Annual Report 2024
Department of Gastrointestinal Medical Oncology
Ken Kato, Atsuo Takashima, Hirokazu Shoji, Hidekazu Hirano, Toshiharu Hirose, Natsuko Okita, Nozomu Ogura, Shotaro Yamaguchi, Shin Kameishi
Introduction
The Department of Gastrointestinal Medical Oncology specializes in the medical treatment of malignant tumors originating in the digestive tract, including gastric cancer, esophagogastric junction cancer, colorectal cancer, and rare cancers of the digestive tract such as anal canal cancer, gastrointestinal stromal tumors (GISTs), and small intestine cancer. They collaborate with the Departments of Gastrointestinal Surgery, Colorectal Surgery, and Gastrointestinal Endoscopy to provide comprehensive treatment.
The Team and What We Do
In the outpatient setting, the staff takes the lead, with each member responsible for clinic sessions for 2 to 3 days per week. In the inpatient setting, a team consisting of full-time physicians, cancer specialist trainees, and residents collaboratively provide patient care. Treatment strategies are determined through departmental conferences, as well as weekly conferences specifically for gastric cancer and colorectal cancer cases.
In the fiscal year 2024, there were a total of 357 new patients. The breakdown is as follows: 100 with gastric cancer, 170 with colorectal cancer, 5 with appendiceal cancer, 13 with small bowel carcinoma, 26 with GIST, 16 with neuroendocrine neoplasms, and 17 with other types of cancer. There were 999 inpatients (including overlaps), with the breakdown as follows: 290 with gastric cancer, 319 with colorectal cancer, 19 with appendiceal cancer, 46 with small bowel carcinoma, 21 with GIST, 97 with neuroendocrine neoplasms, and 207 with other types of cancer. In the fiscal year 2024, 147 patients were enrolled in clinical trials or studies.
Research Activities
With the aim of establishing better treatment methods, our department actively engages in clinical research. Our staff members play a central role in conceiving new trials for the Japan Clinical Oncology Group (JCOG) and the West Japan Oncology Group (WJOG), actively participating in the development of new standard treatments. As an institution, we proactively enroll patients in these trials, contributing to the establishment of novel treatment standards.
Furthermore, we participate in domestic and international clinical trials ranging from Phase I to Phase III, not only for late-stage treatment development but also for the development of new drugs, starting from the planning stage. Additionally, we conduct physician-led trials for second-line treatment of gastric cancer. We actively engage in translational research (TR) in collaboration with research institutes and other basic research facilities and propose new physician-led trials to pharmaceutical companies.
The department delivered 97 scientific presentations, of which 62 were at international conferences. In addition, 60 papers were published, comprising 55 original articles in English and 2 English review articles. Among these, 26 publications listed members of our department as either the first author or the corresponding author.
Clinical Trials
In the fiscal year 2024, a total of 133 patients were enrolled in clinical trials (Table 1). Of these, 56 patients participated in 24 clinical trials for gastric cancer, 71 patients in 17 trials for colorectal cancer, and 6 patients in 3 trials for other gastrointestinal malignancies.
Table 1. Clinical trials and the number of registered patients in the fiscal year 2024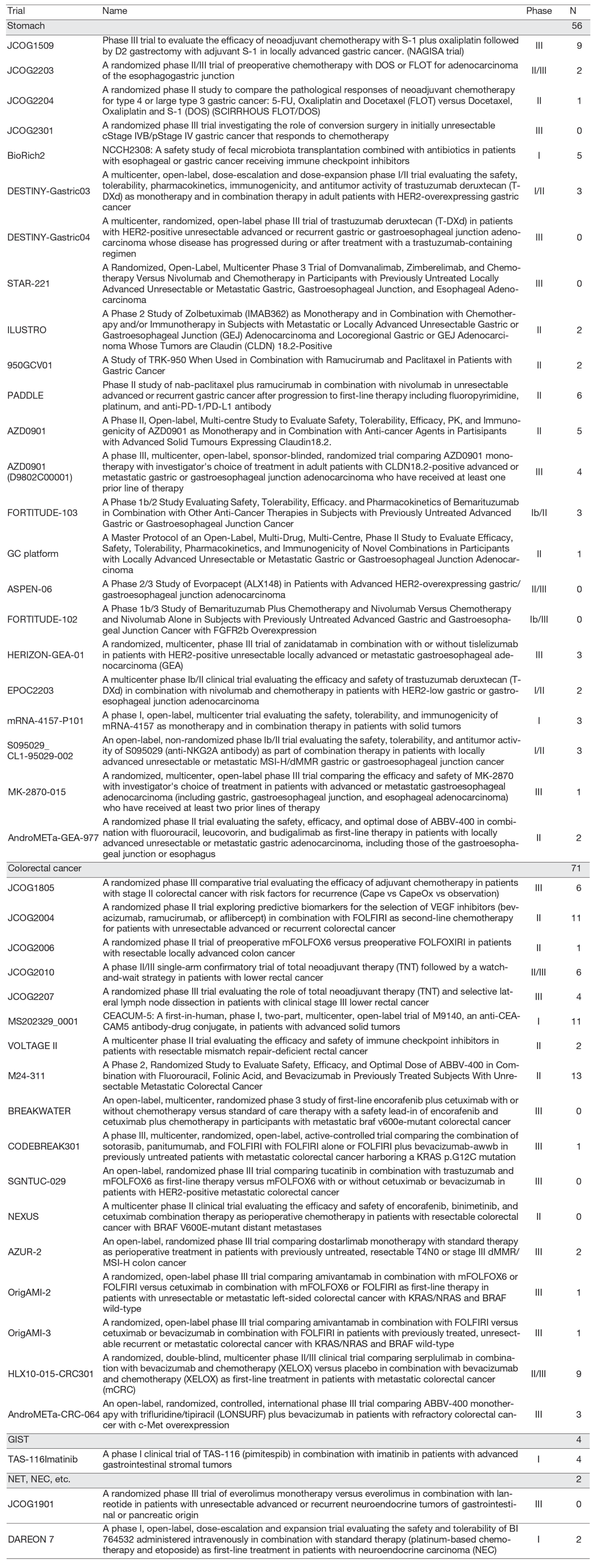

Education
In patient care, full-time physicians collaborate with cancer specialist trainees/residents to form teams and provide guidance through their interactions with assigned patients. Additionally, cancer specialist trainees/residents are assigned research topics and receive active guidance in developing, managing, presenting at conferences, and writing papers related to clinical research. In the fiscal year 2024, cancer specialist trainees/residents in our department presented 22 abstracts as first authors, including 12 at international conferences, and authored 7 articles in English journals.
Future Prospects
We are dedicated to excellence in patient care, research, and education, with the goal of continuously improving treatment outcomes for gastrointestinal cancers. In patient care, we adhere to standard treatments as a foundation while also striving for innovation and continuous improvement by addressing unmet patient needs, always aiming for "better than before." We are also committed to fostering collaboration with local hospitals.
In research, we not only lead clinical trials and multicenter collaborative studies using our specialized expertise but also effectively leverage translational research (TR) in partnership with research institutes. Our objective is to develop treatment approaches that have the potential to become the next standard of care. We actively participate in web conferences with industry partners and international researchers to incorporate a broader perspective into our developmental efforts.
In education, our aim is not only to train residents in cancer pharmacotherapy but also to cultivate young oncologists with a strong research mindset. We are committed to developing the next generation of researchers who are not only highly specialized but also equipped with critical and creative thinking skills.
List of papers published in 2024
Journal
1. Hirano H, Kataoka K, Yamaguchi T, Wagner AD, Shimada Y, Inomata M, Hamaguchi T, Takii Y, Mizusawa J, Sano Y, Shiomi A, Shiozawa M, Ohue M, Adachi T, Ueno H, Ikeda S, Komori K, Tsukamoto S, Takashima A, Kanemitsu Y. Sex differences in toxicities and survival outcomes among Japanese patients with Stage III colorectal cancer receiving adjuvant fluoropyrimidine monotherapy: A pooled analysis of 4 randomized controlled trials (JCOG2310A). European journal of cancer (Oxford, England : 1990), 214:115139, 2025
2. Komori A, Hironaka S, Kadowaki S, Mitani S, Furuta M, Kawakami T, Makiyama A, Takegawa N, Sugiyama K, Hirano H, Ando T, Matsushima T, Chida A, Kashiwada T, Komoda M, Matsumoto T, Oda H, Yabusaki H, Kawakami H, Yamazaki K, Boku N, Hyodo I, Yoshimura K, Muro K. Prevalence and clinicopathological features of microsatellite instability-high metastatic or recurrent gastric and esophagogastric junction cancer: WJOG13320GPS. Gastric cancer, 28:301-308, 2025
3. Fujii H, Shoji H, Hirano H, Hirose T, Okita N, Takashima A, Kato K. Exploring novel therapeutic targets in small bowel adenocarcinoma: insights from claudin 18.2, nectin-4, and HER3 expression analysis. ESMO open, 10:104098, 2025
4. Hirano H, Takahashi N, Amanuma Y, Suzuki N, Takahari D, Kawakami T, Kudo-Saito C, Nagashima K, Boku N, Kato K, Shoji H. Phase II trial of nab-paclitaxel plus ramucirumab in combination with nivolumab for unresectable advanced or recurrent gastric cancer after progression on first-line treatment including fluoropyrimidine, platinum, and anti-PD-1/PD-L1 antibody (PADDLE). BMC cancer, 25:201, 2025
5. Kang YK, Ryu MH, Di Bartolomeo M, Chau I, Yoon H, Kim JG, Lee KW, Oh SC, Takashima A, Kryzhanivska A, Chao Y, Evesque L, Schenker M, McGinn A, Zhao Y, Lee J, Wyrwicz L, Boku N. Rivoceranib, a VEGFR-2 inhibitor, monotherapy in previously treated patients with advanced or metastatic gastric or gastroesophageal junction cancer (ANGEL study): an international, randomized, placebo-controlled, phase 3 trial. Gastric cancer, 27:375-386, 2024
6. Matsumoto T, Yamamoto Y, Kotaka M, Masuishi T, Tsuji Y, Shoji H, Hirata K, Tsuduki T, Makiyama A, Izawa N, Takahashi N, Tsuda M, Yasui H, Ohta T, Kito Y, Otsu S, Hironaka S, Yamazaki K, Boku N, Hyodo I, Yoshimura K, Muro K. A Phase II Study of FOLFIRI Plus Ziv-Aflibercept After Trifluridine/Tipiracil Plus Bevacizumab in Patients with Metastatic Colorectal Cancer: WJOG 11018G. Targeted oncology, 19:181-190, 2024
7. Wakatsuki T, Ishizuka N, Hironaka S, Minashi K, Kadowaki S, Goto M, Shoji H, Hirano H, Nakayama I, Osumi H, Ogura M, Chin K, Yamaguchi K, Takahari D. Exploratory analysis of serum HER2 extracellular domain for HER2 positive gastric cancer treated with SOX plus trastuzumab. International journal of clinical oncology, 29:801-812, 2024
8. Oshima K, Shoji H, Boku N, Hirano H, Okita N, Takashima A, Kato K, Kudo-Saito C. CRP and soluble CTLA4 are determinants of anti-PD1 resistance in gastrointestinal cancer. American journal of cancer research, 14:1174-1189, 2024
9. Arai H, Tsuda T, Sunakawa Y, Shimokawa M, Akiyoshi K, Tokunaga S, Shoji H, Kunieda K, Kotaka M, Matsumoto T, Nagata Y, Mizukami T, Mizuki F, Danenberg KD, Boku N, Nakajima TE. Switching from FOLFIRI plus cetuximab to FOLFIRI plus bevacizumab based on early tumor shrinkage in RAS wild-type metastatic colorectal cancer: A phase II trial (HYBRID). Cancer medicine, 13:e7107, 2024
10. Takashima A, García-Alfonso P, Manneh R, Beşen AA, Hong YS, Cuyle PJ, Yanez P, Burge M, Yoshino T, Kim TW, Cui K, Li C, Jain R, Adelberg D, Taieb J. Olaparib with or without bevacizumab versus bevacizumab plus a fluoropyrimidine as maintenance therapy in advanced colorectal cancer: The randomized phase 3 LYNK-003 study. European journal of cancer (Oxford, England : 1990), 205:114036, 2024
11. Qiu MZ, Oh DY, Kato K, Arkenau T, Tabernero J, Correa MC, Zimina AV, Bai Y, Shi J, Lee KW, Wang J, Poddubskaya E, Pan H, Rha SY, Zhang R, Hirano H, Spigel D, Yamaguchi K, Chao Y, Wyrwicz L, Disel U, Cid RP, Fornaro L, Evesque L, Wang H, Xu Y, Li J, Sheng T, Yang S, Li L, Moehler M, Xu RH. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first line treatment for advanced gastric or gastro-oesophageal junction adenocarcinoma: RATIONALE-305 randomised, double blind, phase 3 trial. BMJ (Clinical research ed.), 385:e078876, 2024
12. Kadono T, Iwasa S, Hirose T, Hirano H, Okita N, Shoji H, Takashima A, Kato K. Impact of immune checkpoint inhibitors on survival outcomes in advanced gastric cancer in Japan: A real-world analysis. Cancer medicine, 13:e7401, 2024
13. Fukuda K, Osumi H, Yoshinami Y, Ooki A, Takashima A, Wakatsuki T, Hirano H, Nakayama I, Ouchi K, Sawada R, Fukuoka S, Ogura M, Takahari D, Chin K, Shoji H, Okita N, Kato K, Ishizuka N, Boku N, Yamaguchi K, Shinozaki E. Efficacy of anti-epidermal growth factor antibody rechallenge in RAS/BRAF wild-type metastatic colorectal cancer: a multi-institutional observational study. Journal of cancer research and clinical oncology, 150:369, 2024
14. Kudo-Saito C, Imazeki H, Ozawa H, Kawakubo H, Hirano H, Boku N, Kato K, Shoji H. Targeting SNCA in the treatment of malignant ascites in gastrointestinal cancer. Translational oncology, 48:102075, 2024
15. Boku N, Omori T, Shitara K, Sakuramoto S, Yamaguchi K, Kato K, Kadowaki S, Tsuji K, Ryu MH, Oh DY, Oh SC, Rha SY, Lee KW, Chung IJ, Sym SJ, Chen LT, Chen JS, Bai LY, Nakada T, Hagihara S, Makino R, Nishiyama E, Kang YK. Nivolumab plus chemotherapy in patients with HER2-negative, previously untreated, unresectable, advanced, or recurrent gastric/gastroesophageal junction cancer: 3-year follow-up of the ATTRACTION-4 randomized, double-blind, placebo-controlled, phase 3 trial. Gastric cancer, 27:1287-1301, 2024
16. Takashima A, Hamaguchi T, Mizusawa J, Nagashima F, Ando M, Ojima H, Denda T, Watanabe J, Shinozaki K, Baba H, Asayama M, Hasegawa S, Masuishi T, Nakata K, Tsukamoto S, Katayama H, Nakamura K, Fukuda H, Kanemitsu Y, Shimada Y. Oxaliplatin Added to Fluoropyrimidine/Bevacizumab as Initial Therapy for Unresectable Metastatic Colorectal Cancer in Older Patients: A Multicenter, Randomized, Open-Label Phase III Trial (JCOG1018). Journal of clinical oncology, 42:3967-3976, 2024
17. Takashima A, Ishiguro M, Sasaki K, Machida R, Nagashima F, Imaizumi J, Hamaguchi T, Yamamoto Y, Masuishi T, Asayama M, Ueno H, Shinozaki K, Kudo T, Machida N, Matsuoka H, Ishida H, Yamaguchi T, Nogami H, Yamada T, Takegawa N, Kito Y, Tonoike Y, Sawada R, Tsukamoto S, Kanemitsu Y. Real-world treatment costs of first-line treatment for metastatic colorectal cancer: a survey of the JCOG colorectal cancer study group. Japanese journal of clinical oncology, 54:1107-1114, 2024
18. Shoji H, Hirano H, Nojima Y, Gunji D, Shinkura A, Muraoka S, Abe Y, Narumi R, Nagao C, Aoki M, Obama K, Honda K, Mizuguchi K, Tomonaga T, Saito Y, Yoshikawa T, Kato K, Boku N, Adachi J. Phosphoproteomic subtyping of gastric cancer reveals dynamic transformation with chemotherapy and guides targeted cancer treatment. Cell reports, 43:114774, 2024
19. Fujii H, Hirano H, Shiraishi K, Shoji H, Hirose T, Okita N, Takashima A, Koyama T, Kato K. Comprehensive Genomic Assessment of Advanced-Stage GI Stromal Tumors Using the Japanese National Center for Cancer Genomics and Advanced Therapeutics Database. JCO precision oncology, 8:e2400284, 2024
20. Yamada Y, Nagashima K, Azuma M, Masutani M, Ichikawa H, Iwasa S, Takahashi N, Hirano H, Kanato K, Machida N, Kinoshita T, Hata H, Kawakami H, Takahari D, Boku N, Kurokawa Y, Terashima M, Yoshikawa T, Sekine S, Hiraoka N. Predictive and prognostic value of excision repair cross-complementing group 1 in patients with advanced gastric cancer. BJC reports, 2:18, 2024
21. Takamizawa S, Hirano H, Takashima A, Shoji H, Hirose T, Okita N, Shiraishi K, Sekine S, Takamizawa Y, Kanemitsu Y, Kato K. Clinicopathological and molecular features of claudin-18 isoform 2 expression in patients with colorectal cancer: a single-center retrospective study. Therapeutic advances in medical oncology, 16:17588359241286774, 2024
