Annual Report 2024
Department of International Clinical Development
Kenichi Nakamura, Kan Yonemori, Yuka Okamoto, Sarah Kruschwitz, Asuka Tsuzuki, Tomomi Hata, Kazuko Ohara, Hiroko Nakahama, Hitomi Okuma, Chiharu Mizoguchi, Rina Inagaki, De Almeida Kathleen Yasmin, Ayako Miyata, Tomozo Yamada, Celia St Clair, Yuta Maruki, Yusuke Okuma, Koichi Ogura, Kazuki Sudo, Shinji Kohsaka, Yuki Kojima, Sho Shiino, Hiroshi Yoshida, Keisuke Watanabe, Tomohiro Matsuda, Mitsumi Terada, Junya Yokobori, Nobuko Ushirozawa, Hisahiro Itoh, Arthitada Hanada, Thoopmanee Kaendiao, Suchanya Leknoi
Introduction
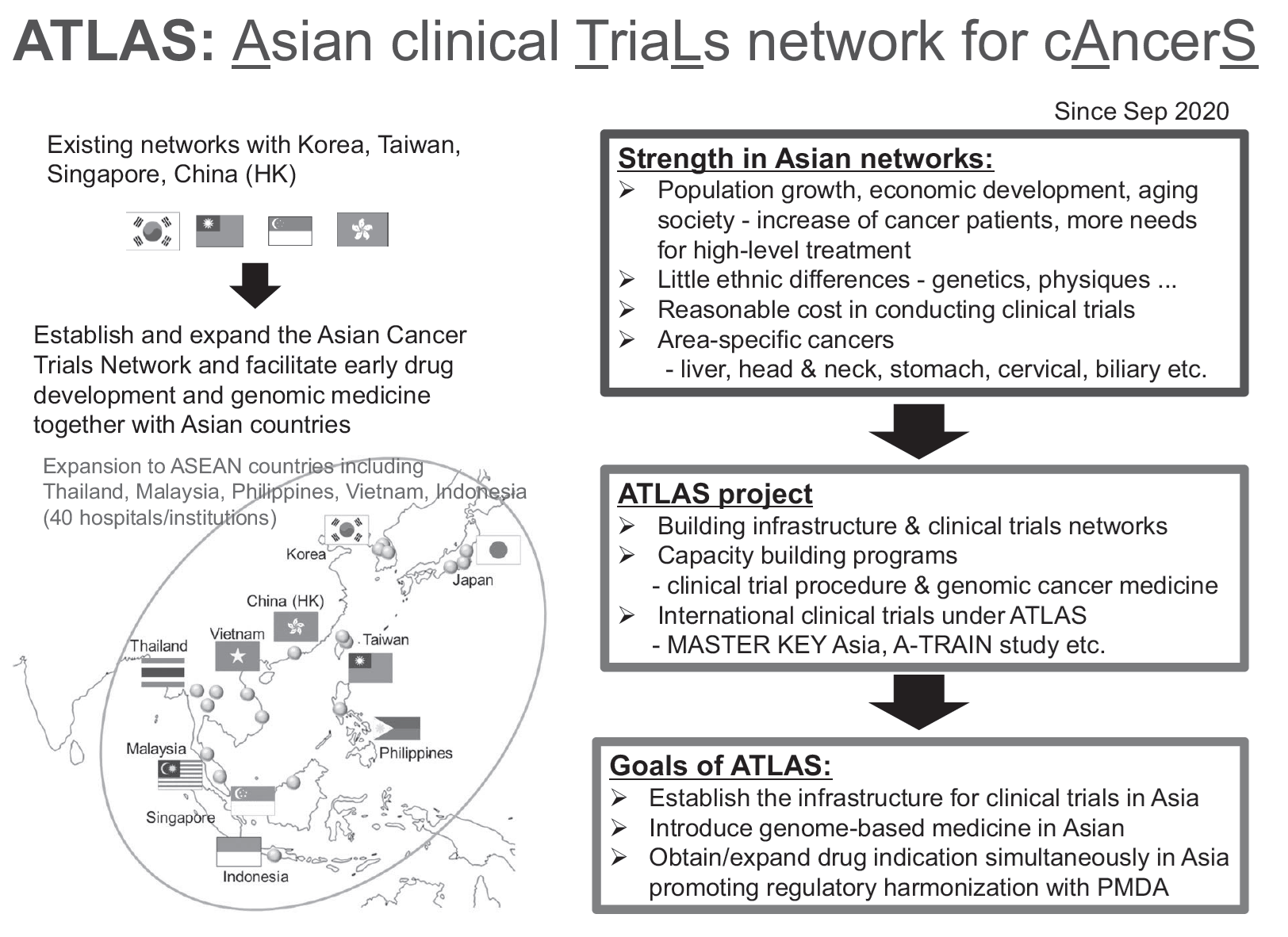
The Department of International Clinical Development (DICD) was established at the National Cancer Center Hospital in 2020 to accelerate international research, education, and treatment under an integrated vision, and to promote the Asian Clinical Trials Network for Cancers project (ATLAS project). In October 2023, the International Medical Care Section, previously part of the DICD, became an independent unit known as the Department of International Medical and Advanced Care.
The Team and What We Do
(International Research and Development Section)
In FY2024, the MASTER KEY project, a platform trial for rare cancers, was further expanded to Asian countries. In addition, a large-scale randomized controlled trial has been conducted with 6 Asian countries to evaluate the effectiveness of an AI-assisted diagnostic tool (CADe) in colonoscopy, and a total of 1,400 patients has been enrolled much earlier than expected. The section also continued internal/external call for international clinical trials in Asia.
(International Professional Education Section)
In FY2023, as part of the ATLAS project, more than 50 English contents were published on the ICRweb, and the section solicited more overseas users for the educational contents. In collaboration with PMDA, a Multi-regional Clinical Trials Seminar in January 2024 was held for participants from overseas regulatory authorities and overseas medical institutions.
(International Translational Research Section)
In FY2024, primary analyses of some cohorts were conducted in the A-TRAIN study, a TR study of liquid biopsy in which 10 Asian countries participate. In addition, the section supported HARMONY study, an observational study using MRD technique in the field of breast cancer.
(Asian Partnerships Section)
As part of the ATLAS project, the NCC Asian Partnership Office has been established in Bangkok, Thailand, which has a local coordination function for international clinical studies and strengthen the network with local investigators, governments and regulatory authorities.
Research Activities
The ATLAS Board, consisting of representative investigators from 8 Asian countries, has been held every 3 months, where new research idea and various initiatives were discussed. Additionally, disease-specific groups such as the head & neck group and the sarcoma/rare cancer group have been established under the ATLAS network. Furthermore, the activities of the ATLAS Project were presented at academic conferences both domestically and internationally.
Clinical Trials
Multiple international clinical studies are ongoing such as: PATHWAY, an investigator-initiated registration-directed trial for breast cancer; MASTER KEY ASIA, a platform study for rare cancer development; and A-TRAIN study, a TR study of liquid biopsy with 10 Asian countries; CHOICE, translational research of genomic medicine for cholangiocarcinoma; HARMONY, a translational research with MRD technique for breast cancer; and Project CAD, a large-scale randomized controlled trial which was launched across 6 Asian countries to evaluate the effectiveness of an AI-assisted diagnostic tool in colonoscopy.
Education
On-the-job training of international research support staff was made through coordination of international clinical studies. In addition, onsite training programs were offered for Asian investigators and clinical research coordinators, which included a phase 1 course, CRC course, and ISO15189 course.
Future Prospects
DICD continues to promote the ATLAS Project and increase the number of international collaborative research. To this end, we will continue to call for new international research once every six months both within and outside the hospital. In addition, the contents of the ICRweb will be enhanced, and various educational seminars will be held jointly with other Asian countries.
List of papers published in 2024
Journal
1. Mizoguchi C, Nishikawa T, Yoshida H, Yasuda M, Kato T, Hasegawa K, Yonemori K. HER2-negative or low expression as an unfavorable prognostic factor in patients with stage I/II uterine carcinosarcoma. Journal of gynecologic oncology, 36:e14, 2025
2. Hirakawa A, Asakawa T, Tokushige K, Ozaki R, Yoshida M, Okuma HS, Saito S, Shimizu Y, Kitabayashi R, Hanazawa R, Sato H, Uemura Y. Planning and Implementing Master Protocol Trials in Japan: Key Considerations of the Japanese Guideline. Clinical pharmacology and therapeutics, 117:627-632, 2025
3. Nishimura R, Sudo K, Kitadai R, Kawachi A, Ito M, Hoshino M, Kita S, Saito A, Kojima Y, Maejima A, Noguchi E, Okuma HS, Koyama T, Shimoi T, Yonemori K. Treatment and Reasons for Choosing Treatment in Breast Cancer Patients Who Underwent Next-Generation Sequencing Test. Oncology, 1-10, 2025
4. Nakamura K, Takeda K, Saito AM, Kato M, Sato S, Nakagawa S, Kawamoto Y, Oki E, Okamoto I, Okamoto H, Katayama H, Mizusawa J, Kaba H, Shibata T, Fukuda H. Transitional dynamics in oncology clinical trials: evaluating the impact of Clinical Trials Act on cooperative groups. Japanese journal of clinical oncology, 54:748-752, 2024
5. Imai T, Kojima Y, Shimoi T, Aiba H, Okuma HS, Saito A, Kita S, Yamamoto K, Maejima A, Nishikawa T, Sudo K, Noguchi E, Yoshida A, Matsui Y, Iwata S, Kobayashi E, Kawai A, Udagawa R, Fujiwara Y, Yonemori K. Predicting Trabectedin Efficacy in Soft Tissue Sarcoma: Inflammatory Biomarker Analysis. Anticancer research, 44:2125-2132, 2024
6. Chiba Y, Kojima Y, Yazaki S, Yoshida H, Takamizawa S, Kitadai R, Saito A, Okuma HS, Nishikawa T, Shimoi T, Sudo K, Noguchi E, Uno M, Ishikawa M, Kato T, Fujiwara Y, Yonemori K. Trop-2 expression and the tumor immune microenvironment in cervical cancer. Gynecologic oncology, 187:51-57, 2024
7. Mizusawa J, Ogawa G, Terada M, Ishiki H, Kikawa Y, Kiyota N. Statistical Analysis Methods and Reporting of Patient-Reported Outcomes in Randomized Controlled Trials for Cancer Conducted in Japan: A Systematic Review. Cureus, 16:e60804, 2024
8. Kitadai R, Shimoi T, Yazaki S, Okuma HS, Hoshino M, Ito M, Saito A, Kita S, Kojima Y, Nishikawa T, Sudo K, Noguchi E, Fujiwara Y, Yoshida M, Yonemori K. Clinicopathological and prognostic features of HER2-null and HER2-low advanced breast cancer treated with eribulin or capecitabine. Breast cancer (Tokyo, Japan), 31:1037-1045, 2024
9. The Selangor Consensus: strengthening clinical trials for local public health in the Western Pacific. The Lancet regional health. Western Pacific, 48:101136, 2024
