Annual Report 2024
Department of Hematology
Koji Izutsu, Wataru Munakata, Suguru Fukuhara, Noriko Iwaki, Shinichi Makita, Tetsuro Ochi
Introduction
The Department of Hematology at the National Cancer Center Hospital is dedicated to providing comprehensive care for patients with hematologic malignancies including leukemia, lymphoma, and myeloma. We strive to offer state-of-the-art diagnostic and therapeutic options, while continuously advancing novel treatment strategies and diagnostic methods through clinical trials and translational research. Our Lymphoma Program has a long-standing tradition of pioneering clinical research in adult T-cell leukemia/ lymphoma and other lymphoid malignancies, and continues to lead the development of innovative agents and cellular therapies for hematologic malignancies in Japan.
The Team and What We Do
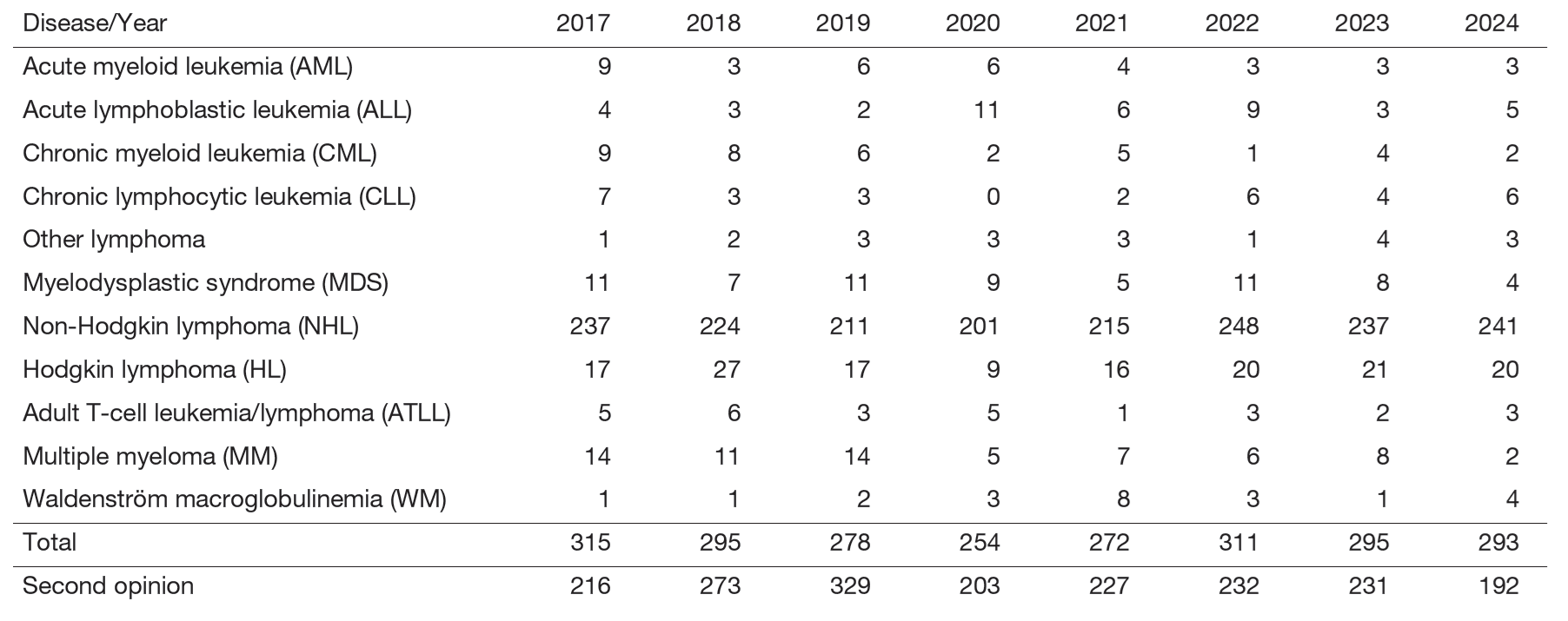
We provide advanced diagnostic procedures and comprehensive care for patients with hematologic malignancies, including leukemia, lymphoma, myelodysplastic syndrome, and multiple myeloma, both on an outpatient and inpatient basis (Table 1). Our therapeutic approaches encompass medical chemotherapy, immunotherapy, and chimeric antigen receptor (CAR) T-cell therapy. We work closely with specialists from the Departments of Hematopoietic Stem Cell Transplantation, Radiation Oncology, and Experimental Therapeutics to ensure multidisciplinary management. In addition, our collaborations with the Department of Pathology and Department of Clinical Laboratories enable state-of-the-art diagnostic support to guide treatment decisions.
Research Activities
We conduct a wide range of clinical trials to develop novel treatments for hematologic malignancies, including global first-in-human, global phase 2-3, and domestic phase 1-3 studies, as well as investigator-initiated clinical trials. In 2023, we completed enrolment of our investigator-initiated phase 2 study of atezolizumab monotherapy in patients with relapsed/refractory extranodal NK/T-cell lymphoma. The results of this study contributed to the submission of an application for label expansion atezolizumab in Japan in 2024 for this indication. In addition, HemeSight®, the first gene panel test for hematologic malignancies in Japan, was approved in September 2024. Our department led the clinical implementation study of this test, which represented a major milestone in the introduction of genomic diagnostics into routine hematology practice. In 2018, in collaboration with other departments, we launched the MASTER KEY HEM registration study for rare hematologic malignancies incorporating biomarker analysis, which has continued to expand. Our department is also deeply engaged in the activities of the Japan Clinical Oncology Group - Lymphoma Study Group (JCOG LSG) and the Japan Adult Leukemia Study Group (JALSG). In 2024, we authored or co-authored 43 peer-reviewed articles in English related to hematologic malignancies.
Clinical Trials
In 2024, we conducted 59 corporate-sponsored clinical trials, including numerous global studies, one first-in-human study, and six new studies that started from April 2024 to March 2025. In addition, we actively participated in several investigator-initiated clinical trials of JCOG and JALSG.
Education
We offer a structured training program for young hematologists and oncologists. Many of our trainees and alumni are now actively contributing to academic societies and playing leading roles in the fields of hematology throughout Japan.
Future Prospects
The Department of Hematology will continue to address unmet needs in the treatment of hematological malignancies. We are particularly focused on developing new therapies for rare hematologic malignancies, including uncommon lymphoma subtypes. Our future initiatives will emphasize the integration of genome-based medicine, as genomic information has become essential for disease classification according to the latest WHO classification of hematologic malignancies, prognostic assessment, and the selection of targeted therapies.
List of papers published in 2024
Journal
1. Kita R, Shimada K, Machida R, Sekita T, Sasaki K, Ishitsuka K, Fukuda H, Maruyama D, Munakata W, Nagai H. Multicenter randomized phase III study of high-dose therapy with autologous stem cell transplantation versus observation for patients with newly diagnosed peripheral T-cell lymphoma who achieved complete metabolic response after induction therapy (JCOG2210, TRANSFER study). Japanese journal of clinical oncology, 55:183-188, 2025
2. Hosoba R, Fukuhara S, Kogure Y, Takano K, Shibata M, Horiuchi T, Kobayashi S, Makita S, Iwaki N, Munakata W, Maeshima AM, Kimura F, Kataoka K, Izutsu K. Whole exome sequencing analysis of a patient with myeloid/lymphoid neoplasms with eosinophilia and tyrosine kinase gene fusions with ETV6::LYN fusion gene. Annals of hematology, 104:809-813, 2025
3. Munakata W, Kumode T, Goto H, Fukuhara N, Shimoyama T, Takeuchi M, Kawakita T, Kubo K, Sawa M, Uchida T, Mishima Y, Ichii M, Hanaya M, Matsumoto A, Kuriki M, Seike T, Izutsu K, Ishizawa K. A phase II study of zandelisib in patients with relapsed or refractory indolent non-Hodgkin lymphoma: ME-401-K02 study. British journal of haematology, 206:541-550, 2025
4. Ri M, Iida S, Saito K, Saito Y, Maruyama D, Asano A, Fukuhara S, Tsujimura H, Miyazaki K, Ota S, Fukuhara N, Negoro E, Kuroda J, Yoshida S, Ohtsuka E, Norifumi T, Tabayashi T, Takayama N, Saito T, Suzuki Y, Harada Y, Mizuno I, Yoshida I, Maruta M, Takamatsu Y, Katsuya H, Yoshimitsu M, Minami Y, Kanato K, Munakata W, Nagai H. Lipidomic profiling of plasma from patients with multiple myeloma receiving bortezomib: an exploratory biomarker study of JCOG1105 (JCOG1105A1). Cancer chemotherapy and pharmacology, 95:29, 2025
5. Tsutsué S, Makita S, Asou H, Mathew A, Kado Y, Idehara K, Kim SW, Ainiwaer D. Real-world assessment to estimate multiple attributes related to treatment cost driver for mantle cell lymphoma in Japan by econometric modeling. BMC health services research, 25:149, 2025
6. Izutsu K, Fukuhara N. JSH practical guidelines for hematological malignancies, 2023: II. Lymphoma-1. Follicular lymphoma (FL). International journal of hematology, 121:577-585, 2025
7. Tsutsué S, Makita S, Asou H, Wada S, Lee WS, Ainiwaer D, Idehara K, Aoyagi SS, Kim SW, Taylor T. Cost drivers associated with autologous stem-cell transplant (ASCT) in patients with relapsed/refractory diffuse large B-cell lymphoma in a Japanese real-world setting: A structural equation model (SEM) analysis 2012-2022. PloS one, 20:e0317439, 2025
8. Nato Y, Miyazaki K, Maruyama D, Takahashi H, Sunami K, Murakami S, Negoro E, Miyazawa Y, Choi I, Okada T, Takayama N, Tomita N, Momose S, Kaneda Y, Yoshida M, Gomyo H, Toyama K, Nishikori M, Saito A, Hiraga J, Masunari T, Takahashi N, Makiyama J, Suzuki T, Tsunemine H, Takizawa J, Kato T, Masaki Y, Fukuhara N, Okamoto M, Tawara I, Asano N, Ohshima K, Izutsu K, Kato K, Suzuki R, Yamaguchi M. Treatments and Outcomes of Newly Diagnosed CD5-Positive Diffuse Large B-Cell Lymphoma: A Multi-Institutional Observational Study. Hematological oncology, 43:e70047, 2025
9. Tan YH, Yoon DH, Davies AJ, Buske C, Boo YL, Somasundaram N, Lim F, Ong SY, Jeyasekharan A, Izutsu K, Kim WS, Chan JY. Improving access to chimeric antigen receptor T-cells for refractory or relapsing diffuse large B cell lymphoma therapy in Asia. Discover oncology, 16:181, 2025
10. Izutsu K, Ishikawa T, Shimada K, Kubo K, Kondo T, Fujimoto K, Fujisaki T, Kurahashi S, Nagafuji K, Sakai R, Jo T, Nakazato T, Sunami K, Kasahara S, Cohen A, Takai M, Zhong J, Takeuchi M. Zanubrutinib in Japanese treatment-naive and relapsed/refractory patients with Waldenström macroglobulinemia and CLL/SLL. International journal of hematology, 121:483-493, 2025
11. Fujimoto A, Miyazaki K, Yakushijin K, Fujino T, Munakata W, Ejima Y, Maruyama D, Kubota N, Maeda T, Takizawa J, Hiramoto N, Takeuchi M, Sakai R, Fukuhara N, Taguchi S, Asano N, Yamaguchi M, Suzuki R. Improved prognosis of advanced-stage extranodal NK/T-cell lymphoma: results of the NKEA-Next study. Leukemia, 39:909-916, 2025
12. Kobayashi T, Ishizawa K, Machida R, Sadachi R, Sasaki K, Kim H, Kataoka K, Munakata W, Fukuhara N, Nagai H. Obinutuzumab maintenance versus observation for patients with newly diagnosed high tumor burden follicular lymphoma who achieved complete metabolic response after obinutuzumab plus bendamustine induction therapy: a multicenter, randomized, phase III study (JCOG2008, MAIN study). Japanese journal of clinical oncology, 55:666-671, 2025
13. Watanabe T, Tobinai K, Wakabayashi M, Maruyama D, Yamamoto K, Kubota N, Shimada K, Asagoe K, Yamaguchi M, Ando K, Ogura M, Kuroda J, Suehiro Y, Matsuno Y, Tsukasaki K, Nagai H. R-CHOP treatment for patients with advanced follicular lymphoma: Over 15-year follow-up of JCOG0203. British journal of haematology, 204:849-860, 2024
14. Saito T, Nagai H, Izutsu K, Ando K, Igarashi T, Izumi T, Ohashi Y, Kamiyama S, Ishizawa K, Tobinai K. A phase II Japanese trial of 90-minute rituximab infusion for untreated B-cell lymphoma. Japanese journal of clinical oncology, 54:444-451, 2024
15. Izutsu K, Ubukawa K, Morishita T, Onishi Y, Ishizawa K, Fujii Y, Kimura N, Yokochi M, Naoe T. Glasdegib with intensive/nonintensive chemotherapy in Japanese patients with untreated acute myeloid leukemia or high-risk myelodysplastic syndromes. Cancer science, 115:1250-1260, 2024
16. O'Connor OA, Ko BS, Wang MC, Maruyama D, Song Y, Yeoh EM, Manamley N, Tobinai K. Pooled analysis of pralatrexate single-agent studies in patients with relapsed/refractory peripheral T-cell lymphoma. Blood advances, 8:2601-2611, 2024
17. Takizawa J, Suzuki R, Izutsu K, Kiguchi T, Asaoku H, Saburi Y, Masunari T, Utsunomiya A, Takeuchi K, Nakamura N, Ohshima K, Gruber M, Jäger U, Aoki S, Suzumiya J. Characteristics of chronic lymphocytic leukemia in Japan: Comprehensive analysis of the CLLRSG-01 study. International journal of hematology, 119:686-696, 2024
18. Yamaguchi K, Yamamoto H, Izutsu K, Yuasa M, Kaji D, Nishida A, Ishiwata K, Takagi S, Yamamoto G, Asano-Mori Y, Uchida N, Taniguchi S. Fatal outcome of BK virus encephalitis in an allogeneic stem cell transplantation recipient. Journal of infection and chemotherapy, 30:1166-1169, 2024
19. Kato K, Izutsu K, Nishikori M, Shibayama H, Maeda Y, Yoshimura K, Tateishi U, Miyamoto T, Matsuda Y, Ishikawa J, Rai S, Takahashi T, Yamauchi T, Matsumura I, Akashi K, Kanakura Y, Suzumiya J. End-of-treatment 18[F]-FDG PET can predict early progression in patients receiving bendamustine-rituximab for follicular lymphoma in first relapse: a prospective West Japan hematology Study Group (W-JHS) NHL01 trial. International journal of hematology, 119:677-685, 2024
20. Kawasaki A, Hatake K, Matsumura I, Izutsu K, Hoshino T, Akamatsu A, Kakuuchi A, Tobinai K. Post-marketing surveillance of the safety and effectiveness of nivolumab for classic Hodgkin lymphoma in Japan. International journal of hematology, 119:667-676, 2024
21. Watanabe T, Matsuno Y, Wakabayashi M, Maruyama D, Yamamoto K, Kubota N, Shimada K, Asagoe K, Yamaguchi M, Ando K, Ogura M, Kuroda J, Suehiro Y, Tsukasaki K, Tobinai K, Nagai H. Analyzing the risk factors for disease progression within 2 years and histological transformation in patients treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone as first-line treatment: A 15-year follow-up of patients with advanced follicular lymphoma in JCOG0203. Hematological oncology, 42:e3272, 2024
22. Tsutsué S, Makita S, Asou H, Matsuda H, Yamaura R, Taylor TD. Cost-effectiveness analysis 3L of axicabtagene ciloleucel vs tisagenlecleucel and lisocabtagene maraleucel in Japan. Future oncology (London, England), 20:1333-1349, 2024
23. Izutsu K, Kato H, Sekiguchi N, Fujisaki T, Kawakita T, Obara N, Matsue K, Nishimoto M, Hatayama T, Inagaki M, Fujikawa E. A phase 2, open-label study of ibrutinib plus rituximab in Japanese patients with Waldenstrom's macroglobulinemia. International journal of hematology, 120:80-90, 2024
24. Ito Y, Marouf A, Kogure Y, Koya J, Liévin R, Bruneau J, Tabata M, Saito Y, Shingaki S, Yuasa M, Yamaguchi K, Murakami K, Weil R, Vavasseur M, Andrieu GP, Latiri M, Veleanu L, Dussiot M, André I, Joshi A, Lagresle-Peyrou C, Magerus A, Chaubard S, Lavergne D, Bachy E, Brunet E, Fataccioli V, Brouzes C, Laurent C, de Leval L, Traverse-Glehen A, Bossard C, Parrens M, Meignin V, Philippe L, Rossignol J, Suarez F, Michot JM, Tournilhac O, Damaj G, Lemonnier F, Bôle-Feysot C, Nitschké P, Tesson B, Laurent C, Molina T, Asnafi V, Watatani Y, Chiba K, Okada A, Shiraishi Y, Tsukita S, Izutsu K, Miyoshi H, Ohshima K, Sakata S, Dobashi A, Takeuchi K, Sanada M, Gaulard P, Jaccard A, Ogawa S, Hermine O, Kataoka K, Couronné L. Comprehensive Genetic Profiling Reveals Frequent Alterations of Driver Genes on the X Chromosome in Extranodal NK/T-cell Lymphoma. Cancer research, 84:2181-2201, 2024
25. Morschhauser F, Dahiya S, Palomba ML, Martin Garcia-Sancho A, Reguera Ortega JL, Kuruvilla J, Jäger U, Cartron G, Izutsu K, Dreyling M, Kahl B, Ghesquieres H, Ardeshna K, Goto H, Barbui AM, Abramson JS, Borchmann P, Fleury I, Mielke S, Skarbnik A, de Vos S, Kamdar M, Karmali R, Viardot A, Farazi T, Fasan O, Lymp J, Vedal M, Nishii R, Avilion A, Papuga J, Kumar J, Nastoupil LJ. Lisocabtagene maraleucel in follicular lymphoma: the phase 2 TRANSCEND FL study. Nature medicine, 30:2199-2207, 2024
26. Rai S, Kim WS, Ando K, Choi I, Izutsu K, Tsukamoto N, Maruyama D, Tsukasaki K, Kuroda J, Ando J, Hidaka M, Koh Y, Kato H, Uchida T, Yang DH, Ishitsuka K, Ishizawa K, Kim JS, Lee HG, Minami H, Eom HS, Kurosawa M, Lee JH, Lee JS, Lee WS, Nagai H, Shindo T, Yoon DH, Yoshida S, Gillings M, Onogi H, Tobinai K. Long-term efficacy and safety of tucidinostat in patients with relapsed or refractory peripheral T-cell lymphoma: final analysis of phase IIb results. Haematologica, 109:3357-3362, 2024
27. Tsutsué S, Makita S, Asou H, Matsuda H, Yamaura R. Cost-utility analysis of second-line axicabtagene ciloleucel versus standard of care in Japan based on the ZUMA-7 trial. Future oncology (London, England), 20:2279-2291, 2024
28. Aruga Y, Ikeda C, Matsushita H, Makita S, Fukuhara S, Munakata W, Izutsu K, Matsui H. The kappa/lambda ratio of surface immunoglobulin light chain as a valuable parameter for MRD assessment in CLL with atypical immunophenotype. Scientific reports, 14:13452, 2024
29. Takahashi Y, Kaimi Y, Taniguchi H, Ochi T, Makino H, Makita S, Iwaki N, Fukuhara S, Munakata W, Izutsu K, Maeshima AM. Clinicopathological differences between T-lymphoblastic leukemia/lymphoma, early T-precursor lymphoblastic leukemia/lymphoma, and mixed-phenotype acute leukemia with T lineage: An analysis of 41 adult cases. Human pathology, 150:78-85, 2024
30. Takizawa J, Teshima T, Ennishi D, Ichikawa S, Suzuki R, Kojima A, Takahashi Y, Hayashi N, Kawasumi H, Murayama K, Cheung P, Kawata T, Izutsu K. Safety and efficacy of acalabrutinib and obinutuzumab in treatment-naive chronic lymphocytic leukemia: a Japanese phase 1 study. Leukemia & lymphoma, 65:1586-1594, 2024
31. Yamaguchi M, Fukuhara N, Takizawa J, Ishitsuka K, Yokohama A, Miyazaki K, Nato Y, Ichikawa S, Mitobe M, Shima K, Miyazawa Y, Izutsu K, Suzuki R, Nagai H, Nakamura N. Alemtuzumab monotherapy for T-cell prolymphocytic leukemia: an observational study in Japan. Journal of clinical and experimental hematopathology : JCEH, 64:216-222, 2024
32. Wierda WG, Shah NN, Cheah CY, Lewis D, Hoffmann MS, Coombs CC, Lamanna N, Ma S, Jagadeesh D, Munir T, Wang Y, Eyre TA, Rhodes JM, McKinney M, Lech-Maranda E, Tam CS, Jurczak W, Izutsu K, Alencar AJ, Patel MR, Seymour JF, Woyach JA, Thompson PA, Abada PB, Ho C, McNeely SC, Marella N, Nguyen B, Wang C, Ruppert AS, Nair B, Liu H, Tsai DE, Roeker LE, Ghia P. Pirtobrutinib, a highly selective, non-covalent (reversible) BTK inhibitor in patients with B-cell malignancies: analysis of the Richter transformation subgroup from the multicentre, open-label, phase 1/2 BRUIN study. The Lancet. Haematology, 11:e682-e692, 2024
33. Kusumoto S, Munakata W, Machida R, Terauchi T, Onaya H, Oguchi M, Iida S, Nosaka K, Suzuki Y, Harada Y, Miyazaki K, Maruta M, Fukuhara N, Toubai T, Kubota N, Ohmachi K, Saito T, Rai S, Mizuno I, Fukuhara S, Takeuchi M, Tateishi U, Maruyama D, Tsukasaki K, Nagai H. Interim PET-guided ABVD or ABVD/escalated BEACOPP for newly diagnosed advanced-stage classic Hodgkin lymphoma (JCOG1305). Cancer science, 115:3384-3393, 2024
34. Maeshima AM, Taniguchi H, Takahashi Y, Kaimi Y, Ochi T, Makino H, Makita S, Iwaki N, Fukuhara S, Munakata W, Izutsu K. Heterogeneity or change in cell of origin in diffuse large B-cell lymphomas determined using hans algorithm. Human pathology, 151:105630, 2024
35. Ito T, Kamimura T, Kiguchi T, Kato K, Takenaka R, Kobayashi M, Ito A, Sakai M, Izutsu K. Venetoclax treatment for chronic lymphocytic leukemia/small lymphocytic leukemia in Japan: post-marketing surveillance. International journal of hematology, 120:613-620, 2024
36. Izutsu K, Ando K, Nishikori M, Shibayama H, Goto H, Kuroda J, Kato K, Imaizumi Y, Nosaka K, Sakai R, Abe M, Hojo S, Nakanishi T, Rai S. Tazemetostat for relapsed/refractory B-cell non-Hodgkin lymphoma with EZH2 mutation in Japan: 3-year follow-up for a phase II study. International journal of hematology, 120:621-630, 2024
37. Hatta Y, Izutsu K, Onizuka M, Dobashi N, Hayakawa F, Yamazaki E. JSH practical guidelines for hematological malignancies, 2023: leukemia-3. Acute lymphoblastic leukemia/lymphoblastic lymphoma: ALL/LBL. International journal of hematology, 120:394-413, 2024
38. Kato K, Nakamura S, Wakana A, Koh Y, Izutsu K. Pembrolizumab in Japanese patients with primary mediastinal large B-cell lymphoma: results from the KEYNOTE-A33 study. International journal of clinical oncology, 29:1977-1983, 2024
39. Maruyama D, Jacobsen E, Porcu P, Allen P, Ishitsuka K, Kusumoto S, Narita T, Tobinai K, Foss F, Tsukasaki K, Feldman T, Imaizumi Y, Izutsu K, Morishima S, Yamauchi N, Yuda J, Brammer JE, Kawamata T, Ruan J, Nosaka K, Utsunomiya A, Wang J, Zain J, Kakurai Y, Yamauchi H, Hizukuri Y, Biserna N, Tachibana M, Inoue A, Horwitz SM. Valemetostat monotherapy in patients with relapsed or refractory non-Hodgkin lymphoma: a first-in-human, multicentre, open-label, single-arm, phase 1 study. The Lancet. Oncology, 25:1589-1601, 2024
40. Zinzani PL, Izutsu K, Mehta-Shah N, Barta SK, Ishitsuka K, Córdoba R, Kusumoto S, Bachy E, Cwynarski K, Gritti G, Prica A, Jacobsen E, Feldman T, Guillermin Y, Ennishi D, Yoon DH, Domenech ED, Zain J, Wang J, Kim JS, Poel MV, Jin J, Wu S, Chen Y, Moriyama T, Inoue A, Nakajima K, Horwitz SM. Valemetostat for patients with relapsed or refractory peripheral T-cell lymphoma (VALENTINE-PTCL01): a multicentre, open-label, single-arm, phase 2 study. The Lancet. Oncology, 25:1602-1613, 2024
41. Iguchi T, Nishikori A, Sato Y, Nishimura MF, Iwaki N, Kojima K, Asahara T, Otsuka F, Maeda Y, Hiraki T. Computed tomography findings of idiopathic multicentric Castleman disease subtypes. Journal of clinical and experimental hematopathology : JCEH, 64:292-296, 2024
