Annual Report 2024
Clinical Research Support Office
Noboru Yamamoto, Kenichi Nakamura
• Clinical Research Coordinating Division
Hideki Ueno, Miki Ito, Chie Miyano, Asako Sakamoto, Sho Murata, Mari Takahashi, Hiroko Kawaguchi, Shinobu Araki, Chiharu Nakano, Asako Kamikawa, Kimiyo Yoshii, Hiroko Takagi, Maki Takayasu, Aya Matsumoto, Yukako Takasaki, Ran Obara, Harue Ui, Kayo Mukai, Nahoko Hiraoka, Hatsuki Ohno, Chikano Yashiro, Yukari Nishiyama, Azumi Seko, Rie Goto, Marie Komagata, Aya Shiroichi, Anna Yashima, Miki Tamaki, Saki Yoshizawa, Yumi Nagashima, Yumi Tada, Asami Kaido, Chiaki Nishikawa, Marina Hirooka, Yuko Shinoda, Aya Kato, Natsumi Fujii, Maki Moriya, Sumika Oka, Kazumi Kojima, Kana Miura, Marie Ito, Ai Kobayakawa, Maya Niigawa, Hiroe Takayama, Mari Kondo, Tomoko Jinbo, Shiho Nishino, Chie Moteki, Kimiko Sega, Nobuko Ushirozawa, Yoshiko Kanazu, Yoko Ebihara, Harumi Maruno, Junko Horie, Eiko Mimata, Kaori Matsushita, Yuki Sezaki, Chiaki Yamamoto, Yoshiko Miyahara, Ken Kato, Keiko Wakakuwa, Mayumi Ikeda, Satomi Nakamori, Harumi Mochizuki, Azusa Komada, Yuri Toyoyama
• Research Management Division
Natsuko Okita, Tomomi Hata, Tetsuya Sasaki, Takako Hitomi, Mitsumi Terada, Syoko Ishiguro, Aayako Miyata, Tomozo Yamada, Anri Uchimura, Li Chia Chien, Hisahiro Ito, Mamiko Kawasaki, Hitomi Okuma, Satoshi Kawashima, Masahiko Ichimura, Chiharu Mizoguchi, Yayoi Ando, Naoko So, Masako Inaba, Reiko Isomura, Kanako Kondo, Nami Shirakawa, Eri Tsutui, Mamiko Nakata, Sachie Kawabata, Eiko Yorikane, Yoshie Shuda, Kenta Anjo, Kaori Izumino, Kazumi Ono, Hiroshi Katayama, Hayato Ogura, Keita Sasaki, Yusuke Sano, Ryosuke Kita, Noriko Mitome, Tetsuya Sekita, Yuko Tominaga, Hisako Ohi, Ayaha Kuriyama, Emi Iwata, Naoko Murata, Toshiyuki Minemura, Junki Mizusawa, Gakuto Ogawa, Ryunosuke Machida, Ryo Sadachi, Masayuki Yokoyama, Taro Shibata, Aya Kuchiba, Shogo Nomura, Akihiro Hirakawa, Kohei Uemura, Riku Kajikawa, Shogo Ando, Yuya Ishikawa, Naomi Konishi, Yuko Minami, Shintaro Iwamoto, Nobuyuki Wakui, Yuki Konda
• Data Management Division
Haruhiko Fukuda, Harumi Kaba, Hiromi Katsuki, Yumi Oshikiri, Yayoi Mitsumori, Tomoko Kojima, Yukari Hoshina, Nobuko Okamura, Takashi Makiuchi, Keiko Ohata, Masahisa Kamikura, Yukari Nagasaka, Kazumi Kurishita, Yoko Kai, Ayako Kumazawa, Mai Naito, Takashi Fuwa, Emi Nakamura, Tomoko Takayama, Keiko Suto
Introduction
The Clinical Research Support Office supports clinical research conducted under the leadership of investigators in the National Cancer Center Hospital (NCCH). Support activities include protocol writing, central/local data management, statistical design and analysis, in-house/on-site monitoring, audits, patient recruitment, and other coordinating jobs. We also provide local support for industry-sponsored registration-directed trials and investigator-initiated registration-directed trials conducted at the NCCH.
The Team and What We Do
- Clinical Research Coordinating Division
The Clinical Research Coordinating Section and the Clinical Trial Administration Section support a lot of industry-sponsored registration-directed trials as well as investigator-initiated registration-directed trials.
The Biobank and Translational Research Support Section has routinely obtained informed consent to participate as an NCC biobank (NCCBB) donor from patients who consult with the NCCH for the first time. CRCs in this section coordinate translational research in several ways, such as assistance for registration for clinical trials, logistics for pathological specimens, data collection for case report forms, and coordination between sections.
- Research Management Division
The Research Management Division is in charge of central research support functions: i) International clinical trial management, ii) Investigator-initiated registration-directed trial (Chiken) management, iii) Monitoring and consultation, iv) Multi-institutional clinical trial support, v) Biostatistics, vi) Medical device and pharmaceutical affairs consultation. The division has the capability to support various types of clinical trials covering both early phase ones including first-in-human trials and late phase multi-institutional trials. For the early phase trials, the division mainly offers comprehensive study management, site visit monitoring and safety information management. One of the strengths of the division is that it can coordinate not only domestic trials but also international investigator-initiated registration-directed trials. The multi-institutional trial support function works as the Japan Clinical Oncology Group (JCOG) Operations Office which engages in protocol development, manuscript drafting, study coordination, etc., for mainly late phase trials.
- Data Management Division
The Data Management Division is responsible for central data management and in-house study monitoring in investigator-initiated clinical trials for cancer therapeutic development. The Data Management Section supports early phase cancer trials mainly for drug development including registration trials which are led by physicians in the NCCH. The Multi-institutional Data Management Section supports mostly late development multi-modality multi-institutional phase II or phase III trials for adult solid cancer conducted by the Japan Clinical Oncology Group (JCOG).
Clinical Trials
- Clinical Research Coordinating Division
The number of registration-directed clinical trials is increasing year by year, and the division supported 547 registration-directed trials including 53 investigator-initiated registration-directed trials in FY 2024 (Table 1).
Table 1. Supported Trials in the Clinical Research Coordinating Division in FY 2024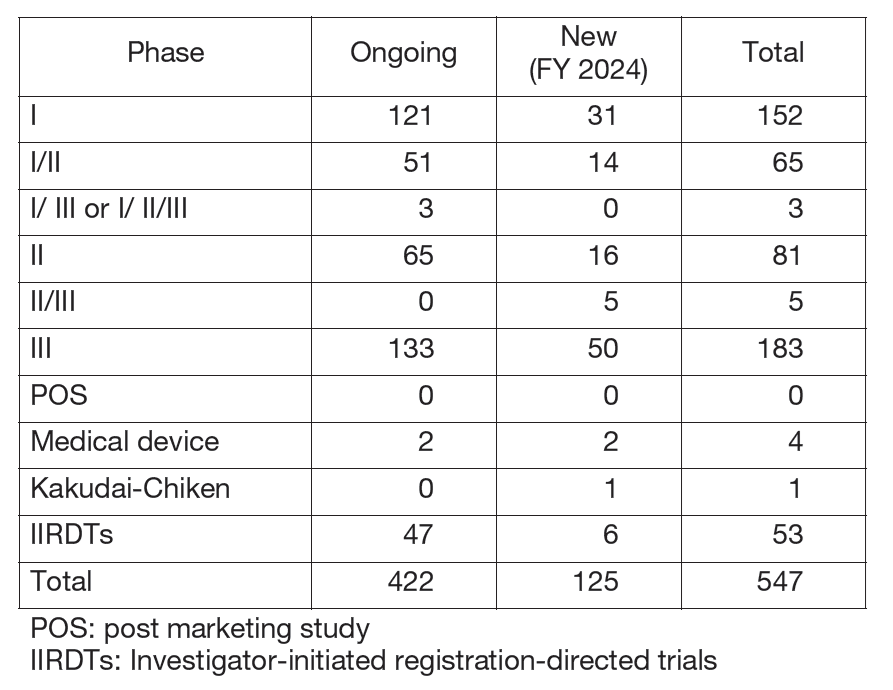

- Research Management Division
The priority areas of the division are genomic medicine, rare cancer and international trials. As of the end of the fiscal year 2024, the Research Management Division supported 19 investigator-initiated registration-directed trials, 14 clinical trials using the Advanced Medical Care system and four trials under patient-proposed health services. This division has been in charge of the coordinating office of rare cancer registry study with basket sub-studies (MASTER KEY Project). This division also supports an international investigator-initiated registration-directed trial (PATHWAY trial), and 4 international investigator-initiated research.
- Data Management Division
The Data Management Section supports 14 IND trials (6 open, 0 in preparation, 8 on follow-up) and 11 non-IND studies (7 open, 0 in preparation, 4 on follow-up). The Multi-institutional Data Management Section supports 106 trials (47 open, 14 in preparation, 45 on follow-up) including 2 open IND trials as the JCOG Data Center.
Education
- Clinical Research Coordinating Division
The staff members received not only day-to-day on-the-job training but also in-house educational seminars in order to learn how to support clinical trials including investigator-initiated registration-directed trials.
- Research Management Division
The staff members received not only day-to-day on-the-job training but also various seminars related to clinical trials held within the National Cancer Center or scientific meetings in order to learn how to support clinical trials including investigator-initiated registration trials.
- Data Management Division
Data managers in the division are encouraged to attend the various seminars related to research ethics and clinical trials held within the National Cancer Center, in-house lectures organized by the Clinical Research Support Office, scientific meetings and seminars organized by the Japan Society of Clinical Trials and Research or the Ministry of Health, Labour and Welfare and so on. The division is also continuously promoting on-the-job training of data managers to teach them clinical trial methodology, data management and research ethics.
Future Prospects
- Clinical Research Coordinating Division
The number of supported clinical trials is increasing, and the supported area covered by CRCs will be expanded to include not only registration trials but also other investigator-initiated clinical trials. Therefore, expansion of CRC staff members is highly anticipated. In view of the plan for the NCCH, all members of this office will work together to contribute to reinforcing clinical research capabilities of the NCCH and to making this office a valuable unit for all members of our hospital. The Biobank and Translational Research Support Section will improve the quality of the NCC biobank’s informatics and storage of serum. The section also aims to establish a support system for higher quality and quantity.
- Research Management Division
Since the number of investigator-initiated registration-directed trials has increased, reinforcement of staff resources is urgently needed. In response to this increase, this division will reinforce the support function for various clinical trials including international clinical trials, investigator-initiated registration-directed trials, Advanced Medical Care system trials, patient-proposed health services trials, and a new type of clinical trial (platform trials). Since 2023, two investigator-initiated registration-directed trials utilizing decentralized clinical trial elements have been under preparation. This division will also establish optimal ways to cope with the Clinical Research Act and revised Ethical Guidelines.
- Data Management Division
The Data Management Division has introduced a web-based electronic data capturing (EDC) system and is promoting standardization of all aspects of data management, such as data formats, case report forms and monitoring reports for increased data integrity, and cost effectiveness of day-to-day work.
List of papers published in 2024
Journal
1. Sekino Y, Sonoda Y, Shibahara I, Mizusawa J, Sasaki K, Sekita T, Ichikawa M, Igaki H, Kinoshita M, Kumabe T, Shibahara J, Ichimura K, Arakawa Y, Fukuda H, Narita Y. Protocol digest of a phase III randomized trial of gross total resection versus possible resection of fluid-attenuated inversion recovery-hyperintense lesion on MRI for newly diagnosed supratentorial glioblastoma: JCOG2209 (FLAMINGO). Japanese journal of clinical oncology, 55:75-79, 2025
2. Matsuda S, Takeuchi H, Kato K, Machida R, Ito Y, Tsubosa Y, Daiko H, Koyanagi K, Ogata T, Fukuda T, Fujita T, Abe T, Bamba T, Watanabe M, Kawakubo H, Shibuya Y, Otsubo D, Kakisita T, Hashimoto T, Sasaki K, Kitagawa Y. Prognostic Impact of Thoracic Duct Resection in Patients Who Underwent Transthoracic Esophagectomy Following Neoadjuvant Therapy for Esophageal Squamous Cell Carcinoma: Exploratory Analysis of JCOG1109. Annals of surgical oncology, 32:293-301, 2025
3. Nakagawa K, Watanabe SI, Wakabayashi M, Yotsukura M, Mimae T, Hattori A, Miyoshi T, Isaka M, Endo M, Yoshioka H, Tsutani Y, Isaka T, Maniwa T, Nakajima R, Suzuki K, Aokage K, Saji H, Tsuboi M, Okada M, Asamura H, Sekino Y, Nakamura K, Fukuda H. Risk Factors for Locoregional Relapse After Segmentectomy: Supplementary Analysis of the JCOG0802/WJOG4607L Trial. Journal of thoracic oncology, 20:157-166, 2025
4. Kita R, Shimada K, Machida R, Sekita T, Sasaki K, Ishitsuka K, Fukuda H, Maruyama D, Munakata W, Nagai H. Multicenter randomized phase III study of high-dose therapy with autologous stem cell transplantation versus observation for patients with newly diagnosed peripheral T-cell lymphoma who achieved complete metabolic response after induction therapy (JCOG2210, TRANSFER study). Japanese journal of clinical oncology, 55:183-188, 2025
5. Kita R, Ito K, Machida R, Sekino Y, Nakamura N, Nakajima Y, Saito T, Imano N, Fukuda H, Ito Y, Mizowaki T. Randomized phase III study comparing re-irradiation stereotactic body radiotherapy and conventional radiotherapy for painful spinal metastases: Japan Clinical Oncology Group study JCOG2211 (RESCORE study). Japanese journal of clinical oncology, 55:189-193, 2025
6. Okuno T, Morizane C, Mizusawa J, Yanagimoto H, Kobayashi S, Imaoka H, Terashima T, Kawakami H, Sano Y, Okusaka T, Ikeda M, Ozaka M, Miwa H, Todaka A, Shimizu S, Mizuno N, Sekimoto M, Sano K, Tobimatsu K, Katanuma A, Gotoh K, Yamaguchi H, Ishii H, Furuse J, Ueno M. Influence of major hepatectomy on gemcitabine-based chemotherapy for recurrent biliary tract cancer after surgery: a subgroup analysis of JCOG1113. International journal of clinical oncology, 30:83-91, 2025
7. Sekino Y, Hishida T, Yoshioka H, Wakabayashi M, Mitome N, Shiono S, Kenmotsu H, Nosaki K, Aokage K, Horinouchi H, Fukuda H, Ohe Y, Watanabe SI. Protocol summary of a randomized phase III study: comparing systemic therapy with and without debulking surgery (primary tumour resection) for clinical stage IVA (cT1-2bN0-1M1a) non-small cell lung cancer with radiologically undetermined pleural dissemination JCOG2103 (DEBULK-LUNG). Japanese journal of clinical oncology, 55:176-182, 2025
8. Funauchi Y, Tsukushi S, Hiraga H, Sakamoto A, Kunisada T, Nagano A, Hiraoka K, Kikuta K, Yonemoto T, Ae K, Kawai A, Endo M, Sano Y, Machida R, Sekita T, Fukuda H, Oda Y, Ozaki T, Tanaka K. Study protocol: randomized phase III trial of neo-adjuvant and adjuvant chemotherapy vs. immediate surgery and adjuvant chemotherapy for localized soft tissue sarcoma: Japan Clinical Oncology Group study JCOG2102 (NACLESS). Japanese journal of clinical oncology, 55:297-303, 2025
9. Hirano H, Kataoka K, Yamaguchi T, Wagner AD, Shimada Y, Inomata M, Hamaguchi T, Takii Y, Mizusawa J, Sano Y, Shiomi A, Shiozawa M, Ohue M, Adachi T, Ueno H, Ikeda S, Komori K, Tsukamoto S, Takashima A, Kanemitsu Y. Sex differences in toxicities and survival outcomes among Japanese patients with Stage III colorectal cancer receiving adjuvant fluoropyrimidine monotherapy: A pooled analysis of 4 randomized controlled trials (JCOG2310A). European journal of cancer (Oxford, England : 1990), 214:115139, 2025
10. Kita R, Yasufuku I, Takahashi N, Mizusawa J, Sano Y, Fukuda H, Kurokawa Y, Boku N, Terashima M, Yoshikawa T. Randomized controlled phase III study comparing chemotherapy alone versus conversion surgery after a remarkable response to chemotherapy in patients with initially unresectable cStage IVB or pStage IV gastric cancer (JCOG2301, Conversion study). Japanese journal of clinical oncology, 55:304-310, 2025
11. Fukushi K, Imaoka H, Ikeda M, Mizusawa J, Morizane C, Okusaka T, Kobayashi S, Sasahira N, Shimizu S, Yamazaki K, Okano N, Miwa H, Hara K, Satoi S, Sano K, Sakai K, Sugimoto R, Nakamura K, Terashima T, Ozaka M, Ueno M. Assessment of heterogeneity according to hospital or medical experience factors in outcomes of chemotherapy for advanced biliary tract cancer: a post-hoc analysis of JCOG1113. Japanese journal of clinical oncology, 55:355-361, 2025
12. Ri M, Iida S, Saito K, Saito Y, Maruyama D, Asano A, Fukuhara S, Tsujimura H, Miyazaki K, Ota S, Fukuhara N, Negoro E, Kuroda J, Yoshida S, Ohtsuka E, Norifumi T, Tabayashi T, Takayama N, Saito T, Suzuki Y, Harada Y, Mizuno I, Yoshida I, Maruta M, Takamatsu Y, Katsuya H, Yoshimitsu M, Minami Y, Kanato K, Munakata W, Nagai H. Lipidomic profiling of plasma from patients with multiple myeloma receiving bortezomib: an exploratory biomarker study of JCOG1105 (JCOG1105A1). Cancer chemotherapy and pharmacology, 95:29, 2025
13. Shinomiya H, Matsuura K, Onimaru R, Ohkoshi A, Saito Y, Tachibana H, Shiga K, Ueda T, Asada Y, Uemura H, Beppu T, Seto A, Yasumatsu R, Nakahira M, Omura G, Asakage T, Minami S, Fujii T, Hirayama Y, Yoshida D, Nakamura K, Sasaki K, Mizusawa J, Fukuda H, Homma A. Dose-finding and efficacy confirmation trial of the superselective intra-arterial infusion of cisplatin and concomitant radiation therapy for locally advanced maxillary sinus cancer (JCOG1212): final analysis. International journal of clinical oncology, 30:489-496, 2025
14. Tsukushi S, Tanaka K, Kunisada T, Machida R, Takenaka S, Kawai A, Katagiri H, Takeyama M, Endo M, Hayashi K, Nakayama R, Hatano H, Emori M, Yoshida S, Kojima T, Sakamoto A, Imanishi J, Kita R, Ozaki T, Iwamoto Y. Clinicopathological background of local recurrence in high grade sarcoma of the extremity with preoperative chemotherapy: a supplementary analysis of JCOG0304. Japanese journal of clinical oncology, 55:603-609, 2025
15. Shibuki T, Ikeda M, Yokoyama M, Sano Y, Furuse J, Kobayashi S, Ohba A, Todaka A, Horie Y, Shioji K, Kanai M, Nishina T, Kumamoto Y, Fujimori N, Katanuma A, Takayama Y, Tsumura H, Miwa H, Ozaka M, Ueno M. Depth of response in patients with locally advanced pancreatic cancer treated with first-line chemotherapy: A supplementary analysis of JCOG1407. Pancreatology, 25:275-283, 2025
16. Kobayashi T, Ishizawa K, Machida R, Sadachi R, Sasaki K, Kim H, Kataoka K, Munakata W, Fukuhara N, Nagai H. Obinutuzumab maintenance versus observation for patients with newly diagnosed high tumor burden follicular lymphoma who achieved complete metabolic response after obinutuzumab plus bendamustine induction therapy: a multicenter, randomized, phase III study (JCOG2008, MAIN study). Japanese journal of clinical oncology, 55:666-671, 2025
17. Sato Y, Mizusawa J, Katayama H, Doki Y, Takiguchi S, Kurokawa Y, Katai H, Suzuki K, Saji H, Aokage K, Kobayashi H, Arimoto T, Kasamatsu T, Sano T, Ito S, Fukuda H. Validation of the Japan Clinical Oncology Group postoperative complications criteria against CTCAE for evaluation of postoperative complications (JCOG1903A). European journal of surgical oncology, 51:109669, 2025
18. Ando Y, Yanagisawa S, Ohno M, Kurihara H, Ito K, Ichimura M, Anjo K, Inaba M, Nagasaka Y, Sadachi R, Shibata T, Okita N, Nakamura K, Matsumoto H, Yoshii Y, Narita Y. Efficacy of radioactive hypoxia-targeting therapeutic agent 64Cu-ATSM on recurrent malignant glioma: a study protocol for a phase-III, investigator-sponsored, randomized controlled trial. Japanese journal of clinical oncology, 55:543-546, 2025
19. Okada M, Suzuki E, Morizane C, Ogawa G, Sano Y, Imaoka H, Kobayashi S, Ikeda M, Okano N, Miwa H, Todaka A, Shimizu S, Mizuno N, Satoi S, Sano K, Tobimatsu K, Katanuma A, Masutomi K, Okusaka T, Ozaka M, Ueno M. Impact of clinically diagnosed liver cirrhosis in patients with intrahepatic cholangiocarcinoma treated with systemic chemotherapy: a subgroup analysis of JCOG1113. Japanese journal of clinical oncology, 55:594-602, 2025
20. Tohi Y, Yokomizo A, Kimura T, Wakabayashi M, Shiota M, Mori K, Kato T, Tsuzuki T, Kato M, Sasaki K, Kawahara T, Eto M, Nishiyama H, Kitamura H, Sugimoto M. Nomogram predicting the outcome of salvage radiation therapy for prostate-specific antigen failure following radical prostatectomy: an exploratory analysis of a randomized, multicenter, open-label, phase 3 trial (JCOG0401). International journal of clinical oncology, 30:993-1001, 2025
21. Hakozaki T, Tanaka K, Shiraishi Y, Sekino Y, Mitome N, Okuma Y, Aiba T, Utsumi T, Tanizaki J, Azuma K, Hara S, Morita R, Niho S, Yokoyama T, Toyozawa R, Horinouchi H, Okamoto I, Hosomi Y, Ohe Y. Gut Microbiota in Advanced NSCLC Receiving Chemoimmunotherapy: An Ancillary Biomarker Study From the Phase III Trial JCOG2007 (NIPPON). Journal of thoracic oncology, 2025
22. Hiraga H, Machida R, Kawai A, Kunisada T, Yonemoto T, Endo M, Nishida Y, Nagano A, Ae K, Yoshida S, Asanuma K, Toguchida J, Furuta T, Nakayama R, Akisue T, Hiruma T, Morii T, Nishimura H, Hiraoka K, Takeyama M, Emori M, Tsukushi S, Hatano H, Kawashima H, Isu K, Tanaka K, Kataoka T, Fukuda H, Iwamoto Y, Ozaki T. Methotrexate, Doxorubicin, and Cisplatin Versus Methotrexate, Doxorubicin, and Cisplatin + Ifosfamide in Poor Responders to Preoperative Chemotherapy for Newly Diagnosed High-Grade Osteosarcoma (JCOG0905): A Multicenter, Open-Label, Randomized Trial. Journal of clinical oncology, 43:1886-1897, 2025
23. Kanda S, Niho S, Kurata T, Nomura S, Kawashima Y, Iwama E, Yokoyama T, Watanabe Y, Tanaka H, Fujiwara Y, Zenke Y, Azuma K, Taniguchi H, Toyozawa R, Hosomi Y, Murakami H, Hara S, Bessho A, Yamamoto N, Ohe Y. Randomized Phase III Study of EGFR Tyrosine Kinase Inhibitor and Intercalated Platinum-doublet Chemotherapy for Non-small Cell Lung Cancer Harboring EGFR Mutation. Clinical cancer research, 31:2317-2326, 2025
24. Aoyama I, Takizawa K, Kataoka K, Ogawa G, Sano Y, Yamamoto Y, Tanaka T, Setoyama T, Kadota T, Saito Y, Nagami Y, Hikichi T, Nagata S, Urabe Y, Ono H, Doyama H, Nagino K, Tomita T, Tamaoki M, Ohno K, Kobayashi M, Abe S, Yano T, Muto M. Endoscopic balloon dilation with steroid injection versus radial incision and cutting with steroid injection for refractory esophageal anastomotic stricture: a randomized study. Endoscopy, 2025
25. Nakashima T, Suzuki T, Machida R, Shimada K, Kobayashi T, Maruyama D, Munakata W, Iida S, Ohmachi K, Kinoshita T, Ando K, Nagai H. Prognostic impact of RDI of vincristine in patients with DLBCL receiving R-CHOP: a supplementary analysis of JCOG0601. Blood neoplasia, 2:100077, 2025
26. Watanabe T, Matsuno Y, Wakabayashi M, Maruyama D, Yamamoto K, Kubota N, Shimada K, Asagoe K, Yamaguchi M, Ando K, Ogura M, Kuroda J, Suehiro Y, Tsukasaki K, Tobinai K, Nagai H. Analyzing the risk factors for disease progression within 2 years and histological transformation in patients treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone as first-line treatment: A 15-year follow-up of patients with advanced follicular lymphoma in JCOG0203. Hematological oncology, 42:e3272, 2024
27. Kataoka K, Ouchi A, Suwa Y, Hirano H, Yamaguchi T, Takamizawa Y, Hanaoka M, Iguchi K, Boku S, Nagata K, Koyama T, Shimada Y, Inomata M, Sano Y, Mizusawa J, Hamaguchi T, Takii Y, Tsukamoto S, Takashima A, Kanemitsu Y. Localized colorectal cancer database integrating 4 randomized controlled trials; (JCOG2310A). European journal of surgical oncology, 50:108354, 2024
28. Tane K, Shiono S, Wakabayashi M, Kataoka T, Mitome N, Fukuda H, Aokage K, Watanabe SI. A randomized phase III trial of postoperative surveillance for pathological stage II and IIIA non-small cell lung cancer (JCOG2012, PHOENIX). Japanese journal of clinical oncology, 54:926-929, 2024
29. Hayashi M, Yoshikawa T, Mizusawa J, Hato S, Iwasaki Y, Sasako M, Kawachi Y, Iishi H, Choda Y, Boku N, Terashima M. Prognostic Impact of Post-operative Infectious Complications in Gastric Cancer Patients Receiving Neoadjuvant Chemotherapy: Post Hoc Analysis of a Randomized Controlled Trial, JCOG0501. Journal of gastrointestinal cancer, 55:1125-1133, 2024
30. Imamura Y, Kiyota N, Tahara M, Kodaira T, Hayashi R, Nishino H, Asada Y, Mitani H, Iwae S, Nishio N, Onozawa Y, Hanai N, Ohkoshi A, Hara H, Monden N, Nagaoka M, Minami S, Kitabayashi R, Sasaki K, Homma A. Risk prediction model for cisplatin-induced acute kidney injury in patients with head and neck cancer receiving chemoradiotherapy: A re-analysis of a phase II/III JCOG1008 trial. Oral oncology, 154:106868, 2024
31. Osawa T, Sasaki K, Machida R, Matsumoto T, Matsui Y, Kitamura H, Nishiyama H. Real-world treatment trends for patients with advanced prostate cancer and renal cell carcinoma and their cost-a survey in Japan. Japanese journal of clinical oncology, 54:1062-1070, 2024
32. Imaoka H, Sasaki K, Machida R, Nagano H, Satoi S, Ikeda M, Kobayashi S, Yamashita T, Okusaka T, Ido A, Hatano E, Miwa H, Ueno M, Nakao K, Shimizu S, Kuramochi H, Sakamori R, Tsumura H, Okano N, Shioji K, Shirakawa H, Akutsu N, Tsuji K, Ishii H, Umemoto K, Asagi A, Ueno M. Current status of the cost burden of first-line systemic treatment for patients with advanced hepatocellular carcinoma in Japan, 2021-22. Japanese journal of clinical oncology, 54:1071-1077, 2024
33. Kato K, Machida R, Ito Y, Daiko H, Ozawa S, Ogata T, Hara H, Kojima T, Abe T, Bamba T, Watanabe M, Kawakubo H, Shibuya Y, Tsubosa Y, Takegawa N, Kajiwara T, Baba H, Ueno M, Takeuchi H, Nakamura K, Kitagawa Y. Doublet chemotherapy, triplet chemotherapy, or doublet chemotherapy combined with radiotherapy as neoadjuvant treatment for locally advanced oesophageal cancer (JCOG1109 NExT): a randomised, controlled, open-label, phase 3 trial. Lancet (London, England), 404:55-66, 2024
34. Shien T, Tsuda H, Sasaki K, Mizusawa J, Akiyama F, Kurosumi M, Sawaki M, Tamura N, Tanaka K, Kogawa T, Takahashi M, Hayashi N, Mukai H, Masuda N, Hara F, Iwata H. Comparison of proportions and prognostic impact of pathological complete response between evaluations of representative specimen and total specimen in primary breast cancer after neoadjuvant chemoradiotherapy: an ancillary study of JCOG0306. Breast cancer research and treatment, 208:145-154, 2024
35. Sano Y, Kanai M, Morizane C, Sasaki K, Yoshimura M, Ito Y, Furuse J, Ozaka M, Fukuda H, Ueno M. Protocol digest of a randomized phase III trial comparing S-1-based chemoradiotherapy with/without nivolumab for unresectable locally advanced or borderline resectable pancreatic cancer: JCOG1908E (PENETRATE). Japanese journal of clinical oncology, 54:1214-1218, 2024
36. Takahashi N, Seki T, Sasaki K, Machida R, Ishikawa M, Yunokawa M, Matsuoka A, Kagabu M, Yamaguchi S, Hiranuma K, Ohnishi J, Sato T. High cost of chemotherapy for gynecologic malignancies. Japanese journal of clinical oncology, 54:1078-1083, 2024
37. Kusumoto S, Munakata W, Machida R, Terauchi T, Onaya H, Oguchi M, Iida S, Nosaka K, Suzuki Y, Harada Y, Miyazaki K, Maruta M, Fukuhara N, Toubai T, Kubota N, Ohmachi K, Saito T, Rai S, Mizuno I, Fukuhara S, Takeuchi M, Tateishi U, Maruyama D, Tsukasaki K, Nagai H. Interim PET-guided ABVD or ABVD/escalated BEACOPP for newly diagnosed advanced-stage classic Hodgkin lymphoma (JCOG1305). Cancer science, 115:3384-3393, 2024
38. Tezuka S, Ozaka M, Furuse J, Yokoyama M, Uemura K, Sano Y, Nakachi K, Imaoka H, Unno M, Shirakawa H, Shimizu S, Kato N, Kojima Y, Sano K, Kobayashi S, Terashima T, Morizane C, Ikeda M, Ueno M. Early tumor shrinkage as a prognostic predictor in chemotherapy-naïve patients with locally advanced pancreatic cancer treated with modified FOLFIRINOX or gemcitabine plus nab-paclitaxel combination therapy: An exploratory analysis of JCOG1407. Pancreatology, 24:909-916, 2024
39. Nishikawa T, Kakunaga S, Tamura K, Ando M, Ozaki T, Kawai A, Ueda T, Kawasaki M, Tomatsuri S, Okamura N, Kamikura M, Hamada A, Yoshida A, Hirakawa A, Shibata T, Nakamura K, Yonemori K. Efficacy and safety of nivolumab monotherapy in patients with unresectable clear cell sarcoma and alveolar soft part sarcoma (OSCAR Trial/NCCH1510). Cancer, 130:3836-3844, 2024
40. Ohba A, Morizane C, Kawamoto Y, Komatsu Y, Ueno M, Kobayashi S, Ikeda M, Sasaki M, Furuse J, Okano N, Hiraoka N, Yoshida H, Kuchiba A, Sadachi R, Nakamura K, Matsui N, Nakamura Y, Okamoto W, Yoshino T, Okusaka T. Trastuzumab Deruxtecan in Human Epidermal Growth Factor Receptor 2-Expressing Biliary Tract Cancer (HERB; NCCH1805): A Multicenter, Single-Arm, Phase II Trial. Journal of clinical oncology, 42:3207-3217, 2024
41. Shimizu H, Kodaira T, Kiyota N, Hayashi R, Nishino H, Asada Y, Mitani H, Hirayama Y, Onozawa Y, Nishio N, Hanai N, Ohkoshi A, Hara H, Monden N, Nagaoka M, Minami S, Fujii T, Tanaka K, Homma A, Yoshimoto S, Oridate N, Omori K, Ueda T, Okami K, Uemura H, Shiga K, Nakahira M, Asakage T, Saito Y, Sasaki K, Kitabayashi R, Ishikura S, Nishimura Y, Tahara M. Incidence and risk factors associated with the development of hypothyroidism after postoperative chemoradiotherapy for head and neck cancer patients with high-risk features: Supplementary analysis of JCOG1008. Oral oncology, 157:106976, 2024
42. Watanabe K, Sasaki K, Machida R, Shimizu J, Yamane Y, Tamiya M, Saito S, Takada Y, Yoh K, Yoshioka H, Murakami H, Kitazono S, Goto Y, Horinouchi H, Ohe Y. High-cost treatments for advanced lung cancer in Japan (Lung Cancer Study Group of the Japan Clinical Oncology Group). Japanese journal of clinical oncology, 54:1084-1092, 2024
43. Shiraishi Y, Nomura S, Sugawara S, Horinouchi H, Yoneshima Y, Hayashi H, Azuma K, Hara S, Niho S, Morita R, Yamaguchi M, Yokoyama T, Yoh K, Kurata T, Okamoto H, Okamoto M, Kijima T, Kasahara K, Fujiwara Y, Murakami S, Kanda S, Akamatsu H, Takemoto S, Kaneda H, Kozuki T, Ando M, Sekino Y, Fukuda H, Ohe Y, Okamoto I. Comparison of platinum combination chemotherapy plus pembrolizumab versus platinum combination chemotherapy plus nivolumab-ipilimumab for treatment-naive advanced non-small-cell lung cancer in Japan (JCOG2007): an open-label, multicentre, randomised, phase 3 trial. The Lancet. Respiratory medicine, 12:877-887, 2024
44. Iwatani T, Sasaki K, Machida R, Shien T, Hara F, Fujisawa T, Takano Y, Kobayashi Y, Saimura M, Koizumi K, Terada M, Sasada S, Saito K, Sumiyoshi M, Iwata H. Status of incremental costs of first-line treatment recommended in Japanese clinical guidelines for metastatic breast cancer patients. Japanese journal of clinical oncology, 54:1093-1099, 2024
45. Nishina T, Boku N, Kurokawa Y, Sasaki K, Machida R, Yoshikawa T. A real-world survey on expensive drugs used as first-line chemotherapy in patients with HER2-negative unresectable advanced/recurrent gastric cancer in the stomach cancer study group of the Japan clinical oncology group. Japanese journal of clinical oncology, 54:1100-1106, 2024
46. Takashima A, Hamaguchi T, Mizusawa J, Nagashima F, Ando M, Ojima H, Denda T, Watanabe J, Shinozaki K, Baba H, Asayama M, Hasegawa S, Masuishi T, Nakata K, Tsukamoto S, Katayama H, Nakamura K, Fukuda H, Kanemitsu Y, Shimada Y. Oxaliplatin Added to Fluoropyrimidine/Bevacizumab as Initial Therapy for Unresectable Metastatic Colorectal Cancer in Older Patients: A Multicenter, Randomized, Open-Label Phase III Trial (JCOG1018). Journal of clinical oncology, 42:3967-3976, 2024
47. Yokoyama K, Wasano K, Sasaki K, Machida R, Nakahira M, Kitamura K, Sakagami T, Takeshita N, Ohkoshi A, Suzuki M, Tateya I, Morishita Y, Sekimizu M, Nakayama M, Koyama T, Shibata H, Miyamaru S, Kiyota N, Hanai N, Homma A. Frequency of use and cost in Japan of first-line palliative chemotherapies for recurrent or metastatic squamous cell carcinoma of the head and neck. Japanese journal of clinical oncology, 54:1115-1122, 2024
48. Takashima A, Ishiguro M, Sasaki K, Machida R, Nagashima F, Imaizumi J, Hamaguchi T, Yamamoto Y, Masuishi T, Asayama M, Ueno H, Shinozaki K, Kudo T, Machida N, Matsuoka H, Ishida H, Yamaguchi T, Nogami H, Yamada T, Takegawa N, Kito Y, Tonoike Y, Sawada R, Tsukamoto S, Kanemitsu Y. Real-world treatment costs of first-line treatment for metastatic colorectal cancer: a survey of the JCOG colorectal cancer study group. Japanese journal of clinical oncology, 54:1107-1114, 2024
49. Motomura K, Sasaki K, Sugii N, Yamaguchi S, Inoue H, Oshima A, Tanaka K, Otani Y, Shirahata M, Shibahara I, Nagane M, Tsuzuki S, Matsutani T, Tsukamoto Y, Kijima N, Asano K, Ohno M, Inoue A, Mineharu Y, Miyake K, Mitobe Y, Hanihara M, Kawanishi Y, Deguchi S, Saito M, Matsuda R, Ujifuku K, Arita H, Sato Y, Yamashita S, Yonezawa U, Yamaguchi J, Momii Y, Ogawa T, Kambe A, Ohba S, Fukai J, Saito N, Kinoshita M, Sumi K, Otani R, Uzuka T, Takebe N, Koizumi S, Saito R, Arakawa Y, Narita Y. Cost of medical care for malignant brain tumors at hospitals in the Japan Clinical Oncology Group brain-tumor study group. Japanese journal of clinical oncology, 54:1123-1131, 2024
50. Mitsunaga S, Ikeda M, Nomura S, Morizane C, Todaka A, Yamamoto N, Kamata K, Yanagibashi H, Mizuno N, Kawamoto Y, Gotoh K, Shirakawa H, Okano N, Nomura T, Tanaka K, Takahashi A, Yagi S, Ohta K, Takayama Y, Miwa H, Nagano H, Kojima Y, Hisano T, Tahara M, Sakuma Y, Arai H, Nakamura I, Katayama H, Konishi M, Ueno M. 5-Fluorouracil metabolic pathway genes predict recurrence risk following adjuvant S-1 therapy: Results of an ancillary analysis from a phase III trial of resected biliary tract cancer (JCOG1202A1). Journal of hepato-biliary-pancreatic sciences, 31:886-896, 2024
51. Yamamoto S, Kato K, Daiko H, Ito Y, Kajiwara T, Kojima T, Miyata H, Nakagawa S, Ueno M, Watanabe M, Tsunoda S, Abe T, Nagatani Y, Okada M, Morita M, Sato T, Mizusawa J, Nakamura K, Kitagawa Y. Factors influencing decision-making between surgery and chemoradiotherapy for esophageal carcinoma from the JCOG0502. Future oncology (London, England), 20:2849-2854, 2024
52. Imamura Y, Kiyota N, Tahara M, Kodaira T, Hayashi R, Nishino H, Asada Y, Mitani H, Iwae S, Nishio N, Onozawa Y, Hanai N, Ohkoshi A, Hara H, Monden N, Nagaoka M, Minami S, Kitabayashi R, Sasaki K, Homma A. Effect of acute kidney injury and overall survival in patients with postoperative head and neck cancer who received chemoradiotherapy with cisplatin: A supplementary analysis of the phase II/III trial of JCOG1008. Cancer medicine, 13:e70235, 2024
53. Ueno M, Morizane C, Ikeda M, Ozaka M, Nagashima F, Kataoka T, Mizusawa J, Ohba A, Kobayashi S, Imaoka H, Kasuga A, Okano N, Nagasaka Y, Sasaki M, Furuse J, Okusaka T. Phase I/II study of nivolumab plus lenvatinib for advanced biliary tract cancer (JCOG1808/NCCH1817, SNIPE). ESMO open, 9:103919, 2024
54. Sasaki K, Mizusawa J, Bando H, Nakamura K, Kataoka T, Katayama H, Fukuda H, Hara H. Consideration of factors of low accrual and methods for setting appropriate accrual periods: Japan Clinical Oncology Group study. Trials, 25:665, 2024
55. Kinoshita M, Fushimi Y, Masumoto T, Sasaki K, Sekita T, Natsume A, Wakabashi T, Komori T, Tsuzuki S, Muragaki Y, Motomura K, Saito R, Sato K, Beppu T, Takahashi M, Kuroda JI, Sonoda Y, Kobayashi K, Mishima K, Mitsuya K, Yamasaki F, Inoue A, Matsutani T, Nakamura H, Yamaguchi S, Ishikawa E, Nakaya M, Tanaka S, Ujifuku K, Uchida H, Kanamori M, Otani R, Kijima N, Nishida N, Yoshino A, Mineharu Y, Arakawa Y, Fukuda H, Narita Y. Image-based Re-evaluation of the JCOG0911 Study Focusing on Tumor Volume and Survival, Disease Progression Diagnosis, and Radiomic Prognostication for Newly Diagnosed Glioblastoma. Magnetic resonance in medical sciences, 2024
56. Komori K, Takii Y, Mizusawa J, Kanemitsu Y, Shiozawa M, Ohue M, Ikeda S, Kobatake T, Hamaguchi T, Katayama H, Fukuda H. Long-term follow-up of the conventional versus no-touch isolation technique for resection of primary colon cancer (JCOG1006): randomized clinical trial. BJS open, 8:zrae133, 2024
