Annual Report 2024
Laboratory of Molecular Carcinogenesis
Naoto Tsuchiya, Yuko Fujiwara, Masae Oomaru, Makiko Saito, Takahiro Shirai, Rumi Tanaka
Introduction
To elucidate the nature of human cancer, it is essential to unravel the network structures established within cancer cells. Our research unit focuses particularly on microRNAs (miRNAs) among non-coding RNAs, aiming to clarify the comprehensive intracellular networks formed in cancer cells through their analysis. Building upon these findings, we are further advancing fundamental research that contributes to the establishment of novel cancer therapies and diagnostic methods, through investigations at the cellular level, in animal models, and using clinical specimens.
Research activities
(1) Development of the D-score Based on miRNA Structural Diversity and Elucidation of Its Functional Basis
Our research unit seeks to elucidate the mechanisms by which microRNAs (miRNAs) regulate gene expression and to clarify how such regulation shapes cancer-specific transcriptional profiles. By uncovering these principles, we aim to lay the foundation for novel diagnostic and therapeutic approaches in cancer medicine.
This year, we focused on the structural diversity of miRNAs, which we consider a critical layer of gene regulation in cancer. We developed a quantitative index termed the "D-score," which measures the dominance of particular structural isoforms of miRNAs. Using this approach, we demonstrated that the D-score enables precise stratification of recurrence risk groups in early-stage non-small cell lung cancer (NSCLC). Importantly, this stratification was independent of conventional clinicopathological factors, underscoring the unique predictive value of the D-score.
Further analyses revealed that the D-score correlates strongly with major malignant features. Tumors with high D-scores showed enhanced genomic instability linked to accelerated cell cycle progression, as well as gene expression profiles consistent with persistent epithelial-mesenchymal transition (EMT), a state associated with metastasis and therapy resistance. High D-score tumors were also enriched in immune-evasive microenvironments, suggesting that miRNA structural diversity not only reflects intrinsic tumor aggressiveness but also influences tumor-host interactions.
To investigate the molecular basis of these findings, we identified the oncofetal RNA-binding protein IGF2BP3 as a key regulator. IGF2BP3 was shown to modulate miRNA biogenesis, altering the balance of structural isoforms within specific miRNA subsets. We further demonstrated that these isoforms differ in their ability to repress target mRNAs, establishing a direct link between miRNA structural diversity and gene silencing efficiency. Taken together, our findings indicate that IGF2BP3-dependent control of miRNA structural diversity contributes to the establishment of cancer-specific gene expression programs (Figure 1).
Figure 1. Prediction of recurrence risk by the D-score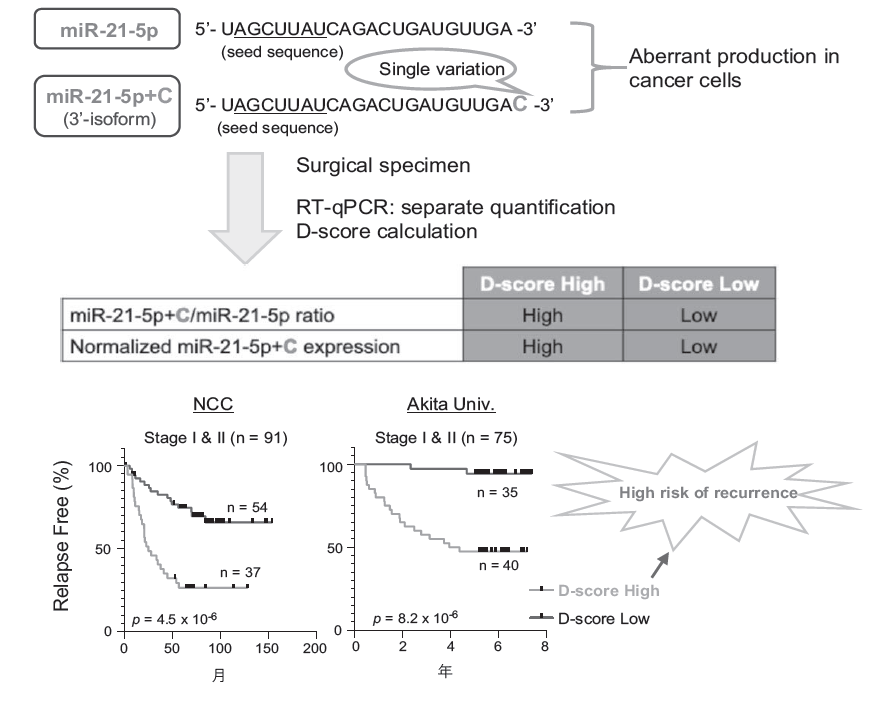

Clinically, this work carries significant implications. D-score-based stratification achieved a negative predictive value of 94% in early-stage NSCLC, making it a promising tool for identifying patients unlikely to relapse after surgery. Such high accuracy could help avoid overtreatment by reducing unnecessary adjuvant chemotherapy, while enabling risk-adapted management of high-score patients.
Recognizing this translational potential, we have initiated collaborative projects with industry partners. A multiplex detection system based on the D-score has been established and validated in clinical specimens, and preparations are underway for formal clinical performance studies. These efforts mark a key step toward the practical implementation of miRNA structural diversity as a biomarker in precision oncology.
In summary, the development of the D-score and the identification of IGF2BP3 as its upstream regulator provide novel insights into cancer biology, highlighting miRNA structural diversity as both a conceptual framework for understanding malignant transformation and a foundation for new diagnostic strategies.
(2) Mechanisms of Tumor Cell and Microenvironmental Regulation via miRNA Secretion
We found that sarcoma cells under nutrient-deprived conditions secrete specific miRNAs that enhance metastatic potential, suggesting that secreted miRNAs contribute directly to malignant phenotypes. As part of this mechanism, we identified CMTM6 as a key intracellular factor. Beyond its known role in PD-L1 recycling, we demonstrated that CMTM6 mediates intracellular transport of miRNAs and secretory molecules. Notably, CMTM6 regulates trafficking of lipid raft-associated proteins critical for receptor signaling, while also modulating extracellular secretory pathways. These findings establish CMTM6 as a unique regulator of tumor malignancy.
Based on these insights, we have begun exploring nucleic acid therapeutics targeting CMTM6, aiming to exploit its role in secretory pathways for therapeutic benefit.
(3) Development of miRNA-Based Diagnostic Kits
Building on the results from section (1), we are collaborating with industry partners to develop diagnostic kits utilizing the D-score principle. A novel detection method optimized for clinical use has been established and validated in clinical specimens, and preparations for clinical performance trials are in progress. These efforts represent a significant step toward practical deployment of miRNA-based diagnostics in oncology.
Education
We are supervising research and presentation skills for graduate students and young scientists.
Future Prospects
This year's findings suggest a new concept of miRNA functional abnormalities in carcinogenesis. Applying these principles could enable a rapid diagnostic method to identify high-risk recurrence groups in early-stage lung adenocarcinoma, serving as a biomarker to guide personalized treatment. Moreover, insights into extracellular miRNA secretion highlight opportunities for novel therapeutic development and a deeper understanding of cancer pathogenesis.
