Annual Report 2024
Department of Laboratory Medicine
Hirotaka Matsui, Kuniko Sunami, Wataru Takeda, Shigeki Yamazaki, Chiaki Hayashi, Shunsuke Tezuka, Yoji Hashimoto, Yasuo Shibuki, Hiroki Kakishima, Tomokazu Suzuki, Chiaki Ikeda, Arisa Hanai, Satoshi Ito, Yusuke Okui, Sayaka Takeuchi, Kyosuke Tosawa, Mizuho Fujima, Sachiko Kobayashi, Risa Otsuka, Hiromi Otokawa, Noriko Takahashi, Saori Nakabayashi, Yuki Kase, Maemi Souma, Tomoe Ito, Kyoko Orihara, Kaori Ueki, Akino Kino, Kana Katsuragi, Ruriko Machida, Haruka Katagiri, Kana Miyajima, Kayo Okauchi, Nanami Sato, Kenta Takehara, Kanako Kanaizuka, Chihiro Yamasaki, Hiroshi Chigira, Kaori Yamaguchi, Kyoko Osanai, Moemi Kasane, Hideya Matsubayashi, Yu Aruga, Misato Tsubokura, Yuka Yasuno, Haruki Mitsuya, Kaho Matsui, Nozomi Shishido, Aya Mikami, Nanako Fujita, Saori Kobayashi, Shingo Nakajima, Saeko Shirahama, Mei Fukuhara, Kumi Nakatani, Hiyori Yatsu, Chika Tokutake, Madoka Kondo, Mayu Takeno, Sakura Ishida, Ayaka Ichikawa, Yusuke Takai, Daichi Yamada, Haruna Omaru, Noa Suzuki, Yuka Oga, Akari Matsui, Eimi Iwama, Yusuke Ozawa, Fuga Hidaka, Shio Kobayashi, Noki Maezawa, Motoi Miyakoshi, Takako Takada, Takashi Kubo, Satoyo Oda, Haruka Hirakochi, Shigeru Tamura, Megumi Masuda, Takeshi Ogasawara, Rio Yamauchi, Miki Nagasawa, Kazuya Nakanishi, Rituko Toyama, Chieko Nozawa, Naoki Komiya, Kozaburo Endo, Hiroki Murayama, Nanami Watanabe
Introduction
The Department of Laboratory Medicine obtained ISO 15189 accreditation, which is an international standard that specifies requirements for quality and competence specific to clinical laboratories, as of September 2012, and has maintained the certification ever since. In March 2025, the department underwent a transition audit to ISO 15189:2022.
The number of test requests to the department in FY2024 was 6,151,602, an increase of 101.6% from FY2023 (Table 1).
Table 1. Number of clinical tests performed

The Team and What We Do
To address the growing demand for laboratory testing and clinical trial-related work, the Department of Laboratory Medicine has established a cross-sectional operational system and continues to improve efficiency.
Hematology Testing: We conduct high-sensitivity analyses for minimal residual disease in hematopoietic neoplasms, as well as examinations of specimens with low cell counts, including cerebrospinal fluid, ascites, and pleural effusion.
Microbiology Testing: We contribute to hospital infection control through rapid organism identification by mass spectrometry and susceptibility testing for drug-resistant bacteria. In addition, genetic testing for various pathogens, including COVID-19, supports timely and effective patient care.
Genetic Testing: With the increasing use of cancer genome profiling, extensive discussions were held in Expert Panels (Table 2). Preparations are underway to implement gene panel testing for hematopoietic neoplasms.
Table 2. Number of sessions and cancer genomic profiling tests in Expert panel meeting 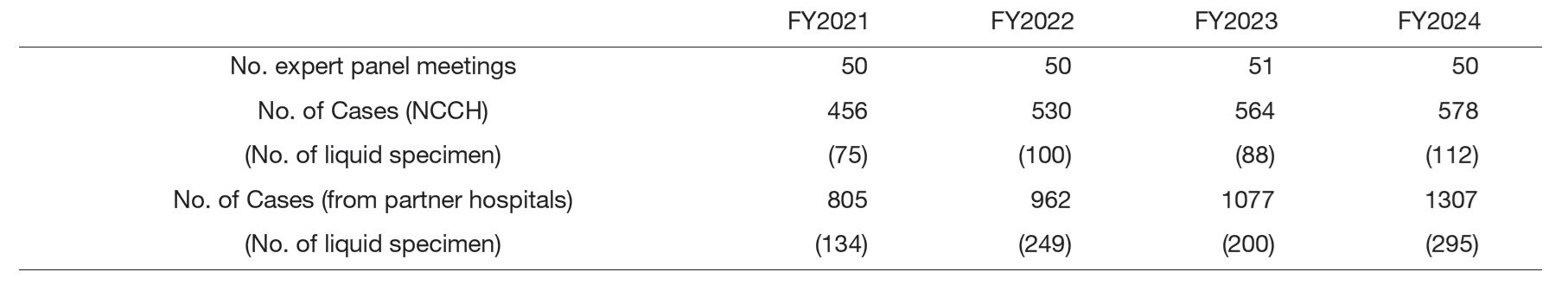

Blood Collection Services: We aim to reduce patient waiting times, targeting fewer than 20 minutes in at least 85% of cases, while ensuring patient safety. The department also supports specimen collection for clinical research, clinical trials, and outpatient treatment centers.
Blood Transfusion Testing and Cell Processing: We have developed a management system for ABO variant transfusions, which are frequently performed in the hospital, and conduct ward rounds in high-demand areas to ensure safe transfusion practice. Continuous support is also provided for chimeric antigen receptor T-cell (CAR-T) therapy.
Physiological Function Testing: Our section actively evaluates the cardiotoxicity of anticancer drugs, diagnoses hepatic sinusoidal obstruction syndrome/veno-occlusive disease (SOS/VOD) after hematopoietic stem cell transplantation, and provides ultrasound examinations prior to breast cancer radiofrequency ablation therapy.
Pathology: Pre-analytical procedures for pathological tissue specimens are carried out in compliance with genomic medicine regulations, contributing to accurate genomic diagnosis and precision treatment.
Research Activities
The Department of Laboratory Medicine has conducted both basic and clinical research into factors affecting test accuracy and has reported findings derived from daily practice and observation.
Tumor Marker Testing: Through collaborative research with clinical departments and manufacturers, we are investigating the stability and clinical usefulness of tumor marker assays.
Cancer Genome Profiling: As the secretariat for the TOP-GEAR project, a comprehensive genome analysis initiative, we are engaged in developing new gene panel tests and conducting clinical research to advance the clinical implementation of whole genome testing.
Hematology: We have established and reported a method for measuring minimal residual disease using flow cytometry.
Blood Transfusion and Cell Therapy: Research is underway on ascites treatment through concentrated ascites filtration therapy (CART), and we also collaborate on clinical research with numerous hospital departments.
Education
Per ISO 15189 education, training, and training procedures, the department fosters personnel who can reliably provide quality-assured and accurate test results in each of the laboratory sections. The department also actively supports academic activities, including taking qualifying examinations, conference presentations, and writing papers, aiming to train clinical laboratory technicians with professional and educational backgrounds. Additionally, under the guidance of the Chief of Laboratory Medicine, we provide structured support for clinical laboratory technologists who pursue master’s and doctoral programs at graduate school while continuing their professional duties. For physicians, we maintain a continuous training framework by accepting residents and equipping them with the expertise required to accurately interpret cancer genome profiling test results.
For those who wish to become clinical laboratory technicians, the department actively accepts students as on-site trainees and appeals to them for clinical laboratory work.
Future Prospects
As an ISO 15189-accredited testing facility, the department guarantees the quality and capabilities meeting international standards and promotes cooperation in clinical trials and clinical studies. With regard to the operations of the clinical laboratory, the testing framework is being reorganized to achieve greater operational efficiency. Additionally, the department is working to increase the personnel who specialize in cell therapy and flow cytometry analyses through professional education to expand these operations' capacity. Regarding cancer genome medicine, the department continues to manage the expert panels in response to the increased number of cancer genome profiling tests. Through carrying out these tasks, the department will establish a clinical laboratory to contribute valuable information from each clinical department and provide it ourselves.
List of papers published in 2024
Journal
1. Takigawa W, Yoshida H, Kitamura S, Tokutake C, Kondo M, Fujima M, Shibuki Y, Kobayashi-Kato M, Tanase Y, Uno M, Ishikawa M. Intraoperative peritoneal cytology for cervical gastric-type adenocarcinoma: Cytopathology and clinical impact. Cancer cytopathology, 133:e22915, 2025
2. Sekita T, Asano N, Kubo T, Totsuka H, Mitani S, Hattori N, Yoshida A, Kobayashi E, Komiyama M, Ushijima T, Nakayama R, Nakamura M, Kawai A, Ichikawa H. Early separation and parallel clonal selection of dedifferentiated and well-differentiated components in dedifferentiated liposarcoma. Neoplasia (New York, N.Y.), 59:101074, 2025
3. Bando H, Naito Y, Yamada T, Fujisawa T, Imai M, Sakamoto Y, Saigusa Y, Yamamoto K, Tomioka Y, Takeshita N, Sunami K, Futamura M, Notake C, Aoki S, Okano K, Yoshino T. A prospective study comparing highly qualified Molecular Tumor Boards with AI-powered software as a medical device. International journal of clinical oncology, 30:172-179, 2025
4. Matsushita K, Ishige T, Watanabe K, Akahane T, Tanimoto A, Yoshimoto M, Yamakuchi M, Hashiguchi T, Okugawa Y, Ikejiri M, Yamaguchi T, Yamasaki T, Takeda M, Hibi M, Akiyama N, Shimizu K, Hashimoto N, Sato H, Tanaka Y, Amari F. Importance of EQA/PT for the detection of genetic variants in comprehensive cancer genome testing. Scientific reports, 15:1036, 2025
5. Ma J, Matsui H, Shao L. Hematopoietic stress in stem cell homeostasis and disease pathogenesis. Front Hematol, 4:1571674, 2025
6. Mochizuki A, Shiraishi K, Honda T, Higashiyama RI, Sunami K, Matsuda M, Shimada Y, Miyazaki Y, Yoshida Y, Watanabe SI, Yatabe Y, Hamamoto R, Kohno T. Passive Smoking-Induced Mutagenesis as a Promoter of Lung Carcinogenesis. Journal of thoracic oncology, 19:984-994, 2024
7. Takamizawa S, Koyama T, Sunami K, Sudo K, Hirata M, Kubo T, Tao K, Cho H, Narita Y, Kato K, Yamazaki N, Ohe Y, Okusaka T, Matsui Y, Ogawa C, Yonemori K, Yamamoto N. Identification of barriers to implementation of precision oncology in patients with rare cancers. Cancer science, 115:2023-2035, 2024
8. Kubo T, Sunami K, Koyama T, Kitami M, Fujiwara Y, Kondo S, Yonemori K, Noguchi E, Morizane C, Goto Y, Maejima A, Iwasa S, Hamaguchi T, Kawai A, Namikawa K, Arakawa A, Sugiyama M, Ohno M, Yoshida T, Hiraoka N, Yoshida A, Yoshida M, Nishino T, Furukawa E, Narushima D, Nagai M, Kato M, Ichikawa H, Fujiwara Y, Kohno T, Yamamoto N. The impact of rare cancer and early-line treatments on the benefit of comprehensive genome profiling-based precision oncology. ESMO open, 9:102981, 2024
9. Aruga Y, Ikeda C, Matsushita H, Makita S, Fukuhara S, Munakata W, Izutsu K, Matsui H. The kappa/lambda ratio of surface immunoglobulin light chain as a valuable parameter for MRD assessment in CLL with atypical immunophenotype. Scientific reports, 14:13452, 2024
10. Matsui H. DDX41 and its unique contribution to myeloid leukemogenesis. Oncotarget, 15:442-443, 2024
11. Fukuda M, Mukohara T, Kuwata T, Sunami K, Naito Y. Efficacy of Trametinib in Neurofibromatosis Type 1-Associated Gastrointestinal Stromal Tumors: A Case Report. JCO precision oncology, 8:e2300649, 2024
12. Nishino T, Yumura M, Sunami K, Kubo T, Ichikawa H, Yasuda T, Furukawa E, Nagai M, Yatabe Y, Kato M, Kohno T. Leveraging non-coding regions to guarantee the accuracy of small-sized panel-based tumor mutational burden estimates. Cancer science, 115:4045-4053, 2024
13. Inoue H, Kuroda JI, Fujioka Y, Hata N, Mizoguchi M, Yoshii D, Sueyoshi H, Takeshima Y, Fujimoto K, Shinojima N, Sunami K, Mikami Y, Nakamura H, Mukasa A. Drug-resistant BRAF V600E-mutant recurrent pleomorphic xanthoastrocytoma, CNS WHO Grade 3 successfully resolved with incidental discontinuation of combined BRAF and MEK inhibitor therapy. Surgical neurology international, 15:417, 2024
14. Shiotsu H, Sueta D, Shinriki S, Ryu M, Usuku H, Nakata M, Uchiumi T, Tsujita K, Matsui H. miR-4485-5p in large extracellular vesicles as a new potent biomarker for diagnosis of deep vein thrombosis. Lab Med Int, 3:95-107, 2024
