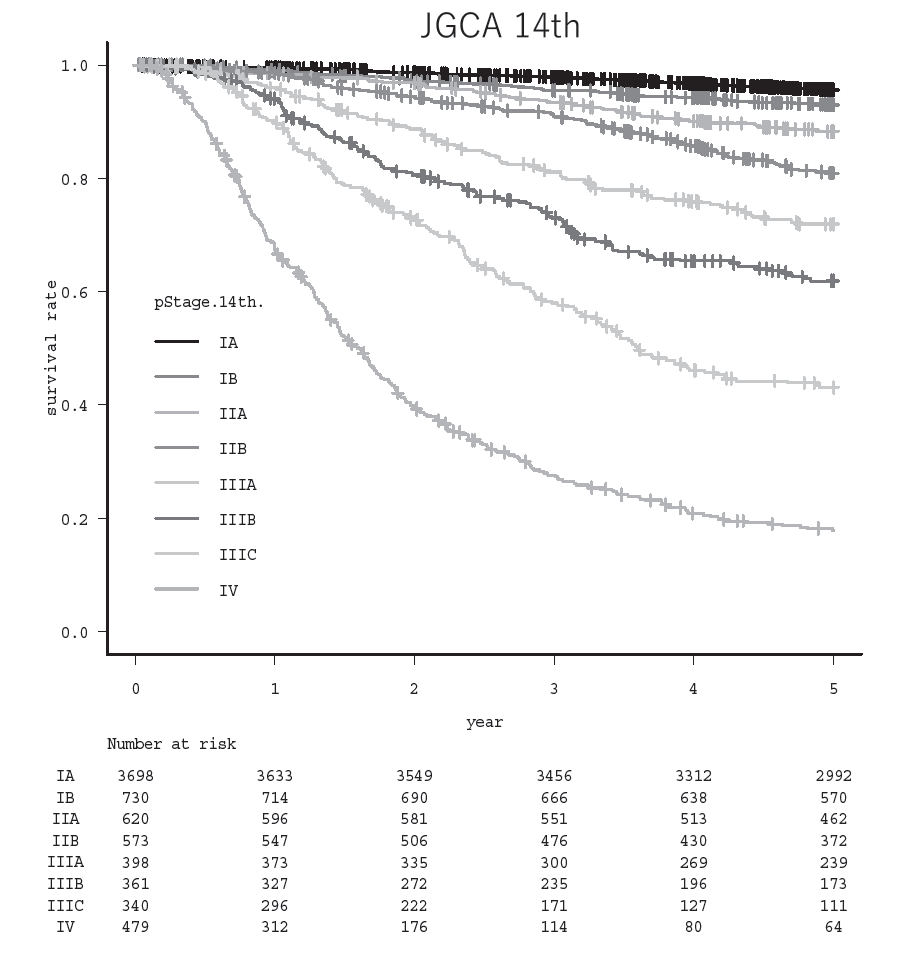
(3) Our division operated a total of 304 cases of gastric cancer, GIST, and others this year. Detailed surgical procedure is shown in Table 1. The proportion of laparoscopic surgery is increased year by year; from 49% (2017) to 65% (2018), 62% (2019), 62% (2020), 74% (2021), 87.7% (2022), 86.1% (2023), and 98.4% (2024) of distal/pylorus-preserving gastrectomy and from 12% (2017) to 21% (2018), 44% (2019), 37% (2020), 46% (2021), 67.8% (2022), 69.0% (2023), and 83% (2024) of total/proximal gastrectomy. Especially, we operated 121 cases of robot assisted-gastrectomy. On the other hand, extended surgery is not rare. We gained experience of numerous cases of D2 plus para-aortic nodal dissection after neoadjuvant chemotherapy for bulky nodal disease or para-aortic nodal metastasis and several left upper abdominal exenteration cases for bulky GIST invading adjacent organs and lower mediastinal dissection through trans-hiatal approach and by right video-assisted trans-thoracic approach for gastric cancer invading the lower esophagus. We can provide any type of surgery for the local control as applicable. Lastly, we presented the overall survival curves of 7,287 patients who received surgery between 2000 and 2019 (Figure 1). The overall 5-year survival rate was 95.5% in stage IA, 93.0% in stage IB, 88.2% in stage IIA, 80.7% in stage IIB, 66.0% in stage IIIA, 54.8% in stage IIIB, 31.9% in stage IIIC, and 18.1% in stage IV following the 15th edition of the Japanese Classification of Gastric Cancer.
Research Activities and Clinical Trials
Ongoing JCOG studies led by our department:
- JCOG1104 (Phase III trial for pathological Stage II gastric cancer, testing the non-inferiority of 6 months of S-1 adjuvant chemotherapy compared to the standard 1 year of S-1; study office: Yoshikawa). The 5-year follow-up was completed in March 2022, and the results were published in Gastric Cancer in June 2024. Subsequently, a letter raising concerns about the results was submitted to the Gastric Cancer editor-in-chief by a Korean medical oncologist, and a rebuttal reply was published in September 2024.
- JCOG1711 (Phase III trial evaluating the non-inferiority of omentum preservation compared to omentectomy in SS/SE gastric cancer; PI: Yoshikawa) completed accrual with 754 cases on March 18, 2025. The second interim analysis is planned within this fiscal year.
- JCOG2301 (Phase III randomized controlled trial on the significance of conversion surgery in initially unresectable cStage IVB/pStage IV gastric cancer responding to chemotherapy; PI: Yoshikawa) was launched in September 2024.
- JCOG1302A2 (prospective registry evaluating the accuracy of preoperative diagnosis for advanced gastric cancer; PI: Yoshikawa, study office: Hayashi). As an associated study, JCOG1302A is investigating prognosis. Manuscripts are under review within the group.
- JCOG2212A (integrated analysis of JCOG0110/0405/1002 to clarify the therapeutic value of para-aortic lymph node dissection; PI: Yoshikawa, study office: Wada). Results were presented at ASCO-GI in January 2025.
- A multi-institutional retrospective study within JCOG gastric cancer group hospitals is ongoing to evaluate the therapeutic value of lymph node dissection in neuroendocrine carcinoma (NEC) (surgical study office: Yamagata). Key results were submitted to and accepted by Gastric Cancer.
- Another retrospective multicenter study evaluating the effect of adjuvant chemotherapy for early gastric cancer requiring extensive lymphadenectomy (PI: Yoshikawa) is also underway. Key results are currently under analysis.
Ongoing JCOG studies (Group Representative: Yoshikawa):
- JCOG1809 (Phase II trial assessing the safety of spleen-preserving splenic hilar lymph node dissection in advanced gastric cancer of the upper greater curvature).
- JCOG1902 (single-arm Phase III trial of endoscopic resection in elderly patients with early gastric cancer).
- JCOG1907 (Phase III superiority trial comparing laparoscopic vs robot-assisted laparoscopic surgery for resectable gastric cancer).
- JCOG2203 (randomized Phase II/III trial of DOS vs FLOT as preoperative chemotherapy for adenocarcinoma of the esophagogastric junction).
- JCOG2204 (randomized Phase II trial exploring the efficacy of FLOT vs DOS as preoperative chemotherapy for large type 3/4 gastric cancers).
Completed accrual / under follow-up JCOG trials:
- JCOG1507 (Phase III superiority trial of postoperative surveillance vs postoperative S-1 adjuvant chemotherapy for elderly Stage II/III gastric cancer) completed accrual in January 2024. The second interim analysis in July 2025 led to early termination for futility. Results will be presented at ASCO-GI in January 2026.
- JCOG1509 (Phase III superiority trial for clinical Stage III gastric cancer: surgery + postoperative adjuvant chemotherapy vs perioperative S-1/oxaliplatin chemotherapy + surgery + adjuvant chemotherapy) completed accrual with 462 patients on March 31, 2025. Currently under follow-up; the first interim analysis is scheduled for FY2025.
- JCOG1704 (Phase II trial of NAC + D3 for advanced nodal metastasis) was published in Gastric Cancer in March 2024. Follow-up is ongoing, with 3-year survival results expected in FY2025.
- JCOG1301C (randomized Phase II trial comparing SP vs SP + trastuzumab as preoperative chemotherapy in HER2-positive, locally advanced gastric cancer with nodal metastasis). Preliminary results were published in Gastric Cancer in May 2024.
Numerous secondary analyses of completed trials have also been reported at conferences and in publications.
Clinical application and innovation:
- Gastric cancer surgery using the da Vinci SP system was initiated in May 2024. A novel hybrid surgical method, MILLER, combining da Vinci SP and laparoscopy, was developed and reported in Asian Journal of Endoscopic Surgery. Safety data from the initial 23 cases were also reported in the same journal.
International collaboration:
- Collaborative research with the Netherlands, UK, Singapore, Germany, and other countries is ongoing. In FY2022, two manuscripts were submitted to Gastric Cancer, both of which were accepted.
International clinical trial:
- Since FY2020, we have participated in an international randomized, double-blind, placebo-controlled Phase III trial evaluating the efficacy and safety of durvalumab combined with FLOT as perioperative therapy for resectable gastric and gastroesophageal junction cancer, followed by durvalumab monotherapy as adjuvant treatment. Final results have been accepted by Journal of Clinical Oncology.
Education
Clinical Practice:
Through open departmental conferences on diagnosis and treatment planning, trainees learn what the current standard treatments are, what problems exist, and what investigational treatments are being developed. They gain experience as second and first assistants to senior staff, as scopists in laparoscopic surgery; and, under the guidance of experienced surgeons, as operating surgeons for standard procedures. In particular, to prepare for obtaining certification from the Japan Society for Endoscopic Surgery, trainees are, in principle, given the opportunity to perform standard laparoscopic distal gastrectomy as the primary surgeon. Regular animal lab training and surgical video conferences are also conducted.
Treatment Development:
All ongoing clinical trial protocols are kept in the conference room and outpatient clinic so that they can be readily referenced during conferences. This allows trainees to learn both standard and investigational treatments in daily practice. In addition, participation as observers in the JCOG Protocol Review Committee and attendance at JCOG Gastric Cancer Group meetings provide them with first-hand experience in the process of treatment development.
Clinical Research:
Under the guidance of the department chair and senior staff, junior staff, oncology fellows, and residents conduct retrospective clinical studies using our departmental database. In fiscal year 2024, a total of five English-language papers were published, including two in Journal of Gastric Cancer, European Journal of Surgical Oncology, and World Journal of Surgery, as well as one in Journal of Gastrointestinal Cancer.
Observers
We received 9 observers from overseas.
Future Prospects
We will continue the high quality treatment, efficient clinical tasks, clinical trials, clinical research, and education in the field of gastric surgery and contribute to the human being.
List of papers published in 2024
Journal
1. Yoshikawa T, Hayashi T, Nishino M, Ogawa R, Fujisaki Y, Honda S, Wada T, Yamagata Y, Seto Y. A new hybrid robotic surgery by minimally invasive laparoscopic and robotic (MILAR) system using da Vinci single-port (SP) in distal gastrectomy for gastric cancer. Asian journal of endoscopic surgery, 18:e13401, 2025
2. Hashimoto I, Komori K, Onuma S, Watanabe H, Suematsu H, Nagasawa S, Kano K, Kawabe T, Aoyama T, Hayashi T, Yamada T, Sato T, Saito A, Ogata T, Cho H, Yoshikawa T, Rino Y, Oshima T. Prognostic impact of adipose tissue loss at 1 month after surgery in patients with gastric cancer. World journal of surgery, 49:472-482, 2025
3. Nishina T, Boku N, Kurokawa Y, Sasaki K, Machida R, Yoshikawa T. Authors' reply to 'RE: A real-world survey on expensive drugs used as first-line chemotherapy in patients with HER2-negative unresectable advanced/recurrent gastric cancer in the stomach cancer study group of the Japan clinical oncology group'. Japanese journal of clinical oncology, 55:89-90, 2025
4. Nishikawa K, Koizumi W, Tsuburaya A, Suzuki M, Morita S, Fujitani K, Akamaru Y, Shimada K, Hosaka H, Nishimura K, Yoshikawa T, Tsujinaka T, Sakamoto J. Differences in efficacy of biweekly irinotecan plus cisplatin versus irinotecan alone in second-line treatment of advanced gastric cancer with or without prior gastrectomy. International journal of clinical oncology, 30:320-329, 2025
5. Kita R, Yasufuku I, Takahashi N, Mizusawa J, Sano Y, Fukuda H, Kurokawa Y, Boku N, Terashima M, Yoshikawa T. Randomized controlled phase III study comparing chemotherapy alone versus conversion surgery after a remarkable response to chemotherapy in patients with initially unresectable cStage IVB or pStage IV gastric cancer (JCOG2301, Conversion study). Japanese journal of clinical oncology, 55:304-310, 2025
6. Yoshikawa T, Hayashi T, Nishino M, Ogawa R, Fujisaki Y, Wada T, Yamagata Y, Seto Y. Short-Term Outcomes of a Novel Hybrid Technique, Minimally Invasive Laparoscopic and Robotic Surgery (MILAR) Using the da Vinci SP System for Gastric Cancer. Asian journal of endoscopic surgery, 18:e70012, 2025
7. Kim TH, Uyama I, Rha SY, Bencivenga M, An J, Wyrwicz L, Koo DH, van Hillegersberg R, Lee KW, Li G, Yoshikawa T, Badgwell B, Lorenzen S, Kim IH, Lee IS, Han HS, Hoon H. Conversion Therapy for Stage IV Gastric Cancer: Report From the Expert Consensus Meeting at KINGCA WEEK 2024. Journal of gastric cancer, 25:133-152, 2025
8. Ishizu K, Takahashi S, Kouno N, Takasawa K, Takeda K, Matsui K, Nishino M, Hayashi T, Yamagata Y, Matsui S, Yoshikawa T, Hamamoto R. Establishment of a machine learning model for predicting splenic hilar lymph node metastasis. NPJ digital medicine, 8:93, 2025
9. Sakon R, Hayashi T, Ogawa R, Nishino M, Ishizu K, Wada T, Yamagata Y, Daiko H, Yoshikawa T. Is Primary Surgery Followed by S-1 Applicable Even for Type 4 or Large Type 3 Gastric Cancer With Positive Peritoneal Lavage Cytology? World journal of surgery, 49:727-733, 2025
10. Ogawa R, Yoshikawa T, Fujisaki Y, Nishino M, Sakon R, Wada T, Hayashi T, Yamagata Y, Seto Y. Risk factors for postoperative liver dysfunction in robot-assisted gastrectomy for gastric cancer. European journal of surgical oncology, 51:109668, 2025
11. Yamagata Y, Yoshikawa T, Sakon R, Ishizu K, Wada T, Hayashi T. Impact of D2 Total Gastrectomy Including Splenectomy for Scirrhous Gastric Cancer in the Era of Effective Adjuvant Chemotherapy. Journal of gastrointestinal cancer, 55:1098-1104, 2024
12. Sodergren SC, Hurley-Wallace A, Vassiliou V, Alkhaffaf B, Batsaikhan B, Darlington AS, Fleitas-Kanonnikof T, Guren MG, Honda M, Kim YW, Kim S, Krishnamurthy MN, Loh SY, Turhal NS, Zhou J, Dennis K, Krishnatry R, Terashima M, Tsironis G, Yoshikawa T, Terada M. Revisiting the use of the EORTC QLQ-STO22 to assess health-related quality of life of patients with gastric cancer: incorporating updated treatment options and cross-cultural perspectives. Gastric cancer, 27:722-734, 2024
13. Sato R, Tokunaga M, Mizusawa J, Sato Y, Ito S, Takahari D, Sano T, Onaya H, Yoshikawa T, Boku N, Terashima M. Clinical impact of skeletal muscle mass change during the neoadjuvant chemotherapy period in patients with gastric cancer: An ancillary study of JCOG1002. World journal of surgery, 48:163-174, 2024
14. Yoshikawa T, Kikko Y, Makino R, Kimijima Y, Nishiyama E, Matsuda Y, Casaes Teixeira B, Tejada M, Carroll R, Hironaka S. Adjuvant and post-recurrent treatment patterns in patients with resectable gastric cancer in japan: a retrospective database cohort study. Gastric cancer, 27:827-839, 2024
15. Hayashi M, Yoshikawa T, Mizusawa J, Hato S, Iwasaki Y, Sasako M, Kawachi Y, Iishi H, Choda Y, Boku N, Terashima M. Prognostic Impact of Post-operative Infectious Complications in Gastric Cancer Patients Receiving Neoadjuvant Chemotherapy: Post Hoc Analysis of a Randomized Controlled Trial, JCOG0501. Journal of gastrointestinal cancer, 55:1125-1133, 2024
16. Ishizu K, Hayashi T, Ogawa R, Nishino M, Sakon R, Wada T, Otsuki S, Yamagata Y, Katai H, Matsui Y, Yoshikawa T. Characteristics of Metachronous Remnant Gastric Cancer After Proximal Gastrectomy: A Retrospective Analysis. Journal of gastric cancer, 24:280-290, 2024
17. Yoshikawa T, Terashima M, Mizusawa J. Reply to letter to the editor: "5-year follow-up results of a JCOG1104 (OPAS-1) phase III noninferiority trial to compare 4 courses and 8 courses of S-1 adjuvant chemotherapy for pathological stage II gastric cancer". Gastric cancer, 27:1155-1156, 2024
18. Hashimoto I, Komori K, Onuma S, Watanabe H, Suematsu H, Nagasawa S, Kano K, Kawabe T, Aoyama T, Hayashi T, Yamada T, Sato T, Ogata T, Cho H, Yoshikawa T, Rino Y, Saito A, Oshima T. Preoperative Visceral-to-Subcutaneous Fat Ratio by Sex as a Predictor of Postoperative Survival in Patients With Gastric Cancer. Anticancer research, 44:3515-3524, 2024
19. Hashimoto I, Komori K, Maezawa Y, Nagasawa S, Kawabe T, Aoyama T, Hayashi T, Yamada T, Sato T, Ogata T, Cho H, Yoshikawa T, Yukawa N, Rino Y, Saito A, Oshima T. Prognostic value of computed tomography‑derived skeletal muscle index and radiodensity in patients with gastric cancer after curative gastrectomy. Oncology letters, 28:458, 2024
20. Wada T, Yoshikawa T, Honda S, Fujisaki Y, Uotani T, Kumamoto Y, Ogawa R, Nishino M, Sakon R, Ishizu K, Hayashi T, Yamagata Y, Seto Y. Para-aortic lymph node dissection following neoadjuvant chemotherapy for type 4 or large type 3 gastric cancer with extensive lymph node metastasis. World journal of surgery, 48:2487-2495, 2024
21. Shoji H, Hirano H, Nojima Y, Gunji D, Shinkura A, Muraoka S, Abe Y, Narumi R, Nagao C, Aoki M, Obama K, Honda K, Mizuguchi K, Tomonaga T, Saito Y, Yoshikawa T, Kato K, Boku N, Adachi J. Phosphoproteomic subtyping of gastric cancer reveals dynamic transformation with chemotherapy and guides targeted cancer treatment. Cell reports, 43:114774, 2024
22. Fujisaki YY, Yoshikawa T, Ogawa R, Nishino M, Wada T, Hayashi T, Yamagata Y, Tokunaga M, Kinugasa Y, Seto Y. Necessity of splenectomy for antral-type scirrhous gastric cancer. European journal of surgical oncology, 108734, 2024
23. Oshima T, Yoshikawa T, Miyagi Y, Morita S, Yamamoto M, Tanabe K, Nishikawa K, Ito Y, Matsui T, Kimura Y, Aoyama T, Ogata T, Cho H, Tsuburaya A, Sakamoto J. Biomarker Study for Selecting Neoadjuvant Chemotherapy Regimens Based on Prognostic Prediction Using Gastric Cancer Biopsy Specimens from a Phase II Randomized Controlled Trial. Anticancer research, 44:4951-4960, 2024
24. Yamada Y, Nagashima K, Azuma M, Masutani M, Ichikawa H, Iwasa S, Takahashi N, Hirano H, Kanato K, Machida N, Kinoshita T, Hata H, Kawakami H, Takahari D, Boku N, Kurokawa Y, Terashima M, Yoshikawa T, Sekine S, Hiraoka N. Predictive and prognostic value of excision repair cross-complementing group 1 in patients with advanced gastric cancer. BJC reports, 2:18, 2024
